Tips for a hospital sepsis prevention program
Takeaways:
- The Surviving Sepsis Campaign international consensus guidelines summarize best practices based on the medical literature, but they don’t illustrate how to customize implementation to specific organizations.
- This organization shares how it individualized its sepsis program infrastructure.
- Other organizations can use this information to guide their own programs.
Improvements in sepsis care have made “good catches” more common than 20 years ago, but sepsis remains a leading cause of inpatient death. Carefully developed processes, protocols, and program infrastructure increase the likelihood of a successful outcome. The Surviving Sepsis Campaign (SSC) international consensus guidelines for sepsis summarize best practices based on the medical literature, but they don’t illustrate how to customize implementation to specific organizations. Consider using the following guiding principles (and examples from our experience) to develop an individualized sepsis program infrastructure for your organization.
Executive sponsorship
Sepsis improvement teams must be interprofessional and formulated via a thorough stakeholder analysis to ensure the most effective representation and support from team members. Although the team may be tempted to bring improvement ideas straight to the bedside and clinicians, begin by mapping out a strategic plan. First, establish an administrative or executive sponsor for support when navigating barriers, such as managing stakeholders, justifying equipment needs, or obtaining resources. Executive sponsors also can be helpful when promoting initiative support to reinforce organizational commitment.
Our experience
Our sepsis improvement team used DMAIC (Define, Measure, Analyze, Improve, and Control) Six Sigma methodology to design and launch the strategic plan for the initiative. We developed a team charter that included project scope, targeted outcomes, deliverable goals, and required resources. The charter also helped affirm which members and leaders would serve on the team as well as their roles and responsibilities. This helped mitigate role confusion and clarified expectations to avoid duplication of effort and oversight. Team members also were expected to take information from team meetings and disseminate key information back to their own units.
Nurse screening
Since 2016, the SSC guidelines have recommended that organizations consider a screening process for early sepsis detection. Generally, the physiologic screening criteria should enable early detection.
But challenges such as alarm fatigue can make screening difficult. Fortunately, electronic triggers and machine learning algorithms are available in many electronic health record (EHR) systems to help reduce alarm fatigue and mitigate the “white noise” some alarm systems create.
Another challenge is that nurses may think they’re expected to diagnose the patient. However, screening isn’t diagnosing. It’s simply evaluating whether certain criteria are met that warrant reporting to the provider. A new positive screen also suggests that the patient’s status is worsening, which would likely be reported to the provider, anyway.
Our experience
Our EHR continuously monitors for sepsis criteria, firing an electronic alert when systemic inflammatory response syndrome (SIRS) and organ dysfunction criteria are met. A “remind me later” button can be clicked if the nurse is performing a more urgent clinical task. However, when sepsis criteria are met, the sepsis response system is activated with a bedside in-person response by the sepsis nurse within 15 minutes. If the sepsis nurse validates that criteria are met, the sepsis bundle is initiated as soon as possible and within 1 hour. Our organization uses a predictive electronic trigger that checks more than 70 pre-existing variables in the EHR.
Response system
Sepsis screening processes can aid early identification, but they must be coupled with response systems to ensure interventions are implemented at the bedside in a timely manner. Sepsis is a time-sensitive condition. According to Seymour and colleagues, septic shock mortality increases by 7% to 8% for every hour of delay in antibiotic administration. The response system may consist of a team that includes a nurse with critical care training, or it may be a modified version of the rapid response team. However the team is constructed, it must be sent to the patient wherever they are in the organization.
A response team can help add a sense of urgency to the situation and may help manage any nurse-provider disagreement about the right course of action. You can customize the members of your response team based on the resources and needs of your organization.
Our experience
Our sepsis alert can be called by any clinician, and a provider order isn’t required. When the code line is called, the operator notifies the team via a paging distribution list. When activated, the team makes every effort to contact the primary provider. However, our hospital intensivist is prepared to intervene as a contingency plan or if clinical needs are more time-sensitive.
Collaboration and performance metrics
Collaboration must occur at the bedside and with documentation. For example, the bedside nurse plays a critical role in the surveillance, monitoring, and early identification of patients who may meet sepsis criteria. However, all of the interventions included in the sepsis bundle require a provider’s order, which requires strong nurse–provider collaboration to increase the likelihood of early identification and timely treatment.
Accurate performance metrics depend on precise documentation and coding. Collaboration with coders and provider documentation specialists can help ensure that the correct severity of illness (SOI) and risk of mortality (ROM) are reflected for the patient. For example, a patient with a history of heart failure who develops pneumonia with hypotension may have SOI and ROM more accurately captured if a diagnostic code of “sepsis” or “severe sepsis” as opposed to “heart failure” is used, as appropriate. The sepsis team also may consider coaching the top ten providers who demonstrate documentation opportunities based on a hospital coder needs assessment.
Our experience
We consider the coding team as members of the sepsis team who can help improve outcomes by creating a more accurate denominator of cases, providing new perspectives, and helping to give the team credit for the excellent care that we know they give.
Hour-1 bundle
In 2018, the SSC issued a guideline update reinforcing the hour-1 bundle to expedite implementing the interventions previously followed in the 3- and 6-hour bundles. Although the hour-1 bundle is controversial in the medical community (from a regulatory standpoint, it would be difficult to hold clinicians accountable for 1 hour), few argue that, in theory, it’s a worthy goal. Completing every element of the sepsis bundle within 1 hour may not be possible, but focus should be placed on attempting to initiate each component in 1 hour if possible. The guiding principle of the hour-1 bundle is similar to the golden hour in trauma, acute stroke, and acute myocardial infarction.
Our experience
In 2014, our organization believed that the 3- and 6-hour bundles didn’t reinforce an ideal sense of urgency, so we implemented what we called “6 things in 60 minutes” to emphasize the time-sensitive nature of resuscitation (See 6 things in 60 minutes.) We promoted this internal goal with the slogan “Time is tissue” to illustrate the clinical significance of the sepsis-induced tissue hypoxia that may be mitigated by promptly implementing the sepsis bundle.
6 things in 60 minutes
The sepsis bundle used in our project—characterized by the slogan
“6 things in 60 minutes”—is based on the Surviving Sepsis Campaign guidelines. Our goal was to at least initiate each of these interventions within 1 hour.
Sepsis bundle
- Measure lactate level. Remeasure if initial lactate is >2 mmol/L.
- Obtain blood cultures before administering antibiotics.
- Administer broad-spectrum antibiotics.
- Begin rapid administration of 30 mL/kg crystalloid solution for lactate ≥4 mmol/L and/or hypotension.
- Start vasopressors if the patient is hypotensive during or after fluid resuscitation to maintain mean arterial pressure ≥65 mmHg.
- Repeat the focused exam (after the initial fluid resuscitation) by a licensed independent practitioner or conduct a dynamic assessment of fluid responsiveness, such as stroke volume, with passive leg raise or fluid challenge.
Sepsis bundle considerations for patients with COVID-19
- Sepsis identification criteria can be applied to patients with COVID-19 the same as for patients with sepsis from other sources.
- For patients with COVID-19 and acute hypoxemic respiratory failure on oxygen, maintain SpO2 no higher than 96%. For patients refractory to conventional oxygen therapy, high-flow nasal cannula is suggested.
- The clinical significance and process for measuring and trending serum lactate applies the same for sepsis from other sources.
- Because of the risk of respiratory failure and acute respiratory distress syndrome (ARDS), the provider may select a conservative over a liberal fluid management strategy.
- For adults with severe COVID-19 who don’t require mechanical ventilation, I.V. remdesivir is suggested, ideally within 72 hours.
- For adults with severe or critical COVID-19, a short course of systemic corticosteroids is recommended.
- For mechanically ventilated adults with COVID-19 and moderate- to-severe ARDS, prone ventilation for 12 to 16 hours is suggested.
- For adults hospitalized with COVID-19, pharmacologic venous thromboembolism prophylaxis is recommended.
Sources: Alhazzani et al 2020, Alhazzani et al 2021, Dellinger et al 2017, Levy et al 2018.
Learn more about maintaining a successful sepsis prevention program during a pandemic at myamericannurse.com/?p=74273.
Lactate measurement
No biomarker exists to diagnose sepsis, but the serum lactate level can help determine severity and aid prognosis. The higher the lactate value, the worse the prognosis. The lactate value also may help identify sepsis sooner than traditional vital signs can, and observed decreases in lactate can help evaluate treatment response.
The question of whether to sample arterial vs. venous lactate or point-of-care vs. laboratory-processed lactate can be controversial, but whatever the method used, the lactate value indicates tissue hypoxia and is a more accurate measure of disease severity compared to a physical exam or urine output. According to SSC, the goal is lactate measurement and associated treatment initiation within 1 hour.
Our experience
At our organization, a key process improvement was implementing “repeat lactate” as a standing order. This feature allows nurses to order and acquire a repeat lactate as long as sepsis is diagnosed and treatment is initiated. The process provides nurses with more autonomy and increases the likelihood of achieving bundle compliance by acquiring the repeat lactate within the first 4 to 6 hours of resuscitation.
Education and training
Education and training are primary components to any strong sepsis program. Simulation is a best practice, when possible or appropriate, to promote retention and knowledge transfer. However, facilities, space, staff, equipment, supplies, cost, and other limitations may create barriers to launching simulation training for any clinician on the team who’s likely to care for patients with sepsis. Leaders frequently are challenged to be creative when spearheading education that’s efficient, cost-effective, and focused on creating the behavioral changes likely to improve patient outcomes. Our hospital was faced with similar limitations and focused on innovative solutions to navigate these common constraints, while also generating a return on our investment.
Our experience
With a financial grant from the hospital’s foundation, our team launched a web-based, virtual simulation program (NovEx™—Novice to Expert Learning) based on SSC guidelines. The program includes case scenarios (based on real-life cases) in either a virtual emergency department, intensive care unit, or unit environment. (See Virtual simulation).
Virtual simulation
In the virtual simulation training used in our organization, a staff member receives a shift handoff and enters and navigates a virtual patient room. The scenarios provide no prompting, so the learner needs to decide what to do. Trainees can navigate the electronic health record or click on anything in the room to obtain more information (such as laboratory data, intake and output, patient history, physical, and assessment data). They also can talk with the patient and check the bedside monitor for sepsis criteria. After evaluating the patient information, participants choose treatments and interventions, for example, by clicking on the lactate machine to check a serum lactate or on the medication cart to administer fluids, vasopressors, or antibiotics.
The program doesn’t prompt case completion in response to interventions or treatments, which allows the participant to undertreat or overtreat the patient before exiting the virtual patient room. This adds a level of realism that quizzes or slideshow-style e-learning modules can’t.
Learners receive instant feedback about their performance based on customizable thresholds for a passing score created by the nursing leadership team. Administrator access allows leaders to see which staff are logged in, their completion rates, the amount of content clicked, and who might require remediation.
The simulations provide experiential learning that reinforces the guiding principles for identifying and treating sepsis. The uniqueness of the simulation experience helped generate a high level of sepsis awareness leading up to the kick-off of the screening and response system. Sepsis simulations are now mandatory for all nurses and patient care technicians as part of the new-employee orientation process.
Data collection
Many conventional sepsis quality improvement programs rely solely on retrospective data collection from the quality department, frequently for patients with the sepsis diagnosis code, to measure team performance and drive process improvement. Although retrospective data can allow for more robust data collection, it’s limited because patient coding can take anywhere from 4 to 6 weeks post-discharge to generate the sample for abstraction. Concurrent, prospective data collection can provide a real-time indicator of sepsis protocol adherence.
Our experience
In our organization, we drafted a paper flowsheet that included the key interventions requiring completion or consideration during the resuscitation period. This flowsheet has been converted to an electronic storyboard tracking tool in our EHR. A flowsheet tool such as this can be customized for any organization’s needs and serve several purposes during workflow, such as a resuscitation and responder checklist, responder-to-responder communication tool, bedside reference, handoff tool, project leader tracking tool, sepsis care plan or care pathway, scripting or care goal resource, and real-time data collection tool.
The reverse-side of the paper flowsheet includes a list of commonly administered, Centers for Medicare and Medicaid Services (CMS)–approved, I.V. sepsis antibiotics from our formulary. The paper tool was particularly helpful during the initial program launch or when sepsis program changes or barriers occurred. It provided a care summary so the sepsis project leader could follow-up with frontline clinicians for feedback or coaching opportunities. The tool encourages sepsis responder awareness, adherence to expectations, and decreased care variation and helped serve as a method for staff to actively participate in data collection and quality. It also included scripting to help guide responders through challenging situations, such as negotiations with providers about implementing the sepsis bundle.
Daily rounding and rapid feedback
A successful sepsis program can’t be launched with tools alone. To improve patient outcomes and meet team and organizational goals, tools must be incorporated into a strong process. For example, the prospective rounding tool should be used to provide rapid, real-time feedback to frontline caregivers. Caregiver behaviors or habits are less likely to change without transparency and feedback, which should be detailed and given soon enough after the patient case so the clinician remembers the patient (face-to-face feedback is preferred, although email feedback can be helpful).
Ideally, feedback should be provided by a trusted subject matter expert (SME) so that it’s viewed as credible, creates accountability, and incentivizes staff. The SME can use tools to guide their patient and staff rounds. When SMEs participate in daily rounding, they can evaluate the current sepsis screening and response system workflow, as well as monitor for ongoing strengths, barriers, or opportunities. Positive reinforcement for expectation adherence or above-and-beyond performance also can be given during these rounds.
SME presence on rounds can help empower staff to ask questions or voice concerns, creating opportunities for relationship-building. Perhaps most importantly, SMEs serve as a living, breathing portrayal of the sepsis program. This helps create daily awareness, accountability, and momentum for the program.
Our experience
A clinical nurse specialist was important not only for daily rounding, but also for spearheading the improvement of patient outcomes through project management, advanced education and training, unit presence as a clinician, and implementation of evidence-based practices.
Continuous quality improvement tools
Sustaining performance and outcomes is the true challenge of a sepsis program. Tools—process maps, workflow algorithms, role grids, operations manual, electronic feedback tools, and report cards—developed in collaboration with the entire clinical team can help. These tools aid consistency, create built-in redundancy, decrease practice variation, support teaching and coaching, and hold team members accountable. They also can be customized based on feedback from the team to navigate barriers and process problems.
Our experience
Based on input from frontline staff, we added piperacillin/tazobactam to our automated medication delivery system so nurses had it on the unit instead of having to wait for a pharmacist to prepare and send it. This change allows us to deliver the antibiotic more quickly. In addition, we use role grids to help define roles and expectations for every member of the sepsis resuscitation team.
Outcomes improvement
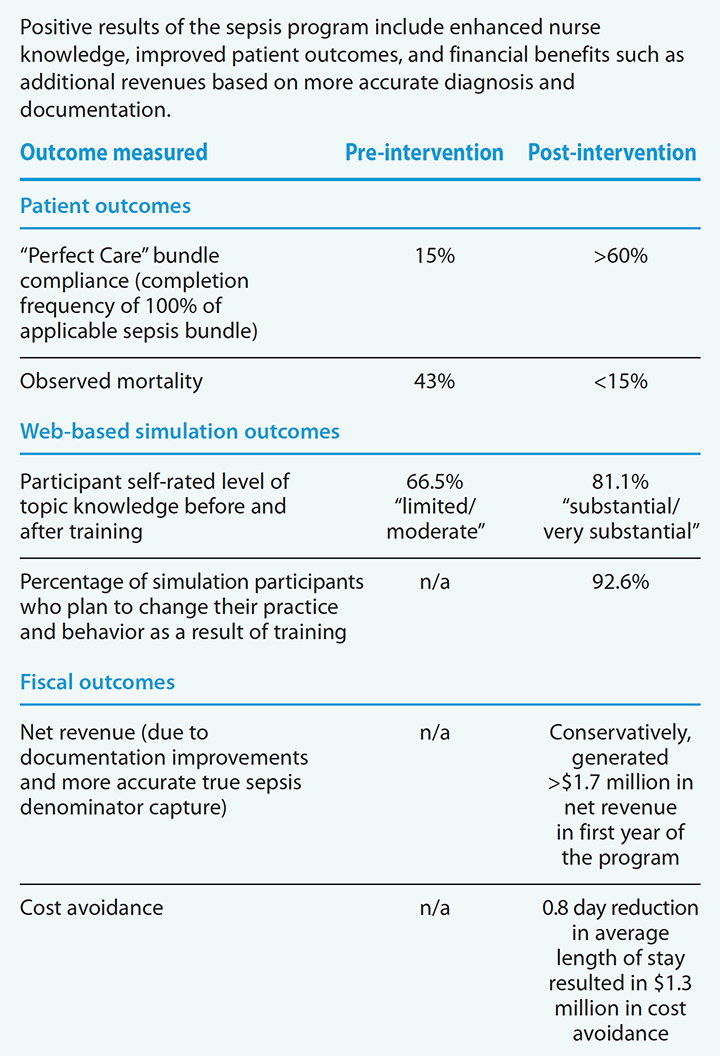
With persistence and patience, you can use these guiding principles to lead your sepsis program to improved patient outcomes, including sepsis bundle compliance, observed mortality, and sepsis identification. Since we launched our sepsis program in 2014, observed mortality has decreased from 43% to less than 15%. Sepsis bundle compliance has improved to consistently exceed 60% (surpassing state and national averages), and outcomes have been sustained or improved since we launched the CMS Core Measure. In 2021, bundle compliance is exceeding 80%.
We’ve also seen fiscal improvements. Not often does a program improve clinical practice and regulatory adherence, save lives, decrease costs, and generate revenue, but that’s been our experience. The program’s ability to drive improved outcomes in all of these areas is the result of contributions from a committed team at all levels of the organization.
A road worth taking
The guiding principles described in this article can be customized to meet the needs of your organization to help you build a successful sepsis program. However, the voices and efforts of nurses and clinical staff will provide the roadmap for implementation and success. As most clinicians know, this road is one worth taking because “Saving one life makes you a hero, but saving 100 likely makes you a nurse.”
Access references and additional outcomes data at myamericannurse.com/?p=74273.
Alexander Johnson is a clinical nurse specialist for critical care at Northwestern Medicine Central DuPage Hospital in Winfield, Illinois. Hillary Crumlett is director of operations for inpatient nursing and the emergency department at Northwestern Medicine Huntley Hospital in Huntley, Illinois.
Disaster planning and preparation
The declaration of a worldwide COVID-19 pandemic in March 2020 challenged all healthcare systems and hospitals to quickly evaluate current disaster plans and begin planning for an influx of patients. During disaster planning, organizations should use a framework for decision-making that prioritizes maximizing staff safety; minimizing staff, patient, and visitor exposure; and delivering high-quality care.
Organizations with a successful sepsis program and infrastructure are at an advantage for managing pandemics because of their established care delivery model. During the COVID-19 pandemic, healthcare organizations quickly realized that efficiency and effectiveness would be critical to successfully deliver care during patient surges, especially in critical care areas. When preparing for a surge, all care domains should be evaluated for potential impact by assessing the environment, supplies and equipment, education and training, and staffing and scheduling. (See Sepsis program advantage in a pandemic.)
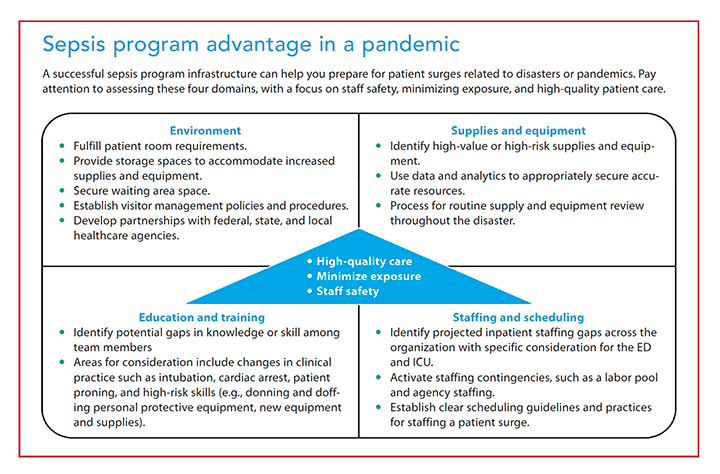

Our experience
We realized that we should be in an “always ready” mode for disaster situations, including COVID-19. We’ve taken several steps, including establishing a disaster-planning team, creating a credible disaster infrastructure, developing internal experts, and building a network of relationships within healthcare agencies. The disaster-planning team (called the incident command team in some organizations) focuses on developing strategic direction and actionable tactics that support three key priorities: maximizing staff safety, minimizing exposure, and providing high-quality care. We established a deliberate infrastructure with support from operations, logistics, communications, and support services. Central to our success was proactive education and simulated training in safe airway management, patient proning, and cardiac arrest.
In addition, interprofessional relationships across the organization ensured a rapid response to frontline healthcare team needs in guaranteeing the delivery of safe, high-quality care. While this experience has been challenging, having an effective and efficient response to the evolving pandemic created an environment of support and resilience.
Outcomes

References
Alhazzani W, Evans L, Alshamsi F, et al. Surviving Sepsis Campaign Guidelines on the Management of Adults with Coronavirus Disease 2019 (COVID-19) in the ICU: First update. Crit Care Med. 2021;49(3):e219-34.
Alhazzani W, Hylander Møller MH, Arabi YM, et al. Surviving Sepsis Campaign: Guidelines on the management of critically ill adults with coronavirus disease 2019 (COVID-19). Intensive Care Med. 2020;46:854-87. doi:10.1007/s00134-020-06022-5. link.springer.com/article/10.1007/s00134-020-06022-5
American Association of Critical-Care Nurses. Coronavirus (COVID-19) resources. aacn.org/clinical-resources/covid-19
American Association of Critical-Care Nurses. Nurses on the front line of sepsis. aacn.org/clinical-resources/sepsis
Dellinger RP, Schorr CA, Levy MM. A users’ guide to the 2016 Surviving Sepsis Guidelines. Crit Care Med.2017;45(3):381-5. doi:10.1097/CCM.0000000000002257
Grek A, Booth S, Festic E, et al. Sepsis and shock response team: Impact of a multidisciplinary approach to implementing Surviving Sepsis Campaign guidelines and surviving the process. Am J Med Qual.2017;32(5):500-7. doi:10.1177/1062860616676887
Ju T, Al-Mashat M, Rivas L, Sarani B. Sepsis rapid response teams. Critical Care Clin. 2018;34(2):253-8. doi:10.1016/j.ccc.2017.12.004
Kumar A. Systematic bias in meta-analysis of time to antimicrobial in sepsis studies. Crit Care Med. 2016;44(4):e234-5. doi:10.1097/CCM.0000000000001512
Levy MM, Evans LE, Rhodes A. The Surviving Sepsis Campaign bundle: 2018 update. Crit Care Med.2018;46(6):997-1000. doi:10.1097/CCM.0000000000003119
Puskarich MA, Trzeciak S, Shapiro NI, et al. Outcomes of patients undergoing early sepsis resuscitation for cryptic shock compared with overt shock. Resuscitation. 2011;82(10):1289-93. doi:10.1016/j.resuscitation.2011.06.015
Rhodes A, Evans LE, Alhazzani W, et al. Surviving Sepsis Campaign: International guidelines for management of sepsis and septic shock: 2016. Crit Care Med. 2017;45(3):486-552. doi:10.1097/CCM.0000000000002255
Seymour CW, Kahn JM, Martin-Gill C. et al. Delays from first medical contact to antibiotic administration for sepsis. Crit Care Med. 2017;45(5):759-65. doi:10.1097/CCM.0000000000002264
World Health Organization. Coronavirus disease (COVID-19) pandemic. who.int/emergencies/diseases/novel-coronavirus-2019?gclid=EAIaIQobChMI07XCrdfr7gIVXebjBx0ZCwtWEAAYASAAEgIgw_D_BwE