Reinforce safe practices in your organization.
Editor’s Note: This is an early release of a web exclusive article for the July 2021 issue of American Nurse Journal.
The Centers for Disease Control and Prevention (CDC), Institute for Safe Medication Practices (ISMP), and the Infusion Nurses Society (INS) agree that syringes and needles should be used only once on only one patient and that aseptic technique is critical to preventing microorganism transmission.
The concept of one and done also applies to the use of prefilled flush syringes (PFS), which have been available since 1999 and are used each time an I.V. catheter is accessed for maintenance flushes or to administer medications. The safety benefits of PFS over manually prepared flush syringes include reducing I.V. catheter occlusions, catheter-related bloodstream infections, and needle stick injuries.
Misunderstandings about the indicated uses for PFS became evident when the ISMP reported that nurses were using them to reconstitute and dilute I.V. push medications. The CDC and ISMP also reported cases where the same PFS were used for multiple patients, which caused infectious outbreaks. But what about reusing a PFS on the same patient? The Infusion Therapy Standards of Practice for flushing and locking recommends that “a syringe or needle/cannula should be considered contaminated once it has been used to enter or connect to a patient’s I.V. solution container or administration set.”
What’s the risk?
Reusing a syringe on the same patient carries the risk of touch contamination, which occurs when patient care activities contaminate healthcare worker’s hands. Microorganisms transfer from high-touch surfaces in the patient zone to caregivers’ hands, which then contaminate other pieces of patient care equipment, including syringes.
The mechanism to transfer microbes on hands to other objects is the same whether or not gloves are used. A contaminated gloved hand offers little protection to the patient. Studies of anesthesia providers and pharmacy compounding staff demonstrate high contamination rates when the hub of a syringe touches nonsterile environmental surfaces and fingers, which is likely to happen when a nurse manipulates a PFS or places it uncapped on surfaces. Recapping syringe tips can increase the contamination rate as well because of syringe tip contact with a contaminated finger or the environment.
PFS can become contaminated in many ways during appropriate use. Partially used PFS frequently are placed uncapped on surfaces immediately adjacent to the nurse and the I.V. catheter—the bed, pillow, top of patient, bedside table, bedside cart, or even a computer keyboard. (See Mechanisms of contamination.)
Mechanisms of contamination
A prefilled flush syringe (PFS) can be contaminated during routine use, including when
• slipping off the connector with one hand
• re-using the flush
• uncapping early and placing uncapped syringe on nearby surface or alcohol swab
• holding multiple syringes
• recapping the PFS with the tip cap
• uncapping the PFS, opening the alcohol swab, and moving around room carrying the uncapped syringe
• placing or dropping the PFS on bedding
• touching the syringe tip to the connector before swabbing the connector.
Prefilled flush syringe survey
The authors conducted a short survey to learn more about the I.V. catheter flushing practices of RNs in the United States. The survey included questions about flushing practices for short peripheral catheters (SPC), peripherally inserted central catheters, central venous catheters, implanted ports, and hemodialysis catheters, all of which are frequently encountered in acute care settings. The survey was sent to a pre-qualified panel selected from the relevant membership segment of a national nursing organization. Participants received a survey if they were a staff nurse, working in a hospital setting on any type of unit, either full-time or part-time. We closed the survey after receiving 100 complete responses.
Demographics
The survey respondents were evenly distributed across U.S. Census regions. Slightly more than one-half (53%) were at least 45 years old. About two-thirds (67%) had a bachelor of science in nursing, and about two-thirds (67%) were employed in acute care settings (intensive care units, emergency departments, med-surg, stepdown, and ambulatory care). Fifty-three percent of respondents had been in nursing for more than 10 years, and 37% had more than 20 years of nursing experience.
PFS size and number
Nurse respondents nearly always used a 10 mL PFS when flushing a central catheter (92% to 96%); 23% used either 3 mL (12%) or 5 mL (11%) PFS with the SPC. This most likely reflects products available and the nurses’ knowledge of the appropriate volume to flush a central catheter versus an SPC.
Flushing practices
Nurses were asked about their practices when giving a single I.V. push medication. More than half of the respondents (53%) said they use only one flush syringe if giving an I.V. medication via SPC; nearly three-quarters would use two PFS when giving one I.V. push medication via any central line. Approximately one-third of nurses “do not always” flush the SPC before administering an I.V. push medication, but nearly all (81% to 92%) flush before I.V. push administration via a central line. Respondents almost always (96% to 99%) flush after an I.V. push medication, regardless of line type. Most (83% to 90%) flush between medications when giving multiple I.V. push medications.
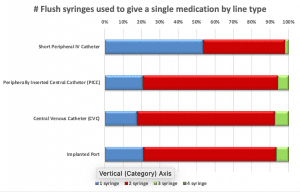

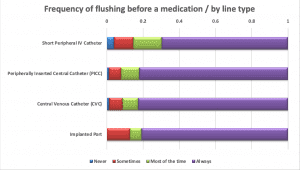

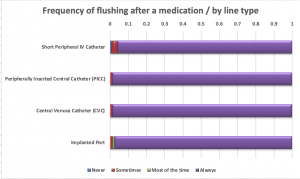

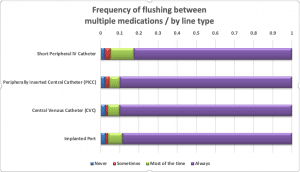

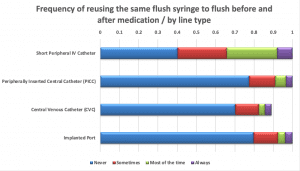
PFS reuse
Only 40% of nurses indicated that they would never reuse a PFS to flush an SPC before and after a medication; 34% noted that PFS reuse was their practice “most or all of the time.” More respondents said they would never reuse the same PFS to flush before and after an I.V. push medication via central line (70% to 83%), but the percentages were still concerning.

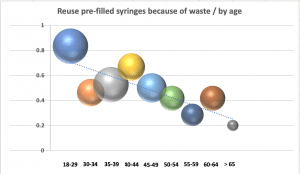
Nurses were asked to select all the reasons why they might reuse the same PFS to flush before and after a medication on the same patient. Saving time (36%) and saving money (25%) were frequently cited, but the reason given most often was to reduce the amount of plastic/waste (50%). Further analysis reveals generational differences about the importance of protecting the environment, with younger nurses more likely to be focused on reducing plastic and waste.
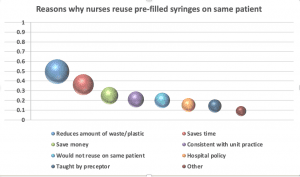


Nearly all respondents (95%) said they would never reuse a PFS for more than one patient.
Results analysis
Much has been written about inappropriate PFS use to reconstitute or dilute I.V. push medications; however, this is the first time nurses have been asked about how they use the PFS when administering I.V. push medications. (The survey included questions about hemodialysis catheters, but more than 80% of participants responded “Not applicable” to the questions, so they were excluded from the analysis and discussion.)
Many hospitals stock 10 mL size PFS because they’re efficient and effective for flushing all sizes of I.V. devices. The survey respondents commented that they reuse the 10 mL size PFS especially for SPCs, because they use only 3 to 5 mL to flush before and after I.V. push medications. The smaller volume PFS can be used for pediatric patients and adult SPCs.
The survey responses indicate that 68% of nurses always flush before and 96% always flush after I.V. push medication administration, but only 47% use more than one PFS when administering an I.V. push medication via SPC. This is a significant practice concern because of the likelihood of touch contamination to the tip of the PFS. The 2021 INS Standards make the following practice recommendation for flushing and locking all vascular access devices: “A syringe or needle/cannula should be considered contaminated once it has been used to enter or connect to a patient’s I.V. solution container or administration set.” In other words, PFS shouldn’t be reused on the same patient, regardless of access type. This recommendation was sound advice from the CDC’s One and Only Campaign in 2010 and remains unchanged today.
The risks of microbial contamination during PFS reuse exists whether the syringe is recapped because the syringe is most likely placed on a high-touch surface during medication administration. Nurses said they reuse the PFS to reduce the amount of plastic/waste, to save time, and to save money. Does the decision to reuse PFS represent a conflict between multiple competing values in the workplace, or is it an example of deviance normalization reinforced by the perception that no harm is done? The survey respondents clearly recognize the risks inherent in reusing a PFS on more than one patient, but they don’t acknowledge or understand the harm from touch contamination for the same patient.
The survey also revealed an unanticipated finding: Nearly one-third of nurses did not routinely flush an SPC prior to medication administration, but they almost always flushed before administering a medication via a central line. The INS standards for flushing and locking don’t differ based on the line type. The responses raise several questions: Do nurses consider an SPC less of a risk for hospital-acquired infections? Has the emphasis on central line–associated blood stream infections inadvertently reduced the perception of risks with SPCs? Is this an issue of knowledge, attitudes, or motivation? Although the focus of this article is on syringe reuse, the findings about flush practices raise concerns and the need for further study.
Protecting the environment is clearly the most important reason for reusing PFS, especially for younger nurses. This is notable for two reasons. First, healthcare organizations need to include environmental protections into the criteria for product selection. Second, clear implications exist for medical device manufacturers to continue to refine and adapt materials that will benefit the planet.
Indications for practice
Based on the results of this survey, nurses and organizations should consider taking steps to reinforce proper PFS use.
Products
Nurses are reusing PFS because they don’t need the full amount to flush, especially with SPCs. Hospitals can re-evaluate which are the appropriate PFS sizes to stock based on patient populations and consider whether smaller volume syringes are needed to support safe practices.
Policies
Although very few nurses cited hospital policy as a reason to reuse PFS, the survey results should be a call to action to review existing policies and procedures. Safe injection practices should reinforce the current evidence and expert guidance from the CDC and INS.
Practice
How are nurses practicing on your unit or in your hospital? Is a unit norm placing patients at risk? If you’re a preceptor, how are you reinforcing best practices with new staff?
Initial and ongoing staff education should include messaging for safe injection practices and expectations for accountability. Infection control and leadership rounding are opportune times to engage staff in conversations that support safe PFS use. In addition, leaders should evaluate the need for additional resources, develop a plan to monitor staff adherence, and consider PFS reuse when investigating bloodstream infections.
A secondary finding from the survey reveals that a gap exists between best practice and actual flushing practices. Nurses aren’t consistently flushing before and after medication administration, especially when using SPCs. This should be a cause for concern to nursing leaders and a reason to examine the organization’s practices.
Indications for education
Nursing students must be taught best PFS practices, especially now when many students are receiving less clinical time in hospitals due to the pandemic and simulation is taking on a larger role. When student nurses are allowed clinical time with a hospital preceptor, that preceptor may unintentionally teach the nursing student that it’s appropriate to use the same PFS to flush an SPC before and after giving an I.V. push medication. If nursing students are taught best practices in nursing programs, they can be a catalyst for change in hospitals across the country.
Make safety a priority
Safe injection practices include the appropriate use of prefilled flush syringes. This survey revealed that nurses reuse PFS on the same patient, especially with SPCs. Many nurses may not perceive PFS reuse as a patient safety risk because they don’t see the immediate harm to patients. In addition, they may experience dissonance between workplace and personal priorities, leading them to reuse PFS out of concern for the environment, costs, and time. However, no other priority should supersede patient safety. The survey also revealed variations in practice for flushing vascular access devices. This report should serve as a catalyst to examine the practices in your setting and to ensure everyone understands that patient safety is the first priority.
Maureen Burger is chief nursing officer at Visante, Inc., in St. Paul, Minnesota. Candy Cross is a nurse educator at Chandler Gilbert Community College in Chandler, Arizona. Both authors are founding members of the National Coalition for IV Push Safety (ncivps.org).
Burger has received professional consulting fees from BD through her work with Visante. This article underwent peer review, and no bias was reported.
References
Centers for Disease Control and Prevention. One and Only Campaign. December 3, 2019. cdc.gov/injectionsafety/one-and-only.html
Gaunt MJ. An unsafe practice: Reuse of prefilled saline flush. Pharm Times. October 2018. pharmacytimes.com/publications/issue/2018/October2018/an-unsafe-practice-reuse-of-prefilled-saline-flush-
Gorski LA, Hadaway L, Hagle ME, et al. Infusion therapy standards of practice. J Infus Nurs. 2021;44(Suppl 1):S1-224. doi:10.1097/NAN.0000000000000396
Institute for Safe Medication Practices. Safe practice guidelines for adult IV push medications. July 23, 2015. ismp.org/guidelines/iv-push
Kwanten LE. Anaesthetists and syringe hygiene: Getting to the pointy end – A summary of recommendations. Br J Anaesth. 2019;123(4):e475-9. doi:10.1016/j.bja.2019.05.036
Stucki C, Sautter AM, Favet J, Bonnabry P. Microbial contamination of syringes during preparation: The direct influence of environmental cleanliness and risk manipulations on end-product quality. Am J Health Syst Pharm. 2009;66(22):2032-6. doi:10.2146/ajhp070681