Increasing nurses’ understanding can reduce errors.
Takeaways
- Maintaining patient safety is pivotal to nursing practice; the complexities of healthcare call for more in-depth attention to recognizing adverse events, utilizing incident reporting systems, and performing root cause analysis (RCA) to improve patient care.
- An organization that promotes just culture encourages nurses and other healthcare providers to speak up when faulty processes and safety hazards are identified, and report concerns in the incident reporting system.
- It is important that institutions prioritize the communication of the findings learned from the RCA process to promote transparency on what happened and the improvements made to prevent recurrence
Nurse leaders play a pivotal role in patient safety. By ensuring an effective incident reporting system is in place and engaging staff in root cause analysis (RCA), leaders can create a collaborative environment where nurses feel free to speak up about errors and participate in finding solutions. This article discusses reporting systems and uses a case study to illustrate RCA in action.
Incident reporting system
Nurses, who are among the highest reporters of adverse events, work most closely with patients and can identify errors that warrant further investigation. Incident reporting systems provide a secure mechanism to report safety concerns about vulnerabilities in the healthcare system and serve as a repository for tracking and trending adverse events. Leaders should reinforce with staff that these tools are meant to identify safety issues, not find blame. In fact, the reports are confidential and protected from discovery in legal proceedings. Some organizations even allow anonymous reporting for those who are afraid to speak up.
Most incident reporting systems are online and are routed directly to trained staff members (including nurses, physicians, and other leaders) who review each event as appropriate for their specialty areas. Unit leaders are responsible for interviewing staff to determine what, how, and why something happened. Lessons learned are shared with other patient care areas and immediate actions aim to mitigate an adverse event and prevent recurrence. When leaders close the loop with the staff member who reported the incident, they provide reassurance that the report won’t disappear into a black hole. This action builds trust with employees and highlights that the organization values staff safety concerns.
Safety reports also can serve as baseline data to identify care deficiencies as well as drive safety, performance, and quality improvement initiatives. The organization’s patient safety team selects adverse events on which to perform in-depth reviews and, depending on the severity of the patient harm, may escalate an event to various leaders within the organization. In many cases, an RCA is performed to gain a better understanding of the system issues underlying the adverse event.
In the following case study, in which a patient receives the incorrect heparin concentration, RCA evaluates the process for system flaws that may have contributed to the event.
Case study
Mary Black*, a 75-year-old woman with a history of hypertension, is admitted for chest discomfort. She’s diagnosed with a pulmonary embolism. After the appropriate laboratory and diagnostic workups are completed, Ms. Black is started on a weight-based heparin protocol infusion. When she develops severe nose bleeds and headaches, the nurse re-assesses the patient and discovers that the incorrect concentration of heparin is infusing. The nurse stops the infusion and notifies the physician. Orders are received for lab work and to perform a brain computed tomography (CT) scan. The lab results reveal an abnormally high partial thromboplastin time (>250 seconds), and the CT scan indicates that the patient suffered a stroke. The nurse immediately transfers Ms. Black to the intensive care unit and later enters an adverse event report into the organization’s reporting system.
Root cause analysis process
An RCA provides a structured and systematic approach to evaluating errors. It’s usually led by an individual with training and experience in investigative and event analysis methodology. An RCA aims to improve patient safety, tighten system vulnerabilities, and facilitate organizational learning. Some organizations include individuals involved in the adverse event as part of the RCA process, whereas others limit participation to subject-matter experts. Critical actions to the process include interviewing the staff involved, gaining an understanding of what happened, determining what usually happens, and identifying interventions to prevent event recurrence.
Information acquired during the interviews, from observing the area’s workflow, and by mapping the sequence of events is essential to understanding what led to the adverse event. Comparing that information to the standard operating procedure or policy set by the organization uncovers the breakdown in the system or process, revealing the event’s causal and contributing factors. Causal factors have a direct cause and effect relationship and tell what caused the event to occur; contributing factors identify the issues that led to the adverse event.
Individuals closest to the work bring valuable information to recognizing safety issues and may be called to help develop solutions. When adverse events occur, rarely is there one individual or system issue contributing to the error. Organizational variables—such as staffing, environmental factors, and lack of organizational policies—signal system issues that should be addressed during the RCA process. Although each organization’s RCA process varies, the most common tools used include an event flow diagram, the 5-whys technique, and the fishbone diagram.
After reviewing Ms. Black’s chart and interviewing the staff involved, an individual skilled in RCA methodology (RCA analyst) creates a timeline and maps out the details of the event using a flow diagram (See Process mapping.) Using a fishbone diagram, the analyst identifies all the possible contributing factors (people, processes, equipment, materials, environmental) leading up to the error. (See Fishbone diagram.) The main headings can be adapted to reflect the process being analyzed. The analyst then uses the 5 whys to get to the root cause of the problem. Each question starts with a problem (Why did this problem occur?). The statement answering each why question is rephrased as a new question until no more “why” questions can be elicited. To get to the root of what caused the adverse event in this case, the analyst asks these questions:
- Why did Ms. Black develop an intracranial hemorrhage?
- Because the heparin dose was too high.
- Why was the heparin dose too high?
- Because the wrong concentration was infusing.
- Why was the wrong concentration infusing?
- Because the scan error was overridden.
- Why was the scan error overridden?
- Because the wrong concentration went unnoticed.
- Why did the wrong concentration go unnoticed?
- Because of the belief that the correct bag was dispensed from the pharmacy.
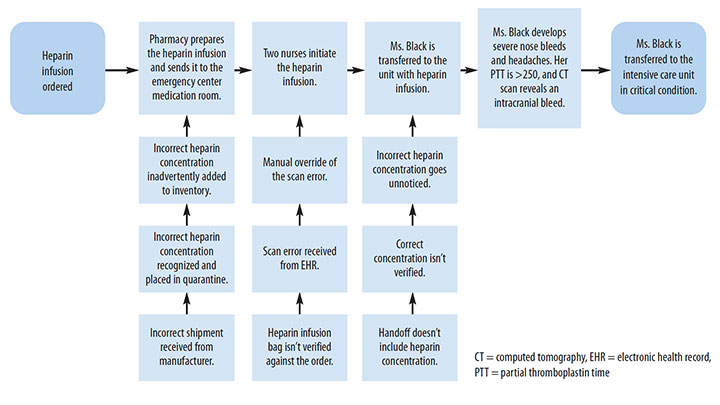
Process mapping
Use a flow chart to map a process and identify errors. This flow chart follows the process of ordering and administering heparin in the case study with Ms. Black.

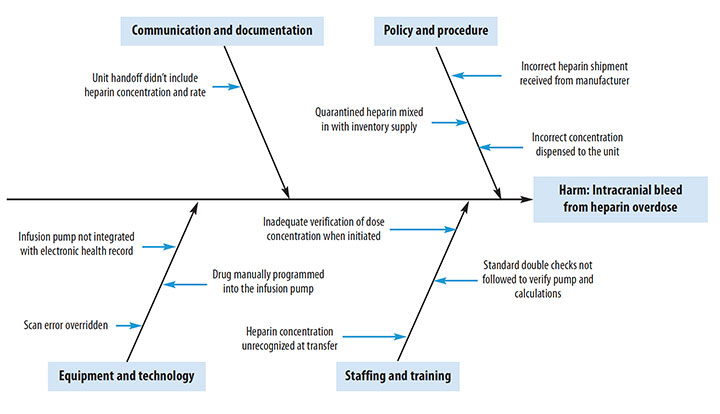
Fishbone diagram
Use a fishbone diagram to identify all factors (people, processes, equipment, materials, and environmental) that may have contributed to an error. This diagram illustrates the factors that contributed to the heparin error in the case study with Ms. Black.

A team of key stakeholders (including nurses, pharmacists, managers, and providers) participates in the RCA to discuss the event and confirm the root causes associated with the heparin overdose. Action plans, which are selected based on the RCA outcomes, should address system issues and any gaps in training and education. Meaningful and sustainable changes start with implementing actions that improve the systems in which people work. For example, adding a force function or automating a system or process can eliminate user error and mitigate future events.
In Ms. Black’s case, the RCA finds six causes behind the adverse event. Corresponding steps are taken to prevent future events.
Cause: Standardized heparin concentrations are used, but the shipment received from the manufacturer was incorrect.
Solution: Report the error to the manufacturer.
Cause: The pharmacy process caught the error, but the quarantined shipment was accidently included with the inventory supply.
Solution: Designate an area or room for quarantined shipments with clear box labeling or area signage.
Cause: The medication was correctly labeled, but the incorrect concentration went unnoticed and was dispensed to the unit.
Solution: Implement independent double-checks before dispensing high-alert drugs to the unit.
Cause: When the heparin was initiated, the scan error from the electronic health record (EHR) was overridden.
Solution: Automate smart infusion pumps with the EHR to prevent manual data entry.
Cause: Standard double checks verifying the pump and calculations weren’t followed.
Solution: Drugs requiring double checks before administration should be programmed into the EHR using a best practice advisory popup alert (BPA) to improve adherence.
Cause: The incorrect concentration was manually entered.
Solution: Implement anticoagulation stewardship to improve monitoring and surveillance.
To help ensure adherence with the solutions, units will observe or audit the double-check process. They’ll also monitor BPA reports to determine the adherence rate with documentation of the double-check process, and the organization will monitor heparin infusion error rates.
If it appears that negligence or risky behavior contributed to the event, RCA investigations can result in referrals to peer review or risk management. Just culture, which is tied closely to the RCA process, is a fair and unbiased approach to addressing systems issues rather than blaming individuals. An organization that promotes just culture encourages nurses and other healthcare providers to speak up when faulty processes and safety hazards are identified and immediately report concerns in the incident reporting system.
Implications for nurse leaders
Nurses frequently share concerns about the lack of closed-loop feedback when an adverse event is reported. Although many organizations have mechanisms for sharing best practices or lessons learned, nurses may not make connections between the incident they reported and the actions taken. To promote transparency, organizations must prioritize communicating findings from the RCA process and the improvements made to prevent recurrence, increase safety issue understanding, and enhance staff buy-in and participation in improvements. In many cases, quality improvement initiatives and policies, algorithm development, and medical equipment and device changes are a direct result of mining an organization’s reporting system or implementing corrective actions from an RCA. Leaders should make that clear to staff so they understand the importance of their reports.
Nurses are vital to identifying safety issues in their work environments. When nurses are educated about the importance of adverse event reporting, leaders can aggregate data from those reports to detect and analyze events. As champions for patient safety, nurses and nurse leaders can use incident reporting and the RCA process to help narrow the knowledge gap, encourage learning from errors, and improve safe nursing practice. AN
*Name is fictitious.
Access references at myamericannurse.com/?p=112415.
Tamu Abreu is the program manager for patient safety at MD Anderson Cancer Center in Houston, Texas.
References
Aboumrad M, Fuld A, Soncrant C, Neily J, Paull D, Watts BV. Root cause analysis of oncology adverse events in the Veterans Health Administration. J Oncol Pract. 2018;14(9):e579-90. doi:10.1200/JOP.18.00159
Aboumrad M, Neily J, Watts BV. Teaching root cause analysis using simulation: Curriculum and outcomes. J Med Educ Curric Dev. 2019;6:2382120519894270. doi:10.1177/2382120519894270
Archer S, Hull L, Soukup T, et al. Development of a theoretical framework of factors affecting patient safety incident reporting: A theoretical review of the literature. BMJ Open. 2017;7(12):e017155. doi:10.1136/bmjopen-2017-017155
Charles R, Hood B, Derosier JM, et al. How to perform a root cause analysis for workup and future prevention of medical errors: A review. Patient Saf Surg 2016;10:20. doi:10.1186/s13037-016-0107-8
Hagley G, Mills PD, Watts BV, Wu AW. Review of alternatives to root cause analysis: Developing a robust system for incident report analysis. BMJ Open Qual. 2019;8(3):e000646. doi:10.1136/bmjoq-2019-000646
Hibbert PD, Thomas MJW, Deakin A, et al. Are root cause analyses recommendations effective and sustainable? An observational study. Int J Qual Health Care. 2018;30(2):124-31. doi:10.1093/intqhc/mzx181
The Joint Commission. Root Cause Analysis in Health Care: Tools and Techniques. 6th ed. Oakbrook Terrace, IL: The Joint Commission; 2017.
National Patient Safety Foundation. RCA2: Improving Root Cause Analyses and Actions to Prevent Harm. 2015. cdn.ymaws.com/www.npsf.org/resource/resmgr/PDF/RCA2_first-online-pub_061615.pdf
Peerally MF, Carr S, Waring J, Dixon-Woods M. The problem with root cause analysis. BMJ Qual Saf. 2017;26(5):417-22. doi:10.1136/bmjqs-2016-005511
Vacher A, El Mhamdi S, dʼHollander A, et al. Impact of an original methodological tool on the identification of corrective and preventive actions after root cause analysis of adverse events in health care facilities: Results of a randomized controlled trial. J Patient Saf. 2017. doi:10.1097/PTS.0000000000000437


















1 Comment.
Excellent article!