Understanding options for patients helps you be a more effective caregiver.
Takeaways
- Transcatheter aortic valve replacement (TAVR) is an alternative to surgical aortic valve replacement for some patients.
- Understanding TAVR approaches, post-procedure monitoring, and potential complications will help nurses better care for patients undergoing TAVR.
For many years, surgical aortic valve replacement (SAVR) was the gold-standard treatment for severe, symptomatic aortic stenosis (AS). For eligible patients, this procedure can improve symptoms and extend life. But it has a major drawback: Whether the surgical approach is open (using a full sternotomy) or less invasive (using a ministernotomy or minithoracotomy), SAVR requires cross-clamping of the aorta and cardiopulmonary bypass.
Improvements in medical therapies and aging of the population mean more elderly patients are living with severe AS and other comorbidities. For the one-third of those patients who aren’t eligible for SAVR, medical management may improve symptoms but can’t extend life or slow disease progression. Without surgery, their life expectancy is 2 to 3 years; only 50% live more than 2 years after symptom onset.
Understanding medical management of aortic stenosis
Transcatheter aortic valve replacement: New hope for patients with aortic stenosis
Vascular closure devices: What you know can prevent serious complications
Enter TAVR: Transcatheter aortic valve replacement
Transcatheter aortic valve replacement (TAVR), which involves a collapsible prosthetic valve placed directly over the native diseased valve, has emerged as a minimally invasive alternative to SAVR. A guidewire is fed through the aorta; then a catheter with a prosthetic valve on the end is fed over the wire and placed over the aortic valve. The prosthetic valve can be deployed percutaneously or through a small incision in the chest wall. The procedure takes 4 to 5 hours and is done in a hybrid cardiac catheterization laboratory.
This article reviews patient eligibility for TAVR, procedural approaches, prosthetic valve types, potential complications, nursing care, and patient education.
Patient assessment and eligibility
Determining if a patient is a better candidate for SAVR or TAVR involves an interdisciplinary evaluation by a heart-valve team with members from cardiac surgery, cardiac imaging, interventional cardiology, and cardiac anesthesia, as well as nursing professionals. The team assesses surgical risk, valve anatomy and function, signs and symptoms, overall cardiovascular disease and health status, comorbidities, physical and cognitive function, and life expectancy.
The first step is to identify the patient’s overall surgical risk. (See Determining surgical risk for patients with heart-valve disease.) The Society of Thoracic Surgeons’ Predicted Risk of Mortality (STS-PROM) can be used to assess risk. It calculates predicted risk of death with surgery or all-cause death after cardiac surgery based on specific patient characteristics (for example, age, gender, height, weight, and race) and clinical variables (such as cardiac history, previous myocardial infarction, heart failure symptoms, endocarditis, previous coronary artery bypass grafting, and other comorbidities).
Once the team establishes surgical risk, they must decide whether SAVR or TAVR is the patient’s best option. (See SAVR or TAVR?) Diagnostic techniques that aid this decision include transthoracic echocardiography (TTE) and multidetector computed tomography (MDCT). MDCT helps find the correct replacement-valve size and identifies possible peripheral vascular complications to ensure the best transcatheter approach. To encourage shared decision making, the team should consider patient and family preferences, goals, and expectations.
TAVR approaches
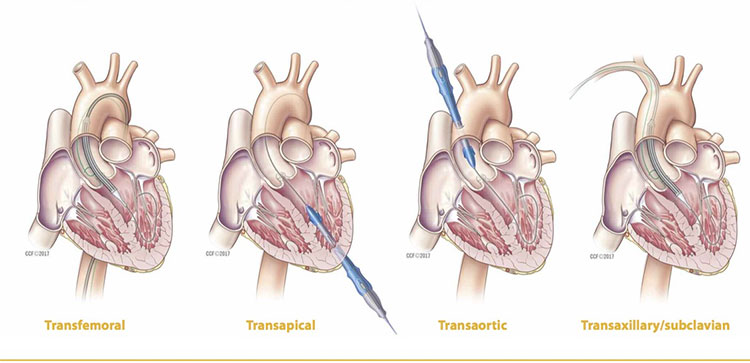
Approved TAVR techniques include percutaneous approaches (transfemoral [TF] and transaxillary/subclavian [TAx]) and traditional open approaches (transapical [TA] and transaortic [TAo]), which require small surgical incisions. Minimally invasive percutaneous procedures (such as TF and TAx) are the most common. (See Approaches to TAVR.)
Percutaneous approaches
About 90% of TAVRs use the TF approach. A sheath is inserted into the femoral artery, through which the guidewire and catheter are fed through the aorta into the heart. The arterial insertion site is typically closed using a vascular closure device, such as a vessel plug, clip, or internal suture. The TF approach may not be suitable for patients with peripheral vascular disease because of potential issues with vessel size.
In the TAx approach, access is gained through the subclavian artery, with a sheath catheter fed into the aortic arch to the aortic valve. Although this approach offers a shorter catheter-insertion route, the smaller vessel can cause difficulty with maneuvering the catheter and may lead to brachial-nerve injury.
Open approaches
A minithoracotomy is used for access in the TA approach. Benefits include avoiding a diseased aorta or femoral artery, unlimited delivery system size, and easier valve delivery. Drawbacks include risk of myocardial injury, increased risk of wall-motion abnormalities, apical bleeding, and incision pain. Additionally, this technique requires a surgical incision through the chest wall and intubation, increasing patient discomfort and pain.
The TAo approach involves direct puncture of the aorta through a partial sternotomy or right thoracotomy. Benefits resemble those of the TA approach, with a small working distance to the valve and no limit to access size. In addition to incision and intubation, drawbacks include limited access if the vessel is diseased or the ascending aorta is heavily calcified.
Replacement-valve options
The Food and Drug Administration (FDA) has approved two valves for TAVR: Edwards LifeSciences SAPIEN® valves and the Medtronic CoreValve®. (See FDA-approved valves for TAVR.)
Made with bovine pericardium, the SAPIEN XT® and SAPIEN 3® valves are attached to an expandable chromium cobalt balloon stent. Both require a smaller sheath size than other valve deployment systems—as low as 14 Fr. The SAPIEN XT is approved for valve-in-valve procedures for patients with failed or deteriorating previously replaced tissue valves. The SAPIEN 3 has a sealing cuff to minimize paravalvular regurgitation (PVR), a TAVR complication.
The trileaflet porcine pericardial Medtronic CoreValve is affixed to a nickel titanium (nitinol) stent. Nitinol provides super elasticity for shape memory, even under stress. This valve can be recaptured and repositioned during placement for the best fit. The CoreValve Evolut R® was approved by the FDA in 2015 for valve-in-valve treatment of failed bioprosthetic valves.
Most patients can be treated successfully using either type of valve, with similar outcomes. However, one valve may be preferable in specific circumstances. For example, a repositionable self-expanding valve may be preferred if a patient has severe valvular calcification, which increases the risk of annulus rupture. Also, a patient who requires a particular approach will need the valve that has been approved for it. For example, a balloon-expandable valve would be used for a TA approach.
Because TAVR was approved only in 2011, no substantive longitudinal valve durability data have been established. Current and future studies should focus on long-term durability, especially in light of FDA approval for use of TAVR valves in intermediate-risk patients who have longer life expectancies than traditionally treated high-risk patients.
Determining surgical risk for patients with heart-valve disease
The American College of Cardiology and the American Heart Association make the following recommendations related to surgical risk for patients with heart-valve disease.
Frailty
To help determine the patient’s level of frailty, assess his or her ability to perform activities of daily living by evaluating:
- ambulation
- bathing
- dressing
- eating
- toileting
- transferring
- urinary continence
If appropriate, care providers may use a scoring system to apply a categorical functional level to the patient’s ability to perform activities of daily living. For example: none, mild, moderate, severe.
Major organ-system compromise
Examples include:
- cardiac compromise (e.g., severe ventricular dysfunction)
- cancer
- central nervous system compromise (e.g., dementia, Alzheimer’s disease, cerebrovascular accident)
- GI compromise (e.g., Crohn’s disease, ulcerative colitis, malnutrition)
- hepatic compromise (e.g., cirrhosis)
- pulmonary compromise (e.g., pulmonary hypertension)
- renal compromise (e.g., stage 3 or higher renal disease).
Open heart vs. percutaneous approach—surgical considerations
- chest malformation
- heavily calcified (porcelain) aorta
- radiation damage
- tracheostomy.
Sources:
Nishimura RA, et al. Circulation. 201;129(23):2440-92; Otto, et al. J Am Coll Cardiol. 2017;69(10):1313-46.
Treatment approach for patients with severe symptomatic aortic stenosis is based partly on the patient’s surgical risk. This table provides risk-level definitions and recommended approaches.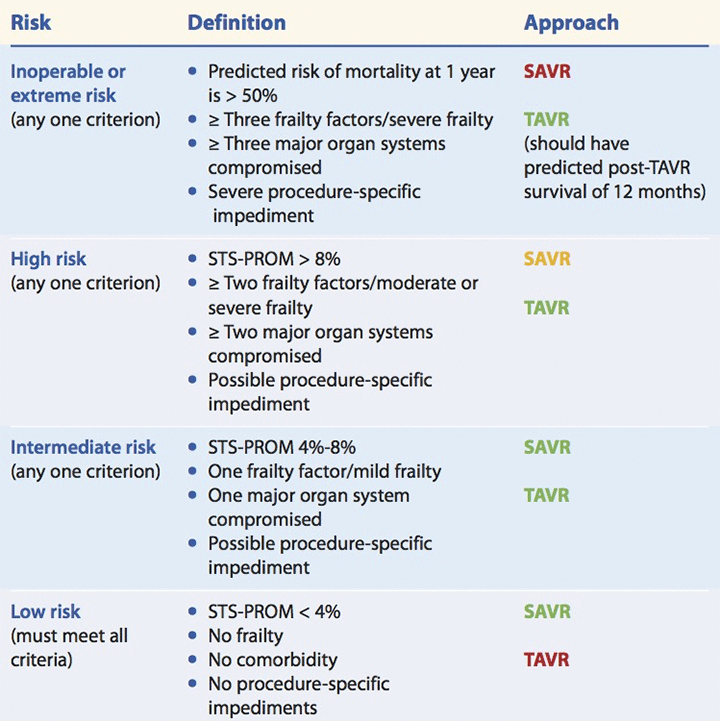

STS-PROM = Society of Thoracic Surgeons’ predicted risk of mortality, SAVR = surgical aortic valve replace- ment, TAVR = transcatheter aortic valve replacement
All approaches are made with a heart-valve team discussion; however, some approaches require other con- siderations. Red = not appropriate. Yellow = not preferred but possible, based on decision of team after evaluation of patient-specific factors. Green = preferred.
Sources: Nishimura RA, et al. Circulation. 201;129(23):2440-92; Otto, et al. J Am Coll Cardiol. 2017;69(10): 1313-46.
Postprocedure complications
Vascular complications associated with femoral access include hematomas, retroperitoneal bleeding, and arterial occlusion.
Hematomas, the most common vascular access complication, develop when blood leaks from the puncture point into the soft tissue. The affected area may be firm, swollen, and discolored, and the patient may complain of tenderness and pain. Because the internal arterial puncture site is proximal to the external incision, apply pressure 1 to 2 cm above the puncture site until hemostasis is achieved. Report the findings to the provider, mark the boundaries of the area, and evaluate the site for changes such as thigh enlargement, discoloration outside the marked boundaries, and changes in vital signs and pain level or location.
Retroperitoneal bleeding is more likely to occur when the femoral artery is punctured above the inguinal ligament (commonly known as a “high stick”). Clinical findings include back, flank, or abdominal pain; decreases in blood pressure, hemoglobin, and hematocrit; and increased heart rate. You may not see obvious signs of bleeding.
Report suspected retroperitoneal bleeding to the provider, who will order computed tomography to diagnose the problem. Patients with this complication must remain on bed rest. Administer I.V. fluids and blood transfusions, as ordered. Surgical repair may be necessary.Suspect an arterial occlusion if the patient has pain, paresthesia, pallor, absent pulses in the affected extremity, and inability to move the limb. Treatment depends on occlusion size and location, as well as symptom severity. The thrombus may lyse spontaneously. If it doesn’t, surgery or thrombolytic agents may be warranted, if not contraindicated.
![]()
![]()
Approaches to TAVR
Minimally invasive transcatheter aortic valve replacement (TAVR) may involve either a percutaneous (transfemoral or transaxillary/ subclavian) approach or an open (transapical or transaortic) approach. The options are illustrated below.

Another complication is PVR, which can be mild, moderate, or severe. It can be caused by a mismatch of the prosthetic valve and the native valve annulus, incomplete valve stent frame positioning due to calcification on the native annulus, or prosthetic valve placement with incomplete annulus sealing of the valve skirt or cuff. Modifications to third-generation valves, including the sealing cuff and the ability to reposition the valve, may decrease risks of PVR.
Other significant complications include stroke or transient ischemic attack, myocardial infarction, cardiac conduction abnormalities, and acute renal injury. (See Nursing care for patients with TAVR complications.)
Nursing care
Up to one-third of patients experience complications after TAVR, so nursing care focuses mainly on patient monitoring and assessment and on educating patients about activity, diet, medications, and pain management.
Postprocedure monitoring
Postprocedure care begins with the handoff report. Obtain pertinent information, including vital signs, medications administered during the procedure, current level of alertness, access difficulties (including placement of the access point and need for reaccess attempts), and overall procedure events, such as complications, arrhythmias or other cardiac events, or difficulties with placement. As ordered, place the patient on continuous telemetry monitoring and observe for heart rate and rhythm changes. Auscultate heart sounds to detect changes from baseline, and monitor vital signs frequently per protocol or orders.
If percutaneous access was performed, monitor the site distal to the puncture for adequate circulation. When checking vital signs, perform neurovascular assessment of the affected extremity, including color, temperature, pulse, numbness, tingling, and swelling. Assess the insertion site for signs of bleeding, hematoma, and infection. Keep dressings clean, dry, and intact and look for bleeding. Promptly report the need for dressing changes due to bleeding. Dressings may typically be removed 24 to 48 hours after the procedure.
Patients at risk for cardiac conduction abnormalities, such as heart block, typically receive a temporary transvenous external pacemaker. Confirm that pacemaker settings match those ordered. Monitor routine laboratory results, such as renal function tests, blood cell counts, electrolytes, and partial thromboplastin times. Report findings of concern.
Patients with AS are preload dependent because of left ventricular hypertrophy and impaired relaxation from cardiac remodeling. Carefully monitor fluid balance for incresed preload, which may cause pulmonary congestion, and decreased
preload, which may impair cardiac output and reduce perfusion to extremities. Track fluid status through intake and output records, laboratory results, and daily weights.
Additional considerations related to open approaches include a chest tube and surgical incisions. Assess incisions for signs of bleeding or infection and observe chest-tube drainage for increases and bleeding.
If the patient received modified anesthesia or conscious sedation, monitor for tolerance and recovery from sedation.
FDA-approved valves for TAVR
The Food and Drug Administration (FDA) has approved two types of valves for transcatheter aortic valve replacement (TAVR).
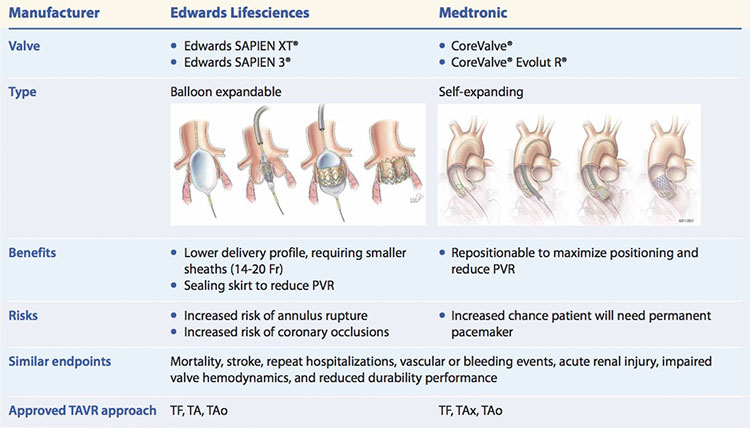

PVR = paravalvular regurgitation, TA = transapical, TAo = transaortic, TAx = transaxillary/subclavian, TF = transfemoral
Activity and diet
After the procedure, patients who had a percutaneous approach typically are placed on 6 hours of bed rest. If the femoral artery was accessed during the procedure, keep the head of the bed elevated 30 degrees or less to minimize bending at the groin and avoid disrupting the closure device at the puncture site.
After bed rest ends, assist the patient with walking as needed to help prevent complications. Check orthostatic vital signs the first time the patient gets out of bed, and assess overall mobility.
Advance the patient’s diet as tolerated once bed rest ends. Encourage the patient to eat meals out of bed to promote greater activity.
Medications and pain management
When patients are able to eat and drink, expect to resume oral medications to control hypertension or other comorbidities. To reduce thromboembolism risk, providers typically prescribe dual antiplatelet medications. Patients will need to take aspirin (75 to 100 mg/day) for the rest of their lives, along with clopidogrel or prasugrel for varying durations. For example, those with self-expanding valves may be prescribed clopidogrel 75 mg daily for 3 months, whereas those with balloon-expandable valves may need to take it for 6 months. Some patients require antithrombotic therapy for other reasons, such as coronary stents or atrial fibrillation, so therapy duration may be managed according to other guidelines or specifications.
Give pain medication, as ordered. Monitor the patient for tolerance and efficacy, and report concerns to the provider.
Nursing care for patients with TAVR complications
This chart describes complications of transcatheter aortic valve replacement (TAVR), along with their causes and corresponding nursing interventions.
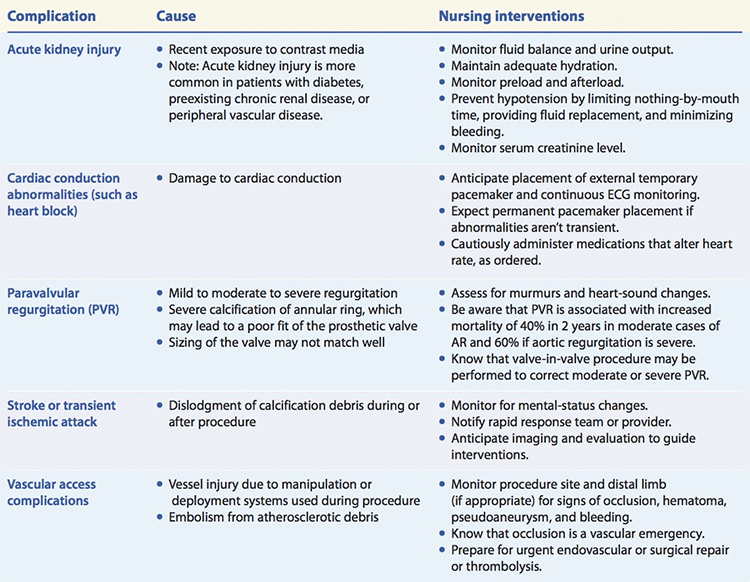

Safety and patient education
Throughout the patient’s stay, make safety a nursing care priority. Be sure to screen for fall risk and put appropriate interventions in place based on the results. In patients with a central line or urinary catheter, assess the need for using them every day, and obtain an order to remove them as soon as possible. This will help reduce the risk of hospital-acquired infections. To reduce the risk of venous thromboembolism, apply sequential compression devices as ordered and provide appropriate education.
Teach the patient and family about the risk of bleeding caused by antiplatelet or antithrombotic medications. Advise patients to contact their cardiologist if another healthcare provider tells them to stop the medications. Explain that ongoing follow-up will include periodic visits with a cardiologist. Inform them that the American Heart Association/American College of Cardiology recommend antibiotic prophylaxis before certain dental procedures for patients with a prosthetic valve.
The future of TAVR
As the population eligible for TAVR expands and more prosthetic valves are approved, you’re likely to encounter more patients who undergo this procedure. By understanding the various TAVR approaches and postprocedure care, you can help ensure the best possible patient outcomes.
Illustrations reprinted with permission, Cleveland Clinic Center for Medical Art & Photography © 2006-2017. All Rights Reserved.
Kelly Haight is a clinical nurse specialist at the Cleveland Clinic in Cleveland, Ohio.
Selected references
Arora S, Misenheimer JA, Jones W, et al. Transcatheter versus surgical aortic valve replacement in intermediate risk patients: A meta-analysis. Cardiovasc Diagn Ther. 2016;6(3):241-9.
Arsalan M, Walther T. Durability of prostheses for transcatheter aortic valve implantation. Nat Rev Cardiol. 2016;13(6):360-7.
Bhatheja S, Panchal HB, Barry N, Mukherjee D, Uretsky BF, Paul T. Valvular performance and aortic regurgitation following transcatheter aortic valve replacement using Edwards valve versus CoreValve for severe aortic stenosis: A meta-analysis. Cardiovasc Revasc Med. 2016;17(4):248-55.
Ellis MF. Transcatheter aortic valve replacement: An evolving option for severe aortic stenosis. Advance Healthcare Network for Nurses. 2015.
Kondur A, Briasoulis A, Palla M, et al. Meta-analysis of transcatheter aortic valve replacement versus surgical aortic valve replacement in patients with severe aortic stenosis. Am J Cardiol. 2016;117(2):252-7.
Malaisrie SC, Iddriss A, Flaherty JD, Churyla A. Transcatheter aortic valve implantation. Curr Atheroscler Rep. 2016;18(5):27.
Merriweather N, Sulzbach-Hoke LM. Managing risk of complications at femoral vascular access sites in percutaneous coronary intervention. Crit Care Nurse. 2012;32(5):16-29.
Nishimura RA, Otto CM, Bonow RO, et al. 2014 AHA/ACC guideline for management of patients with valvular heart disease: Executive summary: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 2014;129(23):2440-92.
Otto CM, Kumbhani DJ, Alexander KP, et al. 2017 ACC expert consensus decision pathway for transcatheter aortic valve replacement in the management of adults with aortic stenosis: A report of the American College of Cardiology Task Force on Clinical Expert Consensus Documents. J Am Coll Cardiol. 2017;69(10):1313-46.
Urena M, Hayek S, Cheema AN, et al. Arrhythmia burden in elderly patients with severe aortic stenosis as determined by continuous electrocardiographic recording: Toward a better understanding of arrhythmic events after transcatheter aortic valve replacement. Circulation. 2015;131(5):469-77.
Vahl TP, Kodali SK, Leon MB. Transcatheter aortic valve replacement 2016: A modern-day “through the looking-glass” adventure. J Am Co



















3 Comments.
While researching TAVR for my elderly mother I found this article. How can I know if the information is up to date for 2019? This article helped me develop a question list for Mom’s primary provider as well as the proceduralist .
I am so happy to get this post. This is a nice post. I read your post. Its helps to patient
.This is great Thanks for published this post.
Hi I am currently working on my masters project for my nurse practitioner program. I was wondering if you might have any test that I might be able to use for my project. I will be educating nurses on TAVR care, complications, and resources that are important when caring for TAVR patients.