Technology aids diabetes self-management and enhances treatment planning.
Learning Objectives:
- Describe continuous glucose monitor (CGM) device options and the differences between personal and professional devices.
- Describe how CGM helps improve diabetes management.
- Discuss nursing implications in caring for patients using CGM devices.
The authors and planners of this CNE activity have disclosed no relevant financial relationships with any commercial companies pertaining to this activity. See the last page of the article to learn how to earn CNE credit.
Expiration: 5/1/25
Takeaways:
- A continuous glucose monitor (CGM) is a wearable sensor that automatically measures the amount of glucose in interstitial fluid.
- CGM delivers readings all day and night compared to self-monitoring blood glucose finger-sticks that reveal glucose values at a particular point in time.
- Nurses play an important role in providing education to people with diabetes and highlighting the importance of CGM.
Blood glucose self-monitoring helps patients with diabetes understand the interrelationships among food, activity, and medication to achieve their glycemic targets. It also aids treatment effectiveness assessment. Patients continue to use blood glucose meters, but in the early 2000s, continuous glucose monitoring (CGM) revolutionized home monitoring. CGM devices comprehensively optimize diabetes management by reviewing activity levels, medication and insulin dosing, food intake, and stress to provide patients with information about how self-care decisions affect glucose levels. As CGM accuracy, reliability, and convenience continue to improve, more healthcare providers use them.
The American Association of Clinical Endocrinology (AACE) and the American Diabetes Association (ADA) recommend CGMs for all patients with diabetes who administer multiple (three or more) injections of insulin per day or use an insulin pump. Although most patients continue to self-monitor and inject insulin, CGM provides an accurate picture of glucose levels throughout the day and night, which allows patients to quickly assess glycemic patterns to prevent hypoglycemia. CGM devices also capture information that helps providers create treatment plans based on real-time glucose results and to remotely view glucose values 24 hours a day, including when the patient hasn’t checked their glucose or while sleeping.
How does CGM work?
Rather than performing finger sticks several times a day to monitor glucose levels, patients with a CGM device insert a small sensor wire under the surface of their skin (usually on the abdomen or back of the arm) with an automatic applicator and secure it with an adhesive patch. A glucose-oxidase platinum electrode attached to the sensor measures the glucose concentration in interstitial fluids throughout the day and night. A transmitter sends real-time glucose readings wirelessly to a handheld receiver or compatible smart device (or insulin pump), which displays current glucose levels and trends.


Depending on the specific CGM device, the patient wears the sensor 7 to 14 days. A CGM can be worn during activities of daily living, including showering and working out. Blood glucose readings are sent to the receiver (smart device) every 5 minutes. Some devices allow custom glucose threshold alerts. (See Patient instructions.)
Patient instructions
Instruct patients to follow these steps when inserting a continuous glucose monitoring device:
- Before opening the sensor package, wash your hands.
- Avoid touching the adhesive area on the sensor.
- Choose a flat and pinchable site for insertion (avoiding scarred or irritated skin), such as the abdomen or back of the arm.
- Clean the area with alcohol and let it dry. (The patient may need to prep the area with a skin preparation or adhesive product before inserting the sensor. These supplies are included in each CGM package.)
- Place the sensor on the area and push on the plunger to remove the introducer needle, which leaves the sensor under the skin. Most patients feel only a slight pinch when inserting the sensor.
Professional vs. personal CGM
Healthcare providers distinguish between two types of CGM devices: personal (owned by the patient) and professional (owned by a healthcare practice for professional use).
Personal devices
Personal CGM systems measure blood glucose continuously and record readings every 5 minutes. Unlike self-monitoring, which provides a snapshot or point-in-time glucose measurement, CGM devices report data in numerical and graph-like formats, indicating current glucose levels and possible patterns. This allows patients to respond quickly to prevent acute glycemic events and permits informed decisions about insulin dosing and self-management. Glucose readings and trend arrows provide information about glucose highs and lows to assist in problem-solving related to meals, physical activity, and illness. Using the personal device, patients can share data remotely with family, caregivers, friends, and healthcare providers.
Most personal CGM devices alert patients when glucose levels are too low or too high. This feature is important for patients who experience frequent hypoglycemia or hyperglycemia unawareness. For patients who don’t, they may choose to disable the alerts. However, to ensure critical lows aren’t ignored, some alerts can’t be disabled.
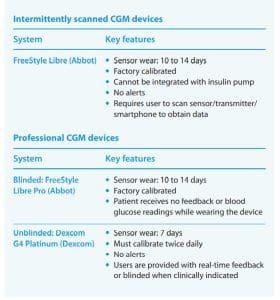
Professional devices
Professional CGM devices are purchased by a healthcare provider’s practice and worn by the person with diabetes for a short period, usually 7 to 14 days. Some professional devices collect glucose data in a “blinded” mode (the patient doesn’t view the data and has no interaction with the sensor), whereas others display real-time data viewable by the patient. After wearing the device for the specified period, the provider uploads the data and meets with the patient to review it and discuss needed changes (including medication adjustments) in diabetes self-management to achieve more targeted glycemic levels. Using a professional CGM device may lead a patient to purchase a personal device.
CGM system options
Two system options exist for both personal and professional CGM devices: real-time CGM (rtCGM) and intermittently scanned CGM (isCGM). rtCGM (continuous CGM) devices automatically transmit a continuous stream of glucose data to a receiver, smartphone, smartwatch, or other compatible device every 5 minutes. They also alert patients to impending hypoglycemia or hyperglycemia.
isCGM devices (previously called flash CGM) provide the same type of glucose data but require the patient to swipe or scan the receiver to obtain it. The information is then sent to a smartphone, smartwatch, or other compatible device. Until 2015, isCGM devices didn’t have the added safeguard of alerts. Newer isCGM devices now offer optional alerts. (See CGM systems.)
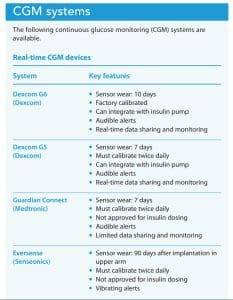


Nurses provide education about CGM advantages and challenges, including the need for self-monitoring. Although CGM devices no longer require calibration against finger blood measurements to ensure accuracy, if glucose data aren’t available (for example, during CGM start-up periods), the CGM malfunctions, the patient runs out of sensor supplies, or the patient feels that their symptoms don’t match the glucose data, a finger stick may be needed to assess blood glucose levels. Any discrepancy between symptoms and sensor numbers requires a finger stick. As a safety precaution, most organizations require a finger stick to establish insulin doses and have policies and care guidelines for charting and documenting decisions based on CGM data. Nurses should encourage patients to provide up-to-date information to ensure appropriate care.


CGM devices don’t automatically adjust insulin delivery unless it’s combined with an integrated, CGM sensor-augmented insulin delivery device. For example, CGM data may identify patterns of high glucose readings. In such cases, patients can treat or prevent high glucose readings themselves before they become a problem. Advances in diabetes technology have combined CGM with continuous insulin infusion pumps that automate insulin dosing and delivery. For patients who use both a CGM device and an insulin pump, insulin dosing and delivery should be done automatically based on data from the CGM.
CGM case study
Michael, a 57-year-old man with a 5-year history of type 2 diabetes and several comorbidities (hypertension, hyperlipidemia, and renal insufficiency), takes glimepiride, 2 mg every morning; metformin, 500 mg twice daily; and one injection of basal insulin (insulin glargine), 30 units every evening. His glycated hemoglobin has been increasing (his most recent was 8.6%) despite initial success with a healthy meal plan and walking three times a week. When first diagnosed with diabetes, Michael routinely checked his glucose, but his efforts have been inconsistent for the past 2 years because of fingertip pain. Michael’s primary care provider recommends meeting with a diabetes care and education specialist to discuss continuous glucose monitoring (CGM).
After meeting with the specialist, Michael reluctantly agrees to try a professional CGM device. Two weeks later, he returns to the specialist to download the glucose data and review it for patterns and possible insulin dose adjustments. When reviewing data before each meal and at bedtime, Michael begins to see a connection between his treatment plan and the impact of meals, physical activity, and medication. After a treatment plan change, Michael believes he’ll be comfortable purchasing a personal CGM device and never wants to return to finger sticks.
Selecting CGM devices
In 2019, the ADA added a new diabetes technology section to the Standards of Medical Care in Diabetes. Guidelines recommend that when patients are properly educated, CGM is a useful tool to lower glucose levels and reduce hypoglycemia. The 2020 standards go further and recommend CGM as a monitoring option for patients with a history of hypoglycemia and those who haven’t met glycemic targets with self-monitoring.
Patients choose personal or professional CGM devices based on several factors. Many who select a personal device want more engagement with their diabetes management. They want a complete picture of their glucose levels throughout the day. Some patients at risk for hypoglycemia or hypoglycemia unawareness prefer a personal device that offers real-time glucose alerts in the event of a hypoglycemic event. Patients with comorbidities who are at increased risk for diabetes-related complications also may benefit from receiving 24/7 glucose values.


Patients who use an insulin pump may be interested in a sensor-augmented pump (SAP) that connects with a specific CGM device. Transmitted CGM data prompt independent SAP basal rate adjustments when blood glucose levels are low or outside of the target range. Sometimes called “non-feedback control” systems, SAPs require manual adjustments and input from the patient, who remains responsible for programming the pump to accommodate the number of carbohydrates they eat at every meal and approving meal-time boluses.
An open-loop system is a step toward a closed-loop (feedback control) system, sometimes called an artificial pancreas. Closed-loop systems automatically adjust insulin with no patient input. (See Closed-loop systems and quality of life.)
Closed-loop systems and quality of life
Boughton and Hovorka describe hybrid closed-loop continuous glucose monitoring systems as rapidly transitioning from research into clinical practice. The systems present various psychosocial benefits and challenges.
Benefits
- Reduced anxiety
- Improved sleep related to confidence in overnight glucose control
- Less restrictive diets
- Fewer diabetes management demands
Challenges
- Technical issues
- Alarm intrusiveness
- Equipment burdens
- Time to trust that the system will work
Ideal candidates for professional CGM devices include those who have difficulty maintaining target glucose levels, those at increased risk for hypoglycemia, and those who don’t want to use a full-time personal CGM. These patients still may benefit from periodic CGM data collection, which helps demonstrate to health insurance carriers the importance of the devices. Providers sometimes ask a patient to wear a professional CGM device 2 weeks before a scheduled visit or at the time of an appointment. Glucose data are downloaded and used as an education tool and to help identify patterns of glucose variability.
Patients with type 2 diabetes and those not receiving insulin therapy also can benefit from professional CGM devices. They provide patients and providers with a comprehensive overview and capture real-time glucose variability so patients can respond to trends before serious complications arise.
Beyond the finger stick
CGM devices go beyond finger sticks to provide glucose monitoring throughout the day. In addition to helping patients better manage their diabetes, the devices benefit family members, caregivers, and friends who also participate in diabetes management.
James A. Fain is associate dean for academic affairs at the University of Massachusetts Chan Medical School, Tan Chingfen Graduate School of Nursing in Worcester, and editor-in-chief of The Science of Diabetes Self-Management and Care.
References
American Diabetes Association. 7. Diabetes technology: Standards of Medical Care in Diabetes—2021. Diabetes Care. 2021;44(Suppl 1):S85-99. doi:10.2337/dc21-S007
Boughton CK, Hovorka R. New closed-loop insulin systems. Diabetologia. 2021;64:1007-15. doi:10.1007/s00125-021-05391-
Edelman SV, Argento NB, Pettus J, Hirsch IB. Clinical implications of real-time and intermittently scanned continuous glucose monitoring. Diabetes Care. 2018;41(11):2265-74. doi:10.2337/dc18-1150
Grunberger G, Sherr J, Allende M, et al. American Association of Clinical Endocrinology clinical practice guideline: The use of advanced technology in the management of persons with diabetes mellitus. Endocr Pract. 2021;27(6):505-37. doi:10.1016/j.eprac.2021.04.008
Longo R, Sperling S. Personal versus professional continuous glucose monitoring: When to use which on whom. Diabetes Spectr. 2019;32(3):183-93. doi:10.2337/ds18-0093
Miller EM. Using continuous glucose monitoring in clinical practice. Clin Diabetes. 2020;38(5):429-38. doi:10.2337/cd20-0043
Olczuk D, Priefer R. A history of continuous glucose monitors (CGMs) in self-monitoring of diabetes mellitus. Diabetes Meta Syndr. 2018;12(2):181-87. doi:10.1016/j.dsx.2017.09.005
Key words: Self-monitoring blood glucose, Continuous glucose monitoring, personal continuous glucose monitors, professional continuous glucose monitors