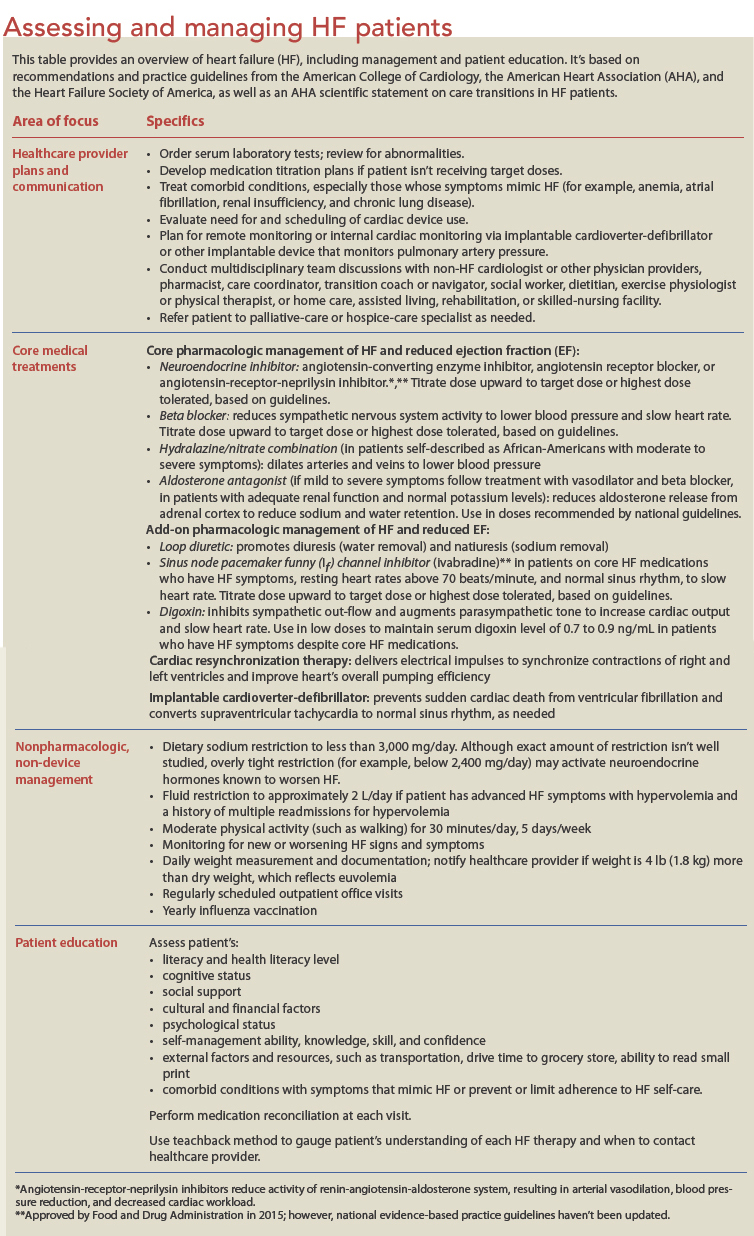
Cardiac resynchronization therapy (CRT) can help reduce readmissions in patients with heart failure (HF). It uses an implanted cardiac device to boost the heart’s efficiency and improve blood flow. CRT is intended for HF patients with ejection fractions (EFs) of 35% or less. (See The failing heart.)
Generally, patients meet the criteria for CRT if they:
- are in normal sinus rhythm
- have current HF symptoms (New York Heart Association [NYHA] functional classes II to IV) despite receiving core HF medications
- have left bundle branch block (LBBB) QRS morphology.
Like pacemaker therapy, CRT entails placing lead wires in the right atrium and right ventricle, with a third lead wire placed in a cardiac vein of the left ventricle. The device sends small, undetectable electrical impulses to both ventricles to improve the synchrony of electrical stimulation, promoting a more synchronized ventricular contraction pattern. When the right and left ventricles contract uniformly, stroke volume increases and mitral regurgitation decreases. As a result, HF symptoms ease and quality of life improves.
