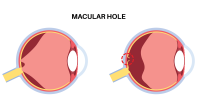
On March 29, the U.S. Food and Drug Administration (FDA) approved Blincyto (blinatumomab) to treat adults and children with B-cell precursor acute lymphoblastic leukemia (ALL) who are in remission but still have minimal residual disease.
Read more via FDA.gov.