Follow current recommendations for prevention, assessment, and treatment.
Takeaways:
- Sexually transmitted infections are epidemic in the United States with numbers steadily rising and reaching an all-time high for the sixth straight year.
- One in five Americans has an STI on any given day.
- 2021 STI Treatment Guidelines, which update the 2015 guidelines, provide clinicians with the latest evidence and important updates about risk assessment, prevention, screening, diagnostic testing, and treatment.
Sexually transmitted infections (STI) have reached epidemic levels in the United States with numbers steadily rising and reaching all-time highs for 6 years in a row. According to the Centers for Disease Control and Prevention (CDC), 68 million people (one in five Americans) have an STI on any given day. Data suggest that individuals experiencing health disparities and inequality (racial and ethnic minorities, individuals between 15 and 24 years of age, and women) bear the greatest STI burden. The CDC estimates that the U.S. spends approximately $16 billion annually in direct medical costs to treat new STIs.
In June 2019, in response to these data, the CDC collaborated with a panel of subject matter experts to conduct a systematic literature review. The 2021 STI Treatment Guidelines summarize these outcomes and provide clinicians with the latest evidence related to risk assessment, prevention, screening, testing, and treatment.
Prevention
Based on current CDC data, those at greatest risk for STIs have remained similar over the past decade. Individuals between the ages of 15 and 24 years represent 50% of all new STIs. Blacks are more likely to be diagnosed with gonorrhea, chlamydia, and syphilis than Whites, and men who have sex with men (MSM) account for more than half of syphilis cases. Each of these infections increase an individual’s risk for acquiring HIV.
Healthcare providers play a critical role in decreasing the physical, emotional, and financial burden of these infections via risk assessment and prevention steps. The CDC recommends that all providers routinely obtain sexual histories as part of the clinical encounter to assess for behavioral and biologic risks. Diagnosis of an STI is a biologic risk marker for acquisition and transmission of other STIs and HIV, so screen patients based on community prevalence. After collecting assessment data, offer risk reduction and prevention counseling in a nonjudgmental and empathetic manner.
The updated guidelines place increased emphasis on pre-exposure vaccination for hepatitis A, hepatitis B, and human papillomavirus (HPV), as well as pre-exposure prophylaxis (PrEP) and post-exposure prophylaxis (PEP) for HIV as appropriate. PrEP effectively prevents HIV in individuals with high-risk sexual behaviors, those who’ve been previously prescribed PEP, women with sexual partners who are HIV positive, and individuals who use I.V. drugs.
For patients who’ve been prescribed PEP, conduct a detailed risk assessment to determine the appropriateness of PrEP. For individuals at high risk for HIV or with possible exposure to HIV, including adolescents, discuss the availability of PrEP and PEP and make referrals for appropriate services if the provider can’t prescribe these medications.
The new guidelines also contain a section on multipurpose prevention technologies—including combined STI, HIV, and pregnancy prevention—currently under investigation. Various microbicides (including a vaginal ring) recommended by the World Health Organization are being considered for approval by the Food and Drug Administration (FDA). If approved for use in the United States, each vaginal ring will provide 3 months of protection against STIs, HIV, and pregnancy.
Screening
The 2021 guidelines update several existing screening recommendations, including for pregnant individuals, adolescents, incarcerated individuals, those at increased risk for HIV, and gender nonconforming individuals.
Pregnant individuals
Between 2014 and 2018, U.S. congenital syphilis rates increased 185%. Most states have guidelines for screening pregnant individuals, but they vary significantly. The 2021 CDC guidelines recommend screening for syphilis at 28 week’s gestation and again at delivery if the patient is high risk for syphilis or lives in a community with significant morbidity related to the infection. Maternal risk factors include multiple partners, sex with drug use, I.V. drug use, late or no prenatal care, incarceration, or unstable housing. Among the new recommendations is universal screening for hepatitis C (Hep C) and repeat screening with each pregnancy if the overall community prevalence of Hep C is greater than 1.1%. This recommendation comes in response to the increased number of pregnant individuals infected with Hep C.
Adolescents
The 2021 guidelines introduced or re-emphasized several recommendations related to adolescents. Although screening for chlamydia and gonorrhea continues to be recommended annually for all sexually active females younger than 25, the new guidelines contain language recommending anorectal and pharyngeal testing for adolescent females based on reported sexual behaviors and exposures. A study by Shannon and colleagues found that approximately half of cisgender females who tested positive for either chlamydia or gonorrhea at rectal and pharyngeal sites were negative at urogenital sites, resulting in undetected infections with urogenital testing alone. The guidelines continue to recommend annually screening adolescent MSM for rectal and pharyngeal chlamydia and gonorrhea.
Incarcerated individuals
Gonorrhea, chlamydia, and trichomonas prevalence in correctional facilities remains high. For that reason, the guidelines recommend opt-out testing for all incarcerated individuals.
High HIV risk
The 2021 CDC guidelines suggest discussing PrEP with adolescents, especially those with high-risk behaviors or those in correctional facilities. Both the CDC and the United States Preventive Task Force recommend offering PrEP to adolescents who weigh 35 kg or more and are at substantial risk of acquiring the virus. Previous HIV guidelines recommended opt-out screening at least annually for all patients in all healthcare settings; however, the new guidelines recommend increased screening based on individual exposure risk factors. In addition, the new guidelines recommend HIV screening for all individuals with genital or perianal ulcers and no previous HIV history.
Gender nonconforming individuals
The 2021 STI Guidelines introduced new screening recommendations for gender nonconforming and sexually diverse individuals. Due to the diversity of gender confirmation surgery, hormone treatment, and sexual practices, guidelines recommend screening for STIs based on anatomy as well as sexual orientation.
Testing and treatment
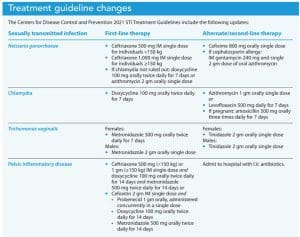
The new guidelines include updates for testing and treatment of gonorrhea, chlamydia, Trichomonas vaginalis, and pelvic inflammatory disease (PID). (See Treatment guideline changes.)

Gonorrhea
Between 2014 and 2018, Neisseria gonorrhoeae infections increased 63% overall and 137% in MSM. The 2020 CDC treatment guidelines for uncomplicated Neisseria gonorrhoeae infections in adolescents and adults are based on antibiotic stewardship and antimicrobial resistance. Studies in 2018 showed that 51% of all tested isolates were resistant to at least one antibiotic. The 2020 update, which is incorporated into the 2021 guidelines, recommends a single dose of intramuscular weight-based ceftriaxone for most patients with uncomplicated urogenital, rectal, and pharyngeal gonorrhea. This update replaces the 2015 recommendation of a single 250 mg ceftriaxone dose.
For individuals with gonorrhea in whom chlamydia hasn’t been ruled out via nucleic acid amplification testing (NAAT), treatment also should include doxycycline. An important change to the 2021 guidelines is that doxycycline is recommended only for patients who either haven’t been tested for chlamydia or for those testing positive for chlamydia in addition to gonorrhea. Cefixime remains the alternate therapy for gonorrhea, but dosages have been revised. The CDC doesn’t recommend a test of cure for uncomplicated urogenital gonorrhea; however, in patients with pharyngeal gonorrhea, a test of cure with either NAAT or culture should be completed 7 to 14 days after treatment. High rates of re-infection require that all patients treated for gonorrhea be re-tested in 3 months.
Chlamydia
Updated testing recommendations for chlamydia include FDA-approved rectal and pharyngeal site NAAT tests in MSM and women. Women with urogenital chlamydia frequently experience concomitant anorectal infections, which can’t always be predicted based on reported sexual activity due to autoinoculation.
First-line treatment for chlamydia has changed from a one-time dose of azithromycin to 7 days of doxycycline. Azithromycin is now alternate therapy as a second-line agent. These changes are based on several randomized controlled trials (RCTs) that compared doxycycline and azithromycin in treating urogenital, anorectal, and pharyngeal chlamydia. Doxycycline proved more effective than azithromycin in anorectal and pharyngeal chlamydia, resulting in the change to a first-line agent. Experts acknowledge concerns related to adherence with the 7-day regimen of doxycycline. As appropriate, they suggest prescribing azithromycin but recommend a test of cure after treatment.
Trichomonas vaginalis
Treatment guidelines for women experiencing Trichomonas vaginalis also changed. The 2015 guidelines recommended metronidazole 2 g or tinidazole 2 g orally in a single dose. Metronidazole remains the drug of choice, but dosing has changed from single-dose therapy to multi-dose therapy based on current evidence. Several RCTs suggest that in women who are HIV negative, extended therapy is more effective than single-dose therapy. In one study, multidose metronidazole decreased by 50% the proportion of women retesting positive 1 month after treatment compared to single-dose therapy.
No data exist to support changes in dosage for tinidazole, so it remains a 2 g, one-time dose and has been moved to second-line therapy for women. No data exist for multidose treatment of males; treatment remains a 2-gram single dose of metronidazole.
Pelvic inflammatory disease
Treatment guidelines for individuals with PID are based on a RCT conducted by Wiesenfeld and colleagues, which suggests that metronidazole was associated with decreased pelvic pain and faster overall symptom improvement. Anaerobic organisms have been isolated from the upper genital tract in some women with PID; in many women bacterial vaginosis is present. Neisseria gonorrhoeae and Chlamydia trachomatis, long considered the major infectious organisms, now account for less than 50% of PID diagnosed in American women
Currently, first-line outpatient PID treatment includes oral metronidazole along with ceftriaxone and doxycycline or a single dose of cefoxitin and probenecid given concurrently along with doxycycline and metronidazole. Close follow-up with oral/intramuscular therapy is necessary, and patients without improvement in 72 hours should be converted to I.V. therapy.
Stay up to date
To help decrease the physical and financial impact of STIs, healthcare providers should stay up to date with the 2021 STI Guidelines to ensure they’re following the most recent prevention, assessment, evaluation, diagnosis, and treatment recommendations. The complete 2021 guidelines are available on the CDC website along with several provider tools. An updated CDC treatment mobile app will be released soon.
Penny Thomas is an assistant professor at Southeastern Louisiana University in Hammond.
References:
AVAC: Global Advocacy for HIV Prevention. Microbicides.
Barrow RY, Ahmed F, Bolan GA, Workowski KA. Recommendations for providing quality sexually transmitted diseases clinical services, 2020. MMWR Recomm Rep. 2020;68(5):1-20. doi:10.15585/mmwr.rr6805a1
Centers for Disease Control and Prevention. Sexually transmitted infections prevalence, incidence, and cost estimates in the United States. January 25, 2021.
Department of Health and Human Services. Sexually transmitted infections: National strategic plan for the United States: 2021-2025. 2020.
Howe K, Kissinger PJ. Single-dose compared with multidose metronidazole for the treatment of trichomoniasis in women: A meta-analysis. Sex Transm Dis. 2017;44(1):29-34. doi:10.1097/OLQ.0000000000000537
Ientille G. An update on CDC STI treatment guidelines. Contemporary OB/GYN. May 5, 2021.
Kissinger P, Muzny CA, Mena LA, et al. Single-dose versus 7-day-dose metronidazole for the treatment of trichomoniasis in women: An open-label, randomised controlled trial. Lancet Infect Dis. 2018;18(11):1251-9. doi:10.1016/S1473-3099(18)30423-7
Shannon CL, Keizur EM, Fehrenbacher A, et al. Sexually transmitted infection positivity among adolescents with or at high-risk for human immunodeficiency virus infection in Los Angeles and New Orleans. Sex Transm Dis. 2019;46(11):737-42. doi:10.1097/OLQ.0000000000001056
St Cyr S, Barbee L, Workowski KA, et al. Update to CDC’s treatment guidelines for gonococcal infection, 2020. MMWR Morb Mortal Wkly Rep. 2020;69(50):1911-16. doi:10.15585/mmwr.mm6950a6
Wiesenfeld HC, Meyn LA, Darville T, Macio IS, Hillier SL. A randomized controlled trial of ceftriaxone and doxycycline, with or without metronidazole, for treatment of acute pelvic inflammatory disease. Clin Infect Dis. 2021;72(7):1181-9. doi:10.1093/cid/ciaa101
Workowski KA, Bachmann LK, Chan PA, et al. Sexually transmitted infections treatment guidelines, 2021. MMWR Recomm Rep. 2021;70(4):1-187. doi: 10.15585/mmwr.rr7004a1