Understand the basics so you can effectively manage your patients.
Takeaways:
- Neuraxial anesthesia is the administration of medication into the subarachnoid or epidural space to produce anesthesia and analgesia.
- An understanding of spine anatomy allows for proper assessment and management of neuraxial anesthesia.
- Prompt recognition and treatment of complications is essential to preventing a lethal outcome.
CNE
1.5 contact Hours
Learning Objectives
- Differentiate types of neuraxial anesthesia.
- Identify complications of neuraxial anesthesia.
- Discuss the nursing care of patients receiving neuraxial anesthesia.
The authors and planners of this CNE activity have disclosed no relevant financial relationships with any commercial companies pertaining to this activity. See the last page of the article to learn how to earn CNE credit.
Expiration: 4/1/23
Neuraxial anesthesia is the administration of medication into the subarachnoid or epidural space to produce anesthesia and analgesia. It can lead to the complete absence of sensory and/or motor function at or below the site of injection. Depending on the dose and concentration of the anesthetic used, neuraxial anesthesia doesn’t always result in a complete absence of motor function. For example, the goal of neuraxial anesthesia administered to a woman in labor is to provide analgesia as she progresses through active labor but not remove her ability to move her lower extremities. The three most commonly used neuraxial techniques are spinal, epidural, and combined spinal-epidural (CSE). (See When is neuraxial anesthesia used?)
Neuraxial anesthesia is used in a variety of clinical situations, including surgical, obstetric, and procedural.
Surgical
- Abdominal
- Orthopedic
- Thoracic
- Urologic
Obstetric/gynecology
- Cesarean delivery
- Labor epidural
- Tubal ligation
Procedural
- Cervical, thoracic, and/or lumbar epidural steroid injections
- Chronic pain management
A major benefit of neuraxial anesthesia is the reduced need for parenteral opioids, which have many side effects (including respiratory depression, delirium, and GI disturbances) that are associated with cardiac, pulmonary, and kidney complications. A recent meta-analysis and systematic review by Meng and colleagues found that neuraxial anesthesia is associated with a lower incidence of these complications as well as decreased blood loss and risk of thromboembolism when compared to general anesthesia. In addition, perioperative and obstetric pain management with neuraxial anesthesia has led to improved patient satisfaction scores because of enhanced pain control and shortened hospitalizations.
To ensure competent care of patients who receive neuraxial anesthesia, you should have a fundamental knowledge of spine anatomy to help you understand the important differences between spinal and epidural anesthesia administration. (See Do you know your spine anatomy?)
You also should know how to effectively monitor patient response to anesthesia, respond to complaints of pain, and address complications. Adverse events must be immediately communicated to the patient’s surgical team to mitigate harm.
Extending from the spinal cord are 31 spinal nerve roots, each of which provides sensory innervation to a dermatome (specific area of skin). For example, a patient who has knee arthroplasty and receives an epidural for postoperative pain management will have analgesia at or below sensory dermatome level L1 (first lumbar vertebrae). Other common procedures and corresponding dermatome levels include:
Dermatome level | Procedure
T4-T6 | Cesarean delivery
T8-T10 | Urologic procedures
T10 | Hip arthroplasty
T11-T12 | Knee arthroplasty
Neuraxial techniques
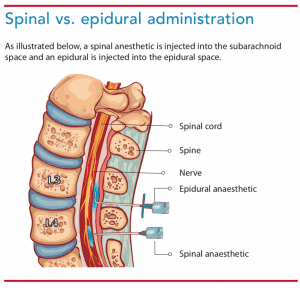
Neuraxial anesthesia can be administered via spinal, epidural, or CSE routes. (See Spinal vs. epidural administration.)
Spinal administration. To administer spinal neuraxial anesthesia and/or analgesia, a needle is inserted through the ligaments between the vertebrae and a one-time injection of medication (approximately 1 to 2 mL of local anesthesia with or without an opioid) is placed into the subarachnoid space (the area between the arachnoid and pia mater).
Epidural administration. An epidural is administered using a Tuohy needle that pierces the ligaments between the vertebrae. The needle is inserted into the epidural space (the area between the dura mater and the vertebrae). A catheter is threaded through the Tuohy into the epidural space, and an infusion of a local anesthetic with or without an opioid is initiated. Epidurals can be used to produce anesthesia but are more commonly used to provide intra- and postoperative analgesia.
CSE administration. CSE anesthesia is a two-step process. First, a Tuohy needle is placed in the epidural space. The spinal needle is then placed through the Tuohy, and a bolus of local anesthetic is administered into the subarachnoid space. After the spinal needle is removed, the epidural catheter is threaded through the Tuohy needle and its position is confirmed. Local anesthetic can be administered either as a bolus or an infusion.

How it works
Anesthesia (the complete absence of sensory and motor function) is achieved when high concentrations of local anesthetic are used, such as 0.75% bupivacaine for spinal administration and 0.25% bupivacaine for epidural administration. Analgesia (absence of pain) is achieved when a lower concentration of local anesthetic is used, such as 0.125% bupivacaine for epidural administration. Analgesic concentrations of local anesthetics are rarely used for spinal administration.
Local anesthetics work by bathing the nerve roots of the spinal cord, inhibiting sodium channel transmission to block pain signals to the central nervous system. The most commonly used local anesthetics for neuraxial anesthesia are lidocaine, bupivacaine, and ropivacaine. Opioids such as fentanyl, morphine, and hydromorphone can be coadministered with local anesthetics to produce a synergistic effect that inhibits pain transmission.
Analgesia duration depends on the type of local anesthetic used and whether it’s administered via a single injection or as a continuous infusion through epidural or CSE technique. Single spinal injections generally last 60 to 150 minutes depending on the anesthetic used; epidural administration achieves anesthesia and/or analgesia as long as the catheter remains in the epidural space and a continuous infusion of medication is running. (See How long will it last?)
Managing neuraxial analgesia
In addition to monitoring analgesic efficacy, side effects, and complications, nurses must assess the catheter insertion site, manage epidural infusions, and potentially remove an epidural catheter if a patient transitions to oral pain medications. Be sure to follow organization protocols and order sets.
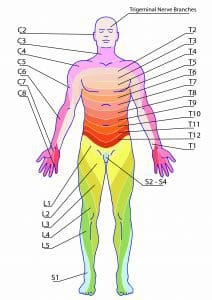
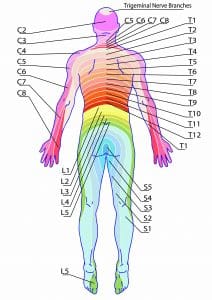
When caring for a patient who’s received neuraxial anesthesia, you’ll closely monitor his or her blood pressure (BP), heart rate (HR), pulse oximetry, respiratory rate, pain perception, and level of consciousness. Use a dermatome map to assess the level of sensory block and determine current motor function to establish a baseline level of analgesia. (See About dermatomes.) The American Society of Anesthesiologists recommends that nurses follow a patient-specific protocol established either by the patient’s care team or the organization’s policy.
If a catheter is present, assess the insertion site. Also assess the infusion system and tubing to identify any detached connections, malfunctioning infusion pumps, or medication errors. Ensure catheters are clearly labeled, and avoid using tubing with any type of injection ports to limit the risk of inadvertently administering a medication intended for I.V. injection into the site. You’ll also want to be vigilant for inadequate analgesia and complications.
Do you know your spine anatomy?
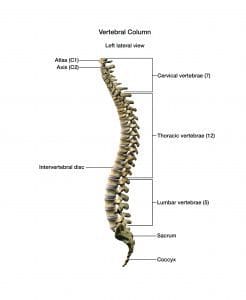
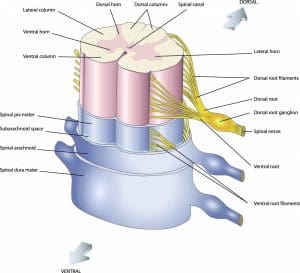
The spine is made up of the vertebrae, spinal cord, and meninges.
Vertebrae
The spine has 33 vertebrae, which are divided into six sections:
- cervical (seven vertebrae)
- thoracic (12 vertebrae)
- lumbar (five vertebrae)
- sacral (five vertebrae)
- coccygeal (four vertebrae)
Ligaments located between and around the vertebrae provide stabilization. Neuraxial anesthesia is delivered via specialized long needles that pierce the ligaments.
Spinal cord
The spinal cord, which extends from the brainstem to the lumbar region, is a thin, cylindrical tube of nerve tissue enclosed by the vertebrae. Extending from the spinal cord are 31 spinal nerve roots, each of which provides sensory innervation to a dermatome (specific area of skin). For example, a patient who has knee arthroplasty and receives an epidural for postoperative pain management will have analgesia at or below sensory dermatome level L1 (first lumbar vertebrae). Other common procedures and corresponding dermatome levels include:
| Dermatome level | Procedure |
| T4-T6 | Cesarean delivery |
| T8-T10 | Urologic procedures |
| T10 | Hip arthroplasty |
| T11-T12 | Knee arthroplasty |
T = thoracic
Understanding how to assess a dermatome level is crucial to determining the adequacy of neuraxial anesthesia and whether a patient is at risk for complications.
Meninges
Three layers of thin membranes (meninges) encapsulate the spinal cord. The dura, arachnoid, and pia mater are the outer, middle, and inner meningeal layers, respectively.
Complications
Prompt recognition and treatment of complications related to neuraxial anesthesia are essential in preventing lethal outcomes.
Inadequate analgesia. If a patient who received a spinal anesthetic begins to complain of worsening pain, parenteral or oral analgesia may be required. If an epidural catheter is present, assess the catheter, the insertion site, and the level of dermatomal block. If an infusion hasn’t been started, initiate it to treat the pain per the anesthesia provider’s order. For infusions already running, the provider may increase the infusion rate. Instruct patients with patient-controlled epidural analgesia (PCEA) how to use the bolus option to control pain. All changes to the infusion should be guided by a clearly written anesthesia provider order. The most common medications used for PCEA are bupivacaine 0.625%-0.25% and ropivacaine 0.625%-0.25% (either drug also may be given with fentanyl, morphine, or hydromorphone). Both are typically administered at a basal infusion rate of 4 to 10 mL/hr and a bolus dose of 3 to 6 mL. The lockout interval is usually 10 to 15 minutes, with the maximum hourly dose 22 to 34 mL.
If the patient continues to complain of inadequate analgesia even after the infusion rate is increased, recheck the infusion system and tubing and ensure the catheter hasn’t migrated (the distance of the catheter from the insertion site should be marked on insertion) or been dislodged. Follow provider orders for breakthrough pain, bolus dosing, and infusion pump settings related to an inadequate or absent level of analgesia. Notify the anesthesia provider if the patient doesn’t experience any improvement in pain.
Hypotension and bradycardia. Neuraxial anesthesia can cause variable BP decreases 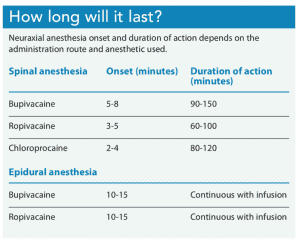

Decreased BP with evidence of hypoperfusion, such as mental status changes, requires prompt treatment. Place the patient in the supine position. If the patient has adequate cardiac and renal function, the provider may order a fluid bolus. Patients who don’t respond to these initial treatments will require vasopressor or inotropic agents, such as ephedrine, phenylephrine, or epinephrine. Treat bradycardia (HR less than 60 beats per minute) as ordered with glycopyrrolate or ephedrine if mildly symptomatic, or atropine if severe.
Respiratory depression. Neuraxial anesthesia can affect the diaphragm and accessory muscles of respiration, resulting in an impaired cough reflex that diminishes the patient’s ability to clear secretions. This effect is more noticeable in patients with preexisting pulmonary disease.
Opioids (particularly morphine) administered into the subarachnoid or epidural space have a duration of action up to 24 hours. In that time frame, any additional parenteral or oral opioids can amplify side effects. Monitor for signs of respiratory depression such as bradypnea (less than eight breaths per minute), desaturation (pulse oximetry less than 90% on room air), grunting, or airway obstruction. If respiratory depression occurs, stop any analgesic infusion and administer oxygen via a face mask at a minimum of 6 L/minute. Elevate the head of the bed to 45 degrees or higher, notify the anesthesia provider, and consider administering naloxone per provider order.
Nausea and vomiting. Nausea and vomiting secondary to neuraxial anesthesia may be attributed to hypotension, neuraxial opioids, or GI hyperperistalsis. Aggressively treating hypotension frequently prevents nausea and vomiting. For symptoms unrelated to hypotension, administer antiemetics as ordered by the provider. Small doses of anticholinergics, such as atropine or glycopyrrolate, also may be ordered.
Urinary retention. Anesthetic block of the lumbar and sacral nerve roots leads to temporary loss of bladder function and an inhibited void reflex. If a urinary catheter isn’t present, assess the patient for bladder distension until he or she is able to void. The incidence of urinary retention has been reported to be as low as 5% and as high as 70%; however, urinary retention may be influenced by factors beyond neuraxial anesthesia, including urology surgery and whether I.V. opioids were used during surgery.
Pruritis. Incidence of pruritis related to opioid administration via spinal or epidural routes ranges from 69% to 83%. Opioid antagonists (such as naloxone) or mixed opioid agonist-antagonists (such as nalbuphine) can effectively treat opioid-induced pruritis. Diphenhydramine also may be effective.
Postdural puncture headache (PDPH). PDPH is a positional headache caused by cerebral spinal fluid (CSF) leakage through a dural puncture. A PDPH is most common after a “wet tap” in which the anesthesia provider inadvertently punctures the dura with the Tuohy needle. A recent meta-analysis by Choi and colleagues showed that although the risk of a dural puncture from a Tuohy needle is only about 1.5%, a greater than 50% chance exists that patients will develop PDPH from that puncture. The constant, throbbing headache typically is located in the frontal or occipital regions. Hallmark symptoms include a headache that worsens with sitting or standing but improves when supine, neck stiffness, visual changes, subjective hearing loss, nausea, and vertigo.
Notify the anesthesia provider if you suspect PDPH. Encourage patient bedrest in the supine position, oral hydration, and increased caffeine consumption. Administer oral analgesics such as acetaminophen, butalbital, or caffeine as ordered and antiemetics as needed. For debilitating PDPH that doesn’t respond to conservative treatment, the anesthesia provider may place an epidural blood patch (injection of a small amount of autologous blood into a patient’s epidural or spinal space to stop a CSF leakage) after patient consent.
High regional block/total spinal anesthesia. A high block or total spinal anesthesia can occur when large doses of local anesthetic are injected via a catheter that’s mistakenly placed in the intrathecal space or migrates after placement. High block symptoms have a rapid onset (usually less than 60 seconds) and include nausea, dyspnea, hypotension, bradycardia, and weakness and numbness in the upper extremities. A total spinal presents with unconsciousness, apnea, and profound hypotension. Notify the anesthesia team immediately and prepare for possible advanced cardiac life support (ACLS) interventions. The incidence of a high regional block or total spinal anesthesia is approximately 0.02%.
Systemic toxicity. Large volumes of local anesthesia can mistakenly be injected into a blood vessel during epidural placement, causing the anesthetic serum level to rise above a toxic threshold and resulting in local anesthesia systemic toxicity (LAST). LAST, which is rare (less than 0.2%), affects the neurologic and cardiovascular systems and may be fatal if treatment is delayed. Early symptoms include tinnitus and a metallic taste in the mouth. Signs of toxicity can rapidly progress to seizure, loss of consciousness, hypotension, arrhythmia, and circulatory collapse. Notify the anesthesia team and prepare for possible ACLS interventions. Lipid emulsion therapy, administered by the anesthesia provider, also is indicated for LAST. Become familiar with where and how to obtain lipid emulsions because timely administration is essential to reduce the risk of cardiac arrest.
Epidural and spinal hematoma. Epidural and spinal hematomas are among the rarest neuraxial anesthesia complications (incidence ranges from 1 in 150,000 to 1 in 220,000). Patients with a hematoma usually will complain of sudden sharp back pain (from the insertion site) that radiates to the leg. Generalized weakness along with bladder and bowel dysfunction also may indicate a hematoma. Because a definitive diagnosis can be achieved only with magnetic resonance imaging and computed tomography, you should have a high degree of suspicion when patients suddenly complain of these symptoms and alert the anesthesia team immediately. Hematomas will be surgically evacuated.
Neuraxial anesthesia and antithrombotic therapy
Some surgical patients take antithrombotic therapy (for example, warfarin, clopidogrel, and aspirin) to treat conditions such as venous thromboembolism or atrial fibrillation or, in the case of those with cardiac stents and/or mechanical heart valves, as a preventive measure. The decision to proceed with neuraxial anesthesia for surgery in patients receiving antithrombotic therapy is based on an assessment of the risks of bleeding and thrombosis when therapy is discontinued. Failure to discontinue antithrombotic medications before neuraxial anesthesia administration predispose these patients to the risk of a spinal or epidural hematoma.
Instruct surgical patients to stop taking antithrombotic medications before the day of surgery. (The time frame for stoppage depends on the medication.) After surgery, monitor patients for signs of a spinal or epidural hematoma and ensure they resume antithrombotic medications per provider orders. Some patients will require postoperative bridge therapy (for example, low molecular weight heparin [such as enoxaparin] or unfractionated heparin). The American Society of Regional Anesthesia and Pain Medicine has developed guidelines for when to remove neuraxial catheters and when to re-initiate antithrombotic medications to reduce bleeding risk. The University of Washington has a an excellent reference that can be found online at bit.ly/3bwqBin.
Ensuring good outcomes
Proper nursing care and management helps ensure good outcomes for patients who receive neuraxial anesthesia. Prompt identification and communication of complications and adverse events coupled with swift intervention can mitigate harm and ultimately prevent permanent injury.
*Name is fictitious.
References
American Society of Anesthesiologists Committee on Pain Management. Statement on the role of registered nurses in the management of continuous regional analgesia. October 17, 2018. asahq.org/standards-and-guidelines/statement-on-the-role-of-registered-nurses-in-the-management-of-continuous-regional-analgesia
Association of Anaesthetists. Best Practice in the Management of Epidural Analgesia in the Hospital Setting. November 2010. aagbi.org/sites/default/files/epidural_analgesia_2011.pdf
Brull R, MacFarlane AJR, Chan VWS. Spinal, epidural, and caudal anesthesia. In: Miller RD, ed. Miller’s Anesthesia. 8th ed. Philadelphia: Saunders; 2015: 1684-720.
Butterworth JF, Wasnick JD, Mackey DC. Spinal, epidural, & caudal blocks. In: Morgan & Mikhail’s Clinical Anesthesiology. 6th ed. New York: McGraw-Hill; 2018: 959-96.
Horlocker TT, Vandermeuelen E, Kopp SL, Gogarten W, Leffert LR, Benzon HT. Regional anesthesia in the patient receiving antithrombotic or thrombolytic therapy: American Society of Regional Anesthesia and Pain Medicine evidence-based guidelines (4th ed). Reg Anesth Pain Med. 2018;43(3):263-309.
Meng T, Zhong Z, Meng L. Impact of spinal anesthesia vs. general anesthesia on peri-operative outcome in lumbar spine surgery: A systematic review and meta-analysis of randomised, controlled trials. Anaesthesia. 2017;72(3):391-401.
New York School of Regional Anesthesia. Spinal anesthesia. nysora.com/techniques/neuraxial-and-perineuraxial-techniques/spinal-anesthesia



















7 Comments.
Very organized notes,I like it
Very well written article for nurses. I loved the graphics and the simple explanations. Thanks so much for taking the time to write this!
I took the test on Overview of Neuraxial anesthesia online on 5/31/20 and successfully passed it. I need to get a copy of my CEU. Please assist me with that. Thank you
Interesting and informative article making it very easy to understand
Interesting and informative.
Very infornative!
Very interesting read.