Nurses are critical to recognizing and managing this life-threatening condition.
Learning Objectives
- Identify the pathophysiology of differentiation syndrome (DS).
- Compare DS with tumor lysis syndrome.
- Discuss DS management.
The authors and planners of this CNE activity have disclosed no relevant financial relationships with any commercial companies pertaining to this activity. See the last page of the article to learn how to earn CNE credit.
Expiration: 1/1/25
Takeaways:
- Differentiation syndrome (DS) is an emerging and potentially life-threatening adverse reaction of isocitrate dehydrogenase inhibitors such as ivosidenib used in the treatment of acute myeloid leukemia.
- Frontline oncology nurses are essential in the assessment, monitoring, and comprehensive management of DS.
- Patient education about DS is aimed at detecting and preventing complications.
John Delaney* is an 85-year-old man with a past history of type 2 diabetes, hyperlipidemia, cigarette smoking (1 pack per day for 60 years; quit 3 years ago), cataract surgery, and recent relapse of acute myeloid leukemia (AML). Originally, the AML was treated with five cycles (1-hour infusions for 5 days) of decitabine. His current medications include prophylactic antifungal (posaconazole) and antiviral (valacyclovir) agents, an oral hypoglycemic (metformin), multivitamins, ascorbic acid, vitamin-D3, and ivosidenib.
Mr. Delaney arrives in the emergency department (ED) with shortness of breath, fever, dizziness, and near-syncopal episodes. On admission to the hematology-oncology unit, his vital signs are temperature 38° C (100.4° F), heart rate 73 beats per minute (bpm), respiration rate 18 breaths per minute, SpO2 96% on room air, and blood pressure 132/58 mmHg. Initial blood work reveals the following: white blood count (WBC) 10.6×103 uL (normal range: 3.4–11.2×103 uL), hemoglobin 8.8 g/dL (normal range: 13.3–17.7 g/dL), hematocrit 25.1% (normal range: 40%–50%), platelets 17,000 (normal range: 150–450×103 uL), and blasts 59% (normal: <5%). The chemistry panel is normal except for a blood urea nitrogen of 30 mg/dL (normal range: 8–20 mg/dL) and creatinine of 1.3 mg/dL (normal range: 0.64–1.27 mg/dL). The oncologist makes a preliminary diagnosis of differentiation syndrome (DS).
DS, previously known as retinoic acid syndrome, is an oncologic emergency that can occur in patients with acute promyelocytic leukemia (APL) and AML. Patients at high risk include those with a high body mass index and active leukemia. A Food and Drug Administration (FDA) study conducted by Norsworthy and colleagues reported that 30- and 60-day mortality were similar in patients who had DS vs those who didn’t. Nurses can help manage patients with DS if they understand its etiology, pathophysiology, and treatment options. (See The ABCs of AML.)
The ABCs of AML

Acute myeloid leukemia (AML) is characterized by infiltration of bone marrow, blood, and other tissues by proliferative and abnormally differentiated hematopoietic cells. Isocitrate dehydrogenase (IDH) mutations disrupt metabolic and epigenetic mechanisms responsible for cellular differentiation and are believed to be an early and critical contributor to oncogenesis. According to DiNardo and colleagues, an estimated one in five AML cases is IDH-mutant, with IDH2-mutant AML being more prevalent than IDH1-mutant AML.
Known risk factors for AML include age, male gender, tobacco smoking, chemical exposure to benzene, other cancers or disorders treated with alkylating agents or topoisomerase II inhibitors, radiation exposure, family history of leukemia, and certain genetic syndromes and blood disorders. In Mr. Delaney’s case, advanced age and smoking may have contributed to his AML.
In 2015, Döhner and colleagues reported that treatment advances in the past 2 decades have resulted in a 35% to 40% cure rate in adult patients younger than 60 years and a 5% to 15% cure rate in those older than 60.
AML treatment
Evidence-based AML treatment consists primarily of chemotherapy but also can include targeted therapy and stem cell transplant. Surgical and radiologic therapies, although not typically used to treat AML, may be used to treat symptoms or complications.
Isocitrate dehydrogenase (IDH) inhibitors (ivosidenib and enasidenib) comprise a newer class of targeted chemotherapeutic agents that inhibit one of the two mutant IDH proteins, promoting differentiation of leukemic myeloblasts. Ivosidenib tablets were approved by the FDA in 2018 as first-line treatment for AML with a susceptible IDH1 enzyme mutation. Blocking the enzyme reduces myeloblast counts and increases the number of mature myeloid cells. Enasidenib is approved for relapsed or refractory mutant IDH2 AML.
Ivosidenib is indicated for adult patients with newly diagnosed AML who are 75 years or older, those who have comorbidities that preclude intensive induction chemotherapy, and adults with relapsed or refractory AML.
Mr. Delaney has been taking ivosidenib, 500 mg daily by mouth, for 6 months and was tolerating the treatment, until a week before admission when he began tiring easily and experiencing dyspnea on exertion. He also noted bilateral leg edema.
DS pathophysiology
To understand DS, consider cellular differentiation (the process by which a cell changes from one type to another). Typically, this change occurs from a less specialized type (such as stem cells) to a more specialized type (such as platelets). In AML, abnormal differentiation of myeloid cells develops. IDH inhibitors promote the myeloblast differentiation, which can lead to myriad symptoms (or DS).
Patients can progress to DS spontaneously or while undergoing induction therapy using all-trans retinoic acid (ATRA), arsenic trioxide (ATO), chemotherapy, or various targeted therapies such as ivosidenib, enasidenib, and gilteritinib. Frankel and colleagues reported DS incidence of 15% to 25% for patients receiving ATRA. According to the FDA, clinical trials of ivosidenib resulted in DS in 25% of patients treated for new AML and in 19% of patients treated for relapsed or refractory AML.
During induction therapy (the first phase of chemotherapy, when a combination of two or three agents is used), leukemic blasts present in large numbers, inducing cellular migration, endothelial activation, and release of interleukins and vascular factors, resulting in tissue damage. Although the pathophysiology of DS is still not fully understood, key features include cytokine storms coinciding with differentiation of blast cells. DS diagnosis is largely based on clinical presentation and imaging findings.
Mr. Delaney’s chest x-ray shows bilateral pleural effusions. He’s given 20 mg of I.V. furosemide, and he voids 400 mL of urine in 4 to 5 hours. Throughout his first shift on the unit, he uses 4 liters of nasal oxygen intermittently with dyspnea on exertion. His maximum temperature is 39° C (102.2° F), and he experiences increased work of breathing. A repeat CBC shows his WBC count is 26x103uL and blasts of 65%.
Clinical and radiologic findings
Onset of DS clinical signs and symptoms vary from days to weeks after induction therapy. According to DiNardo and colleagues, ivosidenib-induced DS occurs within a median of 29 days. Clinical features include pulmonary infiltrates, fever, weight gain, pleural and pericardial effusion, renal failure, cardiac failure, and hypotension. Patients with two or three of these clinical features are considered to have moderate DS, whereas those with at least four are considered to have severe DS. Leukocytosis, anemia, thrombocytopenia, and coagulopathy are commonly associated with AML-induced DS, which may coincide with chemotherapy.
For IDH inhibitor–induced DS, symptom onset can occur as early as 1 day or as late as 3 months after starting treatment. The manifestations resemble those of induction-induced DS, and leukocytosis is notable. Radiologic findings may include pulmonary opacities, pleural effusion, and pericardial effusion due to cytokine-mediated inflammatory response. In addition, prolongation of the QTc interval has been documented in patients with AML who are prescribed ivosidenib, as seen in Mr. Delaney’s case.
Fever, peripheral edema, and cardiac and pleural effusions can occur in patients with IDH inhibitor–induced DS. These patients should be assessed for hypoxia, dyspnea, and pulmonary infiltrates and opacities. A chest x-ray and an echocardiogram will help determine the severity of pulmonary and cardiac effusions and rule out other conditions such as sepsis and pneumonia.
IDH inhibitor–induced DS also can lead to tumor lysis syndrome (TLS) in patients with AML. TLS is an oncologic emergency caused by metabolic imbalance during tumor breakdown after initiation of cytotoxic therapy. Laboratory findings related to released metabolites from lysed tumor cells include hyperuricemia, hyperkalemia, hyperphosphatemia, hypocalcemia, and uremia. These findings put the patient at risk for acute renal failure, cardiac arrhythmias, seizures, and death.
TLS and DS differ in their etiology and clinical presentation. TLS is defined as a massive and sudden release of cellular contents into the bloodstream caused by cell lysing. This differs from the DS etiology of large-scale cellular differentiation. Acute kidney injury may be seen in both TLS and DS but develops later in DS. (See DS and TLS: Key differences.)
Differentiation syndrome (DS) and tumor lysis syndrome (TLS), both oncologic emergencies, have several key differences.
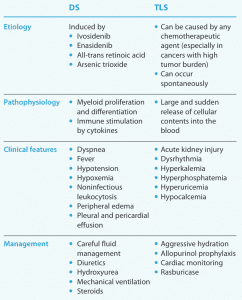

TLS is ruled out in Mr. Delaney’s case because of the lack of tell-tale hyperkalemia and hyperuricemia. In addition, his overall clinical picture is more consistent with DS.
In a study by Roboz and colleagues, noninfectious leukocytosis was noted in patients with AML who were receiving ivosidenib therapy (27% of patients were diagnosed with or without co-occurring DS). A WBC count >25×103 uL or an absolute increase in total WBC >15×103 uL from baseline might signal the presence of noninfectious leukocytosis.
Mr. Delaney’s WBC increases from 10.6 x103 uL to 26×103 uL, an increase of 15.4×103 uL, which meets the current criteria for noninfectious leukocytosis. Additional proof is provided by the lack of any infectious organisms in Mr. Delaney’s blood and urine cultures. The care team treats the noninfectious leukocytosis with hydroxyurea, which induces cytoreduction. The drug disrupts DNA synthesis, alters adhesion molecules of endothelial cells, and lowers WBC counts, which is especially effective in myeloproliferative disorders such as AML.
Management
Comprehensive DS care centers around controlling inflammatory response, maintaining hemodynamic stability, averting a crisis, and symptom management.
On day 2, Mr. Delaney is given 2,000 mg hydroxyurea orally twice and started on 10 mg of I.V. dexamethasone every 12 hours. He is afebrile for the next 72 hours and stabilizes clinically until he experiences chest heaviness and a near-syncopal episode. A 12-lead ECG shows sinus rhythm at 92 bpm with a QTc interval of 580 ms (normal in men is ≤430 ms; in women, ≤450 ms). The cardiologist recommends telemetry monitoring.
When DS is suspected, dexamethasone 10 mg (I.V. or by mouth) twice daily is initiated. If the patient improves clinically within 12 to 24 hours, dexamethasone is continued for 3 or more days, and then doses are tapered. If no improvement is observed, dexamethasone is increased to 10 mg every 6 hours. If clinical features worsen, the full dexamethasone dose should be continued, differentiation agents (for example, ATRA and ATO for APL, and IDH inhibitors for AML) discontinued, and management of possible new clinical DS features initiated. If improvement continues for more than 3 days, dexamethasone is tapered and differentiation agents are continued. If no improvement is observed for more than 3 days, full dose dexamethasone and supportive care are continued until the symptoms improve. When DS has resolved, dexamethasone is tapered and differentiation agents are resumed.
Patients with co-occurring leukocytosis are at risk for TLS and organ damage. To control leukocytosis, in addition to steroid therapy, hydroxyurea 2 to 4 g orally (titrated daily) should be initiated.
Patients with QTc prolongation are at risk for more serious ventricular dysrhythmia such as torsade de pointes and ventricular tachycardia. Medication reconciliation is recommended to determine if the patient is taking other medications (such as ondansetron) that can prolong the QT interval. If possible, a cardiology consult can be initiated to fully evaluate the patient.
For patients with co-occurrence of fevers and hypotension, initiation of empiric antibiotics and fluid management may be needed. Patients in fluid overload may benefit from cautious diuretic therapy such as with furosemide.
On day 3, after receiving two daily doses of ivosidenib in conjunction with dexamethasone, Mr. Delaney’s WBC peaks at 26×103 uL and his blast percentage is 65. His overall clinical status declines with vital signs as follows: temperature 39° C (102.2° F), heart rate 106 bpm, respiration rate 35 breaths per minute, Sp02 100% on 50% oxygen by Venturi mask, and blood pressure 90/50 mmHg.


Nurse continuing education
Continuing nursing education related to emergencies, adverse drug events, and novel chemotherapy and biotherapy is essential for both experienced oncology nurses and those new to the specialty.
Simulations designed in collaboration with pharmacology and oncology teams can help nurses manage these events. In addition, interprofessional rounds facilitate awareness among all disciplines of novel therapies and their inherent reactions. With increasing understanding of emerging adverse drug events, new protocols and treatment algorithms may be developed via interdisciplinary quality improvement efforts.
Nursing care
Specific nursing care depends on DS severity but should focus on control of fever, fluid management, supplemental oxygen and mechanical ventilation, antibiotic administration, vasopressor support, and cardiac monitoring. The primary nurse also can effectively advocate for the patient by monitoring for adverse drug effects, such as leukocytosis and prolonged QTc. Physical assessments, daily weights, strict input and output monitoring, daily 12-lead ECGs to check for prolonged QTc, and medication reconciliation can help determine the need for further supportive care.
To control Mr. Delaney’s fever, the nurse initiates nonpharmacologic cooling measures and provides acetaminophen. He’s kept on 50% oxygen by Venturi mask, the head of his bed is elevated, and incentive spirometry is encouraged to optimize pulmonary function. The nursing staff encourages him to eat and take prescribed supplements. Physical and occupational therapy is initiated to maintain Mr. Delaney’s functional status and prevent deconditioning.
On day 4, Mr. Delaney shows worsening hemodynamic status that doesn’t respond to fluid resuscitation. He’s transferred to the intensive care unit (ICU) where continued steroids, vasopressors, and mechanical ventilation ultimately resolve the DS, and he’s subsequently discharged home.
Critical to care
DS is a potentially life-threatening complication of IDH-inhibitors. Nurses are critical to the comprehensive management of patients with DS, including developing and implementing protocols and treatment algorithms. As patient advocates, nurses ensure patients and families can monitor for signs and symptoms of DS and report them to the care team. (See Ivosidenib and DS: Patient education.)
Ivosidenib and DS: Patient education
Cover the following information when teaching patients and their families about ivosidenib and differentiation syndrome (DS):
- Take ivosidenib with or without food.
- Maintain adequate fluid intake.
- Follow up with blood chemistry monitoring.
- Avoid breastfeeding during treatment and for at least 1 month after the final dose of ivosidenib.
- Swallow tablets whole. Do not split, crush, or chew them.
- Avoid taking ivosidenib with a high-fat meal.
- If a dose of ivosidenib is vomited, do not take an additional dose. Wait until the next scheduled dose.
- If a dose of ivosidenib is missed or not taken at the usual time, take the dose as soon as possible unless the next dose is due within 12 hours.
- Immediately report any symptoms suggestive of DS, such as fever, cough or difficulty breathing, rash, decreased urinary output, low blood pressure, rapid weight gain, or swelling of arms or legs.
- Report dizziness, lightheadedness, and fainting to your healthcare provider. (These may be caused by significant QTc interval prolongation.)
- Report weakness or tingling sensation in the legs, arms, or upper body to your healthcare provider. (These may indicate Guillain-Barré syndrome.)
Encourage patients to ask questions and take an active role in monitoring their response to treatment and watching for potential adverse drug effects. Explain that they can voluntarily report observed or suspected adverse drug effects at www.accessdata.fda.gov/scripts/medwatch.
Daniel Borski is a senior staff nurse at New York Presbyterian Weill Cornell Hospital in New York City, New York. Ivan Biglang-awa is a staff nurse at Montefiore Medical Center in Bronx, New York. Fidelindo Lim is a clinical associate professor at New York University Rory Meyer College of Nursing in New York City, New York.
*Name is fictitious.
References
Belay Y, Yirdaw K, Enawgaw B. Tumor lysis syndrome in patients with hematological malignancies. J Oncol. 2017;2017:9684909. doi:10.1155/2017/9684909
Birendra KC, DiNardo CD. Evidence for clinical differentiation and differentiation syndrome in patients with acute myeloid leukemia and IDH1 mutations treated with the targeted mutant IDH1 inhibitor, AG-120. Clin Lymphoma Myeloma Leuk. 2016;16(8):460-5. doi:10.1016/j.clml.2016.04.006
De Kouchkovsky I, Abdul-Hay M. Acute myeloid leukemia: A comprehensive review and 2016 update. Blood Cancer J. 2016;6(7):e441. doi:10.1038/bcj.2016.50
DiNardo CD, Stein EM, de Botton S, et al. Durable remissions with ivosidenib in IDH1-mutated relapsed or refractory AML. N Engl J Med. 2018;378:2386–98. doi:10.1056/NEJMoa1716984
Döhner H, Weisdorf DJ, Bloomfield CD. Acute myeloid leukemia. N Engl J Med. 2015;373(12):1136-52. doi:10.1056/NEJMra1406184
Food and Drug Administration. FDA approves ivosidenib as first-line treatment for AML with IDH1 mutation. May 3, 2019. fda.gov/drugs/resources-information-approved-drugs/fda-approves-ivosidenib-first-line-treatment-aml-idh1-mutation
Fathi AT, DiNardo CD, Kline I, et al. Differentiation syndrome associated with enasidenib, a selective inhibitor of mutant isocitrate dehydrogenase 2: Analysis of a phase 1/2 study. JAMA Oncol. 2018;4(8):1106-10. doi:10.1001/jamaoncol.2017.4695
Frankel SR, Eardley A, Lauwers G, Weiss M, Warrell RP Jr. The “retinoic acid syndrome” in acute promyelocytic leukemia. Ann Intern Med. 1992;117(4):292-6. doi:10.7326/0003-4819-117-4-292
Golub D, Iyengar N, Dogra S, et al. Mutant isocitrate dehydrogenase inhibitors as targeted cancer therapeutics. Front Oncol. 2019;9:417. doi:10.3389/fonc.2019.00417
Li K, Vo K, Lee BK, Addo N, Coralic Z. Effect of a single dose of I.V. ondansetron on QTc interval in emergency department patients. Am J Health Syst Pharm. 2018;75(5):276-82. doi:10.2146/ajhp161070.
Mamez AC, Raffoux E, Chevret S, et al. Pre-treatment with oral hydroxyurea prior to intensive chemotherapy improves early survival of patients with high hyperleukocytosis in acute myeloid leukemia. Leuk Lymphoma. 2016;57(10):2281-8. doi:10.3109/10428194.2016.1142083
McMahon CM, Canaani J, Rea B, et al. Gilteritinib induces differentiation in relapsed and refractory FLT3-mutated acute myeloid leukemia. Blood Adv. 2019;3(10):1581-5. doi:10.1182/bloodadvances.2018029496
NIH Stem Cell Information Home Page. National Institutes of Health, U.S. Department of Health and Human Services. 2016. https://stemcells.nih.gov
Norsworthy KJ, Mulkey F, Scott EC, et al. Differentiation syndrome with ivosidenib and enasidenib treatment in patients with relapsed or refractory IDH-mutated AML: A U.S. Food and Drug Administration systematic analysis. Clin Cancer Res. 2020;26(16):4280-8. doi:10.1158/1078-0432.CCR-20-0834
Olsen M, LeFebvre K, Brassil K. Chemotherapy and Immunotherapy Guidelines and Recommendations for Practice. Pittsburgh, PA: Oncology Nursing Society; 2019.
Pasquier F, Lecuit M, Broutin S, et al. Ivosidenib to treat adult patients with relapsed or refractory acute myeloid leukemia. Drugs Today (Barc). 2020;56(1):21-32. doi:10.1358/dot.2020.56.1.3078363
Röllig C, Ehninger G. How I treat hyperleukocytosis in acute myeloid leukemia. Blood. 2015;125(21):3246-52. doi:10.1182/blood-2014-10-551507
Roboz GJ, DiNardo CD, Stein EM, et al. Ivosidenib induces deep durable remissions in patients with newly diagnosed IDH1-mutant acute myeloid leukemia. Blood. 2020;135(7):463-71. doi:10.1182/blood.2019002140
Sanz MA, Montesinos P. How we prevent and treat differentiation syndrome in patients with acute promyelocytic leukemia. Blood. 2014;123(18):2777-82. doi:10.1182/blood-2013-10-512640
Schwartz PJ, Woosley RL. Predicting the unpredictable: Drug-induced QT prolongation and torsades de pointes. J Am Coll Cardiol. 2016;67(13):1639-50. doi:10.1016/j.jacc.2015.12.063
Shallis RM, Wang R, Davidoff A, Ma X, Zeidan AM. Epidemiology of acute myeloid leukemia: Recent progress and enduring challenges. Blood Rev. 2019;36:70-87. doi:10.1016/j.blre.2019.04.005
Key words:
Differentiation syndrome, Isocitrate dehydrogenase (IDH) inhibitors, Ivosidenib, Oncology