No guidelines exist for specific medication-plan revisions before CRT placement, with or without an implantable cardioverter-defibrillator, for patients who meet CRT inclusion criteria. When CRT is the treatment of choice, device placement poses a cost, but patients benefit through early and long-term symptom improvement.
HF readmissions
Hospital readmissions are common in HF patients—and are linked to increased morbidity and deaths. From 2007 to 2009, about 25% of HF patients were readmitted within 30 days. More important, readmission occurred soon after discharge. In a large review of Medicare beneficiaries, 61% of readmissions occurred during the first 15 days after discharge.
When patients are readmitted, their diagnoses aren’t always HF. In one study of 30-day readmissions after HF discharge, nearly 20% of patients were readmitted for worsening HF and 10% came in with acute myocardial infarctions. What’s more, 30-day mortality among patients readmitted for cardiovascular reasons exceeded that of patients readmitted for noncardiovascular reasons. Over a 3-year period, patients who weren’t readmitted had significantly lower death rates than those who were readmitted for either cardiovascular or noncardiovascular reasons. These findings highlight the complexity of HF.
When asked the reason for their HF readmission, patients’ rationales tended to reflect preventable causes. Data from medical records showed evidence of multiple medical conditions in patients who were readmitted. Patient interviews shed light on psychosocial factors, self-care adherence, and health-system failures and delays in treatment as rationales for increased readmissions.
The Centers for Medicare & Medicaid Services began public reporting of 30-day risk-standardized readmission rates for HF in an attempt to raise healthcare provider awareness and, ultimately, to lower rates. The National Quality Forum endorses the 30-day readmission measure as a way to improve quality of care and reduce preventable readmissions.
Evidence supporting CRT
Multiple prospective and retrospective research trials have shown that CRT reduces the risk of all-cause mortality and all-cause, cardiovascular, and HF readmissions.
CRT was applied in patients with an LBBB pattern and QRS durations of 150 ms or more, as well as in patients with a non-LBBB pattern and those with QRS durations between 120 and 150 ms. Patients in the first category had significant EF and functional status improvments as well as heart-size reduction (reduced left ventricular end-systolic and diastolic diameters) compared to the other groups. Surprisingly, though, CRT is underused in patients who meet the criteria. In fact, 60% of eligible patients don’t receive CRT despite known benefits.
Adaptive CRT
An improvement to CRT, called adaptive CRT (aCRT), is now available. This dynamic physiologic
pacing algorithm enhances CRT by adjusting device parameters automatically with changes in patient activity levels and conduction status. Rather than using echocardiographic (echo) optimization, the technology automatically adjusts to patients’ minute-by-minute needs and creates customized therapy. Compared to echo optimization, aCRT reduced the odds of 30-day HF hospital readmission by 59% and the odds of all-cause 30-day hospitalization by 46%. It also reduced all-cause and HF readmissions beyond 30 days. Ultimately, aCRT enhanced optimization, and research participants had reduced short- and long-term hospitalizations.
Case study
Mr. R, a 79-year-old Caucasian male, has a history of HF and reduced EF, along with hypertension, obesity, and gout. He was diagnosed with dilated cardiomyopathy about 4 years ago after complaining of fatigue and shortness of breath that didn’t subside with rest. He was unable to complete activities of daily living (ADLs) without frequent breaks and rest periods. His primary care physician placed him on evidence-based medication therapies, including an angiotensin-converting enzyme (ACE) inhibitor, a beta blocker, a loop diuretic, and an aldosterone antagonist.
Initially, Mr. R’s signs and symptoms improved. But in the last 6 months, they’ve become progressively more severe, so he is referred to a cardiologist. An echocardiogram shows Mr. R has an EF of 23%. Over the next 4 months, the cardiologist titrates his ACE inhibitor and beta blocker upward to target dosages without adverse effects. Subsequently, Mr. R’s EF improves to 35%. He states he is able to complete ADLs for short periods (improvement from a NYHA functional class IV [symptoms at rest] to III [moderate symptoms with activities]). However, his quality of life is poor and he is depressed because he can’t carry out activities for which he feels responsible and enjoys.
