Education and surveillance are key for patients at risk for this rare but fatal complication.
Takeaways:
- Progressive multifocal leukoencephalopathy (PML) is a rare demyelinating opportunistic viral infection of the central nervous system that occurs in the presence of a compromised immune system.
- PML has increased in the last decade due to a surge in immunosuppressive medications for various conditions.
- Treatment is still emerging, so awareness of risk factors and symptoms (cognitive changes, progressive weakness, visual and speech changes, ataxia, and hemiparesis) is critical.
- Nurses play an important role in the fight against this disorder by providing exemplary patient teaching.
Many of us tune out during commercials that list the rare side effects of a medication. However, understanding the side effects of disease-modifying and immunosuppressive drugs can help nurses recognize progressive multifocal leukoencephalopathy (PML), a rare and potentially fatal condition, and enhance patient education.
What is PML?
PML, a demyelinating opportunistic infection of the central nervous system, is caused by the John Cunningham (JC) virus in the presence of a compromised immune system. How PML develops isn’t well understood, but it can result in severe disability or death. Neurologic symptoms, which include weakness, paralysis, and mental status changes, vary depending on the location of lesions in the brain.
Between 30% to 90% of people harbor a latent version of the JC virus in their kidneys, bone marrow, or brain, but patients who are immunocompromised are at risk for virus reactivation. In some patients, the virus can infect the myelin-producing cells, which transmit nerve impulses in the brain. Physicians initially described PML in patients with lymphoproliferative cancers such as leukemia and lymphoma in the late 1950s.
In the past decade, disease-modifying treatments have successfully slowed progression of multiple sclerosis (MS) and other immune system diseases by preventing the inflammatory cascade. The success of these medications, which alter the immune system, has resulted in the widespread prescription of immunosuppressants. The result is an increased incidence of PML. The monoclonal antibody therapy natalizumab was the first disease-modifying drug reported to cause PML, but more cases have been detected as new drugs emerge. According to Garry and Kelly in the 2020 MS Research Update, the Food and Drug Administration (FDA) has approved 13 disease-modifying drugs to treat MS.
Risk factors
The relatively low risk of developing PML is associated primarily with disease-modifying drugs. For example, natalizumab blocks lymphocytes from migrating into the peripheral system, resulting in decreased central nervous system inflammation in patients with MS and intestinal endothelium inflammation in those with Crohn’s disease. These medications reduce the number of relapses in autoimmune disorders, but they also lead to immunodeficiency in some people, which increases the risk for JC virus reactivation. According to Berger, PML incidence varies from 1/30,000 with rituximab use in lymphoproliferative disorders (cancers such as lymphoma and leukemia) to 1/1,000 with natalizumab. Risk increases if the medication is prescribed for more than 2 years.
PML has increased in the past decade due to the surge in immunosuppressive medication use for various conditions. According to the 2020 MS Research Update, at least 14 FDA-approved drugs warn of increased PML risk. In addition to increased risk in patients treated with natalizumab, those with inflammatory bowel disorders, rheumatologic disorders, and plaque psoriasis treated with disease-modifying therapies also are at risk. Other at-risk individuals include those with leukemia, lymphoma, and HIV (because of underlying disease processes and treatments) and those with organ transplants (as a result of immunosuppressant medications). (See Medications associated with PML.)


Prognosis
PML prognosis depends on the underlying condition, how promptly PML is diagnosed, and the patient’s ability to mount an immune response. Maas and colleagues performed a retrospective study of 326 PML cases and discovered a death rate ranging from a low of 12% in patients with MS to as high as 83.3% in patients with cancer. Dunham and colleagues’ 2020 review reported that patients with HIV/AIDs who receive antiretroviral treatment have a mortality rate as low as 20%. The National Institute of Neurological Disorders and Stroke noted a 30% to 50% mortality rate for those diagnosed with PML. However, patients who survive are left with mild-to-severe neurologic deficits.
Signs and symptoms
PML symptoms vary depending on lesion location. They can include cognitive changes, progressive weakness, visual and speech changes, ataxia, and hemiparesis. Symptoms can be mistaken for a stroke or an MS exacerbation, creating diagnostic challenges. As the myelin progressively and quickly demyelinates, causing cell death, neurologic symptoms increase, eventually causing death unless the immune system can be restored. The earlier the disease is diagnosed, the better the prognosis, making excellent patient education imperative.
Diagnosis
PML diagnostic tests include magnetic resonance imaging (MRI) to detect white matter lesions in the brain and cerebrospinal fluid analysis to confirm the presence of the JC virus. However, spinal fluid may not always contain the virus DNA. In fact, Maas and colleagues’ study of 326 PML cases found that 22.7% of patients with PML had no JC virus DNA. If the clinical presentation and MRI point to PML but spinal fluid is negative, a brain biopsy may be ordered. However, depending on lesion location, surgery may negatively impact the patient’s quality of life. Small lesions may present on an MRI before a patient becomes symptomatic. If diagnosed early, restoring immune function at this stage can improve outcomes.
A study by Anand and colleagues of 91 patients with PML discovered a median delay of 40 days from symptoms to diagnosis. Diagnostic delays decrease the chances of successfully restoring the immune system, making it critical for healthcare professionals to recognize the signs and symptoms.
Treatment
No current FDA-approved treatment exists for PML, making the withdrawal of the responsible drug the only option to restore the immune system. Neurologists believed that plasmapheresis could quickly remove the responsible drug from the body, but a retrospective study by Landi and colleagues, comparing mortality and outcomes of those treated with plasmapheresis and those who weren’t, found no differences between the two groups. However, many neurologists continue to use this treatment, especially with natalizumab-associated PML.
Cancer’s underlying disease process makes restoring the immune system challenging, and patients with organ transplants must consider the risk of organ rejection when stopping immunosuppressant drugs. Patients with HIV who are diagnosed with PML have improved outcomes when started on combination antiretroviral therapy. Unfortunately, this therapy hasn’t been found beneficial for patients with other diseases who’ve been diagnosed with PML.
Several treatments have been tested to determine their effectiveness in treating PML and its associated symptoms. They include mefloquine, mirtazapine, checkpoint inhibitors, T-cells, corticosteroids, and filgrastim.
Clifford and colleagues tested the antimalaria drug mefloquine for its anti-viral effects in a randomized parallel-group trial of 38 patients. The researchers terminated the trial early when no lack of difference occurred among groups and the patients’ disability continued to progress. A review by Jamilloux and colleagues of 75 case reports, in which mirtazapine (an antidepressant that blocks the glial receptor cells for the JC virus) was used, found only minimal benefit for patients with MS treated with natalizumab and no benefit for other patients.
Checkpoint inhibitors (such as pembrolizumab and nivolumab), which interrupt programmed cell death on T cells and may improve viral clearance, is an emerging therapy for patients in whom boosting the immune system isn’t possible. In 2019, Cortese and others at the National Institutes of Health initially treated eight patients with PML with pembrolizumab every 4 to 6 weeks for one to three doses at 2 mg/kg of body weight. Five of the eight patients showed clinical improvement or stabilization, three showed no improvement or worsening of their symptoms, and two died within 6 months. According to Volk and associates, as of May 2021, 31 patients have been treated with checkpoint inhibitors with mixed results.
In a study by Muftuoglu and colleagues, three patients (who had cancer or HIV and PML) were infused with matched or partially matched T-cells to boost their immune systems. The researchers reported a decrease in the JC viral load for all three, with two patients improving clinically. The third patient died 8 months later. A case report by Grimley and colleagues reported viral and clinical improvement after T-cell therapy for a woman with leukemia and PML.
Some patients develop immune reconstitution inflammatory syndrome (IRIS), which results in worsening neurologic symptoms that are corroborated with MRI changes. IRIS is treated with aggressive corticosteroid therapy to prevent fatal cerebral edema. According to Scarpazza and colleagues, corticosteroids may interfere with JC virus elimination, so they aren’t currently recommended as a treatment for PML unless the patient develops IRIS.
Filgrastim stimulates white blood cell production in patients undergoing chemotherapy. In a study by Stefoski and colleagues, the drug was used in 17 patients with MS diagnosed with natalizumab-associated PML. Although 15 of the 17 developed IRIS after filgrastim initiation, all survived and seven recovered to their baseline neurologic function. Unfortunately, filgrastim may be ineffective in patients with leukemia or lymphoma.
Without a definitive treatment, neurologists may propose any of these experimental options. A lack of suitable animal PML models makes treatment research challenging. In addition, length of time to diagnosis, disease rarity, and lack of research sites complicate patient study enrollment.
Risk mitigation
The natalizumab manufacturer implemented a monitoring program in response to the increased risk of PML for patients with MS and Crohn’s disease. Providers who prescribe the drug must be enrolled in the TOUCH Prescribing Program through the manufacturer and comply with screening MRIs, testing for JC virus antibodies, education, and patient monitoring. Before initiating natalizumab on a patient, a blood test is checked for JC antibodies. Those who do not have the antibodies are at lower risk of developing PML than those who do. MRI screening can detect small brain lesions 3 to 6 months before patients become symptomatic.
Only the natalizumab manufacturer has developed a PML monitoring program, possibly because of lower PML incidence related to other disease-modifying drugs and conditions. However, if all of these drugs had a monitoring program, more patients, nurses, and providers would know the risk factors, signs, and symptoms. Until a PML treatment or screening program is adopted for all the drugs that cause the disease, surveillance for signs and symptoms of PML and leukopenia and patient teaching are the most effective management.
Nursing implications
A study in Australia by Rath and colleagues appears to be the only one that has assessed the PML knowledge of patients with MS when enrolled in a risk-mitigation program for natalizumab therapy. The researchers asked 37 patients three basic questions about PML and found only 43.2% answered all three correctly. They found no difference between the length of treatment and knowledge about PML, raising suspicion that the risk-mitigation program may not be sufficient for teaching patients about signs and symptoms.
According to Skelton and colleagues, patient education for chronic diseases should be individually tailored, culturally competent, and easily understood, as well as provide information on contacting healthcare providers. A literature review by Ciciriello and colleagues verified the effectiveness of using multimedia to teach about medications compared with no education or only verbal instruction. Implementing various types of education (videos, questionnaires with pictures, and patient stories) over several sessions can aid knowledge retention. (See Educational stories.)
Case studies can help patients identify risk factors and symptoms. Paul, the patient in the first case study, was my husband. Progressive multifocal leukoencephalopathy (PML) risk was mentioned briefly at the beginning of treatment and promptly forgotten in the stress of learning he had cancer. No one from two oncology offices spoke of PML risk. Although he had leukopenia, treatment continued. The other case studies are from a Facebook PML group and used with permission. When I asked for volunteers, 13 members contacted me. They or their surviving family members wanted to tell their stories to help healthcare workers and other patients become aware of PML risks and symptoms.
Paul
Paul, a middle-aged man with a history of non-Hodgkin’s lymphoma and leukopenia, under treatment with rituximab every 2 months for 16 months, presented to his primary care physician with complaints of numbness in his left hand. After transfer to a local hospital, a magnetic resonance image (MRI) of his brain showed a sizeable lesion on the right frontal lobe. The leukopenia was treated with pegfilgrastim, a long-acting form of filgrastim. After a few days, Paul’s white blood cell count returned to a very low level. The physicians didn’t understand the connection between leukopenia and Paul’s symptoms.
Paul was discharged home with a diagnosis of stroke. The weakness progressed, causing left arm paralysis over the next several weeks, then left leg weakness. The neurologist recommended he follow up at a large teaching hospital. After a week of bone marrow biopsies, an MRI, and multiple blood tests, Paul was diagnosed with PML. He had been misdiagnosed for 84 days, losing precious time to prepare for his last days. Paul was discharged home with hospice and died 7 weeks later.
Gina
Gina, a 22-year-old woman with MS for 2 years and many exacerbations, was prescribed natalizumab. She gained most of her vision and mobility back with the disease-modifying drug. However, the neurologist neglected to order follow-up MRIs. Five years later, Gina developed extreme weakness on her left side and diminished speech volume. An MRI and lumbar puncture revealed PML. Plasmapheresis was performed to remove the natalizumab from her system, and she was prescribed fingolimod for the MS. Gina also had PML-related seizures and hallucinations, which eventually were controlled with medication. The left-sided weakness progressed to paralysis, which persists today despite no other PML lesions and continued physical therapy. Gina, who is left-handed, had to learn how to function with her right hand. Her spouse continues to help her dress and shower.
Susan
After 2 years of treatment with natalizumab, Susan, a 38-year-old woman with MS, was found to have an abnormal lesion on a routine MRI as a participant in the medication manufacturer’s risk-mitigation program. She was diagnosed with natalizumab-associated PML after a lumbar puncture. Previously, Susan had no PML symptoms. The neurologist ordered plasmapheresis to remove the natalizumab from her system. She developed seizures, memory loss, and weakness. Filgrastim was administered, and she also received mefloquine and mirtazapine. With intensive therapy, Susan gained back full function within 1 year. She credits her recovery to screening, early diagnosis, and aggressive treatment before the appearance of neurologic symptoms.
Because of PML’s poor prognosis, close monitoring and prompt diagnosis are critical. Teaching about the disease and surveillance for signs and symptoms should occur at each follow-up appointment with any patient who’s at risk. The current medication guide provided by the natalizumab manufacturer (bit.ly/3DFSL95), although not appropriate for patients, provides useful information nurses can incorporate into a more engaging presentation, such as the one created by the author. (See PML screening tool.)
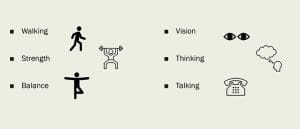
Administering a simple nonvalidated screening tool at each office visit can help nurses identify progressive multifocal leukoencephalopathy symptoms. I created the following tool using Microsoft PowerPoint, which can be adapted to specialties and healthcare organizations.
Do you have MS, RA, Crohn’s, HIV, blood cancer, or an organ transplant? You may be at risk for PML. Let your provider know if you have new or worsening problems with:

For patients diagnosed with PML, nurses should consider suggesting a support group. Facebook has two private social media groups for PML survivors and family members. These nonmedical groups provide support and treatment advice. Each drug manufacturer has a toll-free number for patients to call for information and to report symptoms. In addition, patients can enroll in a clinical monitoring trial through the National Institutes for Health or search for a PML trial at centerwatch.com.
Education for detection
With no PML treatment options other than stopping the medication responsible for causing the condition, nurses must diligently monitor and screen patients at high-risk. Frequent patient and family education can help ensure an early diagnosis before it’s too late.
Helen Condry is a nursing instructor at Charleston Southern University in Charleston, South Carolina.
References
Anand P, Hotan GC, Vogel A, Venna N, Mateen FJ. Progressive multifocal leