Many patients facing surgery for mitral valve disease now have an alternative to valve replacement: valve repair. The outcomes for mitral valve repair are similar to those for replacement. Plus, a repair often allows the patient to avoid a lifetime of anticoagulant therapy.
Causes of mitral valve disease
Changes in mitral valve function are caused by leaflet regurgitation or stenosis. These structural abnormalities usually result from one of four disease processes—rheumatic disease, valve degeneration, coronary artery disease, and infective endocarditis. Less common causes of mitral valve disorders include congenital abnormalities, trauma, chest radiation, and Marfan syndrome.
Rheumatic disease may affect the mitral valve after a beta-hemolytic streptococcal infection. The rheumatic process can produce regurgitation or stenosis.
Valve degeneration can develop with age. The gradual deterioration of the valve allows deposits of calcium and other fibrous substances on the valve leaflets and surrounding support structure.
Coronary artery disease reduces blood flow to the myocardial tissue, producing ischemia. Two anatomic changes can affect valve structure. Ischemic coronary artery disease can cause the left ventricle to enlarge, stretching the natural elliptical, or football, shape of the ventricles into a round, or basketball, shape. As the ventricle stretches, so does the mitral valve opening, resulting in mitral regurgitation. Ischemic coronary artery disease may also limit blood flow to the papillary muscle, the anchor for the chordae tendineae that tether the mitral valve leaflets. Valve leaflets not anchored by strong chordae can’t maintain complete closure during left ventricular contraction. The result is valve regurgitation that allows blood to flow backward from the left ventricle into the left atrium.
Infective endocarditis occurs when bacteria, usually streptococcal and staphylococcal bacteria, attach to the lining of the mitral valve leaflet. The bacteria cause large vegetations on the valve. Bacterial invasion may produce abscesses in the valve (or the heart chambers), erode the chordae tendineae, or tear the valve leaflet.
Abnormal structure and function
Mitral stenosis is characterized by narrowing of the annulus (the ring-shaped structure supporting the leaflets), distortion of the leaflets, and fusion of the valve edges. Leaflet movement is limited. The stenotic valve gradually narrows the path of blood leaving the left atrium. In response, atrial wall thickness increases to generate more power to push blood through the stenotic valve. If the atrium can’t increase its wall strength, the stenosis may cause atrial dilation and pulmonary congestion, putting the patient at risk for atrial fibrillation. (See A look at normal and abnormal mitral valves.)
In mitral regurgitation, one or both leaflets may enlarge and move excessively when the valve opens and closes. The resulting annular dilation doesn’t allow the leaflets to close completely when ventricular contraction ends, producing a backflow of blood into the left atrium. The results are congestion and an enlarged left atrium. An enlarged left ventricle or left atrium can’t empty completely, resulting in a vicious circle of enlargement and congestion.
Mitral valve repairs
According to the current guidelines by the American Heart Association and the American College of Cardiology, the preferred surgical option for mitral valve disease is valve repair, which can correct both mitral stenosis and regurgitation. Repairing a mitral valve is more demanding than replacing one. A surgeon must be able to reshape and reconstruct the malformed leaflets and annulus, so the repaired valve is as functional as it was before the disease. By contrast, removing a diseased valve and replacing it with a prosthetic one is more straightforward.
Before the repair procedure, a surgeon orders echocardiography, so he or she can analyze the valve, leaflet movement, and the size of the opening during left atrial contraction. To repair the valve, a surgeon may use a median sternotomy, a partial sternotomy, or a thoracotomy. An open surgical approach remains the gold standard for this surgery.
Mitral stenosis from calcification or infection produces inflexible or fused leaflets. Surgical repair can include removing calcium deposits and correcting damaged leaflets with pericardial patches. Stiff, stenotic leaflets may be reshaped or separated, so the mitral annulus allows better blood flow.
In mitral regurgitation, the valve leaflets may be enlarged or stretched and may billow and leak when the left ventricle contracts in systole. Stretched or misshapen leaflets can be reshaped, and excess valve tissue can be resected to achieve a more normal shape. In ischemic coronary artery disease, the chordae tendineae may be stretched or ruptured. Without the tether of the chordae tendineae, leaflets may prolapse into the left atrium or block outflow from the left ventricle. Surgical repair may include transferring the support of a valve leaflet from a ruptured chord to a patent one. Or a surgeon may substitute suture material for a ruptured chord. As an alternative, a surgeon may choose Gore-Tex material, which will not stretch or deteriorate like the patient’s tissue.
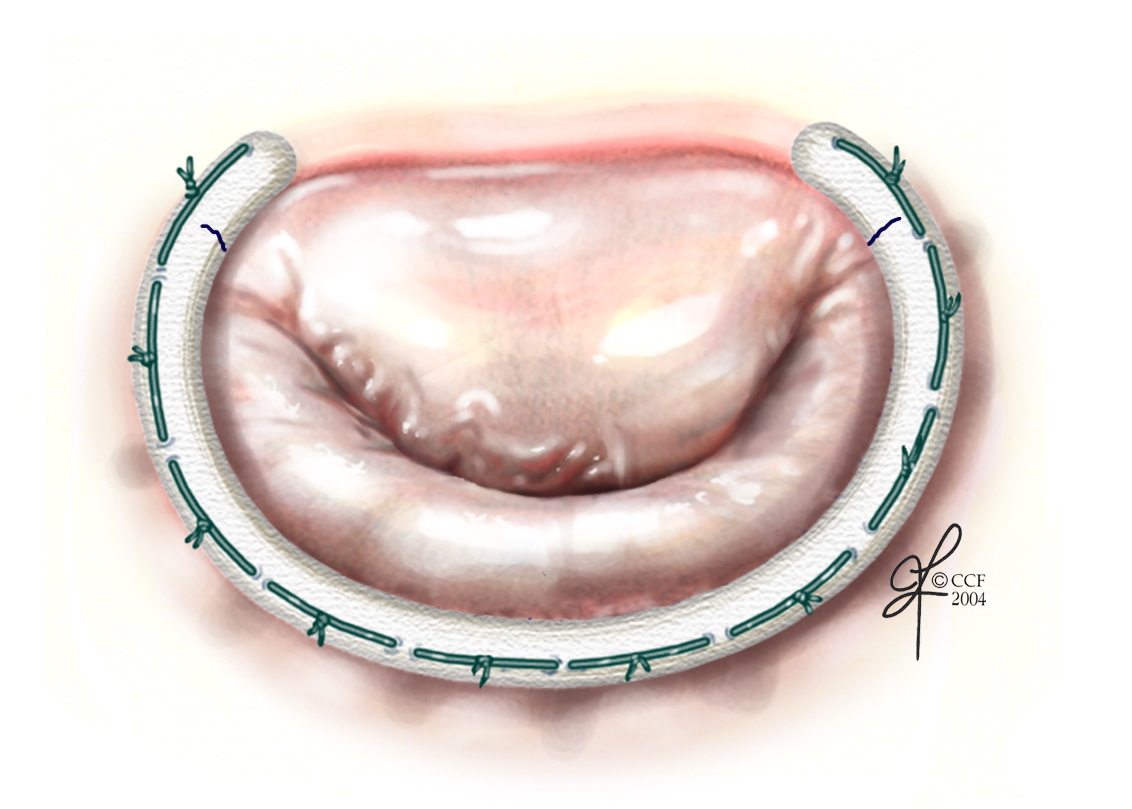
Valve repair surgery may also include shoring up and reducing the size of a dilated annulus. Inserting a supportive ring around the annulus helps restore its natural shape. (See Fixing the chordae tendineae and annulus.)
Less invasive techniques
Mitral valve repair combining the expertise of interventional cardiology and cardiac surgery is being refined in clinical studies. Here are three promising approaches:
- balloon valvuloplasty, in which a balloon is placed inside the stenotic mitral valve, then inflated and deflated several times to widen the valve opening
- a clip that can attach regurgitant mitral leaflets and allow the valve to close completely
- an annuloplasty ring that can be placed by way of the coronary sinus to correct regurgitation.
Advantages of mitral valve repair
After 5 years, the durability of a mitral valve repair compares favorably with that of mitral valve replacement. Plus, repair allows many patients to avoid life-long anticoagulant therapy and the risk of hemorrhage. By keeping their natural valve, patients also reduce their risk of infective endocarditis. And, of course, repair avoids the complications associated with prosthetic valve replacements, such as thromboembolism and structural failure. Repair patients with underlying infective endocarditis become more resistant to repeat infections.
If mitral valve regurgitation results from ischemia, the repair won’t prevent left ventricular remodeling and dysfunction from coronary artery disease, and regurgitation may recur. But the mortality risk from coronary artery disease decreases with mitral valve repair.
Nursing care
Postoperative nursing care for patients undergoing valve repair is similar to nursing care for other cardiothoracic surgeries. During the acute recovery phase, you monitor the patient for unstable hemodynamic values, bleeding, arrhythmias, surgical-site infection, and complications from mechanical ventilation.
The risk of postoperative complications is lower than or similar to the risk after valve replacement. Patients who have mitral valve repair for ischemic coronary artery disease face the same risk of bleeding and stroke as those who have valve replacement. A patient’s risk of atrial fibrillation is 27% for either procedure.
Patient teaching
The stress of surgery and hospitalization makes it likely that patients and families will forget much of what you teach them preoperatively. Anticipate repeating the details of the valve repair during this emotionally intense period.
If an annuloplasty ring is implanted, the patient will need short-term anticoagulant therapy until endothelial tissue surrounds the ring. A patient with a condition such as atrial fibrillation may also need anticoagulant therapy. Teach these patients about the anticoagulant and the importance of taking it as prescribed.
A patient and family may think that because the surgery is “only a repair,” the postoperative recovery will be uneventful. If so, you need to dispel this myth. Be sure to translate the technical details of surgery. Also, explain the need for an intensive care unit stay and the expectations for the recovery period. Make sure the patient and family know the possible postoperative complications: new-onset atrial fibrillation, delayed weaning from mechanical ventilation, and impaired cardiac output.
Better options
Today, mitral valve repair using an open surgical approach may be a better option than mitral valve replacement for many patients. And in the future, mitral valve repairs using less invasive procedures may be even better options for patients with mitral valve stenosis and regurgitation.
Selected references
ACC/AHA 2006 guidelines for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 2006;114:84-231.
Gillinov AM, Faber C, Houghtaling PL, et al. Repair versus replacement for degenerative mitral valve disease with coexisting ischemic heart disease. J Thorac Cardiovasc Surg. 2003;125:1350-1362.
Rankin J, Hammill B, Ferguson T, et al. Determinants of operative mortality in valvular heart surgery. J Thorac Cardiovasc Surg. 2006;131:547-557.
Savage E, Ferguson T, DiSesa V. Use of mitral valve repair: analysis of contemporary United States experience reported to the Society of Thoracic Surgeons national cardiac database. Ann Thorac Surg. 2003;75:820-825.
Topol EJ, ed. Textbook of Cardiovascular Medicine. 3rd ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2007.
Visit www.AmericanNurseToday.com/journal for a complete list of selected references.
Kathleen M. Hill is the clinical nurse specialist for the surgical intensive care unit at the Cleveland Clinic in Ohio.