During an office visit with his advanced practice nurse provider, he becomes emotional and starts to cry when explaining that all he wants to do is walk his dog around the block and use his riding mower to cut the lawn. Mr. R states his wife is in charge of his food and he eats whatever she cooks. He adheres to all of his medications and doesn’t have financial, social, or cultural barriers that would keep him from following the prescribed HF management regimen. His serum electrolyte levels and blood chemistries are within normal ranges, his vital signs are stable, and he is in normal sinus rhythm with an LBBB, a heart rate of 82 beats/minute, and a QRS duration of 163 ms.
Analysis
Mr. R believes he’s following all the recommended therapies, yet he remains symptomatic without the quality of life he desires, leading to depression. Although he seems to be well-managed pharmacologically, his nurse needs to reconcile his medications at the beginning and end of each encounter to ensure that his actions meet the healthcare provider’s recommendations.
What’s more, she must thoroughly assess Mr. R’s self-care behaviors that could affect his quality of life and possibly lead to readmission. For instance, perhaps his wife is still buying and preparing low-sodium foods, but she and her husband are now eating at restaurants a few days each week because he feels better.
Many factors could explain Mr. R’s suboptimal functional status, including deconditioning and lack of a physical activity routine, comorbid conditions, hidden dietary sodium indiscretion from restaurant food, and poor absorption of his loop diuretic. He requires further assessment to determine if his blood counts and thyroid function are normal and if he has critical (but asymptomatic) coronary artery disease, sleep-disordered breathing, or another medical condition that could account for his symptoms. He appears to meet the criteria for CRT with an implantable cardioverter-defibrillator and additional medications, if worsening symptoms stem from hypervolemia.
Mr. R’s nurse should be sure to discuss CRT as an option, if appropriate. Also, Mr. R may meet the criteria for newer HF medications that increase vasodilation or slow the heart rate; research suggests these agents improve both symptom burden and quality of life. His nurse also may need to discuss more advanced HF therapies, such as ventricular assist device support or palliative and end-of-life care. (See Assessing and managing HF patients.)
Nurse’s role in managing HF
As Mr. R’s scenario shows, nurses are uniquely positioned to identify ways to help reduce HF readmissions. Given the high risk of early readmission after discharge, multidisciplinary care is crucial. Nurses need to be assertive advocates to ensure patients’ needs are met during the transition from hospital to home, as well as after discharge. Patient and family caregiver education should include signs and symptoms that herald worsening of the patient’s condition, pharmacologic and nonpharmacologic plans of care, and new interventions and technologies.
To provide thorough education and patient care, nurses must remain current in their knowledge of HF management—including understanding and applying guideline-directed medical therapies consistent with the highest evidence-based practices. And we must consider social, cultural, financial, and other factors that could pose barriers to care.
In addition, we must learn about new or improved drug and device therapies known to further reduce HF readmissions. For example, two new medications approved by the Food and Drug Administration are available for HF patients with reduced EF—ivabradine and the combination agent sacubitril/valsartan. Although each has unique applications and inclusion criteria for use, they both reduced HF hospitalizations compared to placebo (ivabradine) and usual care (sacubitril/valsartan).
When appropriate, nurses also may need to discuss use of CRT or aCRT with both patients and healthcare providers if symptoms don’t improve after optimization of HF medication. (See Indications for CRT.)
To help ensure optimal collaboration among the many stakeholders of HF care, we must promote and enhance communication among the multiple teams treating HF. By documenting the level of service the patient requires, along with changes in therapies, gaps, barriers to care, and patient and family preferences and needs, we can enhance care coordination among all care providers.
For nurses who focus on care coordination, connecting patients to the right care provider is crucial to ensure the patient is assessed thoroughly to determine optimal next steps. At every encounter, evaluate the patient’s self-confidence, as well as level of knowledge and skills in carrying out HF recommendations. Teach patients about self-care expectations related to diet, physical activity, and self-assessment of worsening symptoms, as well as the importance of keeping appointments with healthcare providers, even if they feel fine. For patients with refractory symptoms despite optimal medical therapies, palliative care may be needed to improve quality of life.
References
Albert NM, Barnason S, Deswal A, et al.; American Heart Association Complex Cardiovascular Patient and Family Care Committee of the Council on Cardiovascular and Stroke Nursing, Council on Clinical Cardiology, and Council on Quality of Care and Outcomes Research. Transitions of care in heart failure: a scientific statement from the American Heart Association. Circ Heart Fail. 2015;8(2):384-409.
Desai AS, Claggett B, Pfeffer MA, et al. Influence of hospitalization for cardiovascular versus noncardiovascular reasons on subsequent mortality in patients with chronic heart failure across the spectrum of ejection fraction. Circ Heart Fail. 2014;7(6):895-902.
Dharmarajan K, Hsieh AF, Kulkarni VT, et al. Trajectories of risk after hospitalization for heart failure, acute myocardial infarction, or pneumonia: retrospective cohort study. BMJ. 2015;350:h411.
Dupont M, Rickard J, Baranowski B, et al. Differential response to cardiac resynchronization therapy and clinical outcomes according to QRS morphology and QRS duration. J Am Coll Cardiol. 2012;60(7):592-8.
Go AS, Mozaffarian D, Roger VL, et al.; American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics—2013 update: a report from the American Heart Association. Circulation. 2013;127(1):e6-e245.
Enguidanos S, Coulourides Kogan AM, Schreibeis-Baum H, et al. “Because I was sick”: seriously ill veterans’ perspectives on reason for 30-day readmissions. J Am Geriatr Soc. 2015;63(3):537-42.
Fang JC, Ewald GA, Allen LA, et al. Advanced (stage D) heart failure: a statement from the Heart Failure Society of America Guidelines Committee. J Card Fail. 2015;21(6):519-34.
Heart Failure Society of America, Lindenfeld J, Albert NM, Boehmer JP, et al. HFSA 2010 Comprehensive Heart Failure Practice Guideline. J Card Fail. 2010;16(6):e1-e194.
Heidenreich PA, Albert NM, Allen LA, et al.; American Heart Association Advocacy Coordinating Committee; Council on Arteriosclerosis, Thrombosis, and Vascular Biology; Council on Cardiovascular Radiology and Intervention; Council on Clinical Cardiology; Council on Epidemiology and Prevention; Stroke Council. Forecasting the impact of heart failure in the United States: a policy statement from the American Heart Association. Circ Heart Fail. 2013;6(3):606-19.
Lund LH, Benson L, Ståhlberg M, et al. Age, prognostic impact of QRS prolongation and left bundle branch block, and utilization of cardiac resynchronization therapy: findings from 14,713 patients in the Swedish Heart Failure Registry. Eur J Heart Fail. 2014;16(10):1073-81.
Peterson PN, Greiner MA, Qualls LG, et al. QRS duration, bundle-branch block morphology, and outcomes among older patients with heart failure receiving cardiac resynchronization therapy. JAMA. 2013;310(6):617-26.
Retrum JH, Boggs J, Hersh A, et al. Patient-identified factors related to heart failure readmissions. Circ Cardiovasc Qual Outcomes. 2013;6(2):171-7.
Starling RC, Krum H, Bril S, et al. Impact of a novel adaptive optimization algorithm on 30-day readmissions: evidence from the Adaptive CRT Trial. JACC Heart Fail. 2015;3(7):565-72.
Tracy CM, Epstein AE, Darbar D, et al. 2012 ACCF/AHA/HRS focused update of the 2008 guidelines for device-based therapy of cardiac rhythm abnormalities: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2012;60(14):1297-313.
Yancy CW, Jessup M, Bozkurt B, et al; American College of Cardiology Foundation; American Heart Association Task Force on Practice Guidelines. 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2013;62(16):e147-e239.
Nancy M. Albert is associate chief nursing officer for Cleveland Clinic’s Office of Nursing Research and Innovation and a clinical nurse specialist at in Cleveland Clinic’s Kaufman Center for Heart Failure.