The U.S. Food and Drug Administration (FDA) recently granted accelerated approval to Global Blood Therapeutics’ voxelotor (Oxbryta), a treatment for adults and pediatric patients (12 years and older) with sickle cell disease (SCD).
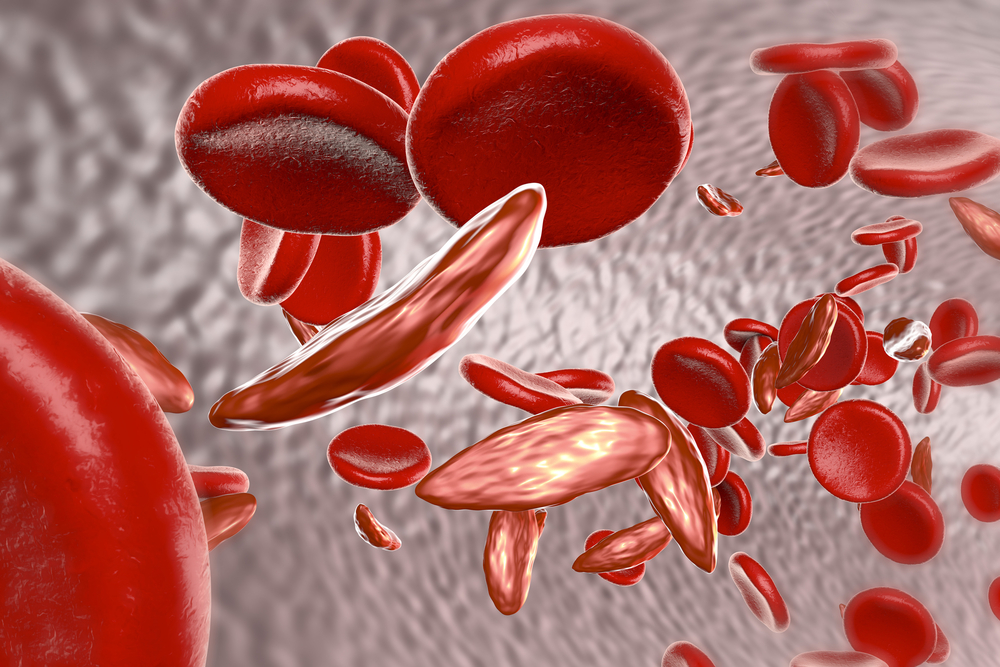
SCD is an inherited blood disorder that causes abnormally shaped red blood cells. These abnormal shapes can restrict blood flow and limit the amount of oxygen that’s delivered to tissue.
The director of the FDA’s Oncology Center of Excellence, Richard Pazdur, MD, describes the impact of the treatment: “Oxbryta is an inhibitor of deoxygenated sickle hemoglobin polymerization, which is the central abnormality in sickle cell disease… With Oxbryta, sickle cells are less likely to bind together and form the sickle shape, which can cause low hemoglobin levels due to red blood cell destruction. This therapy provides a new treatment option for patients with this serious and life-threatening condition.”
Previously, voxelotor received its fast track and orphan drug designation from the FDA, which highlights its potential impact for the SCD community.
Please read more about the approval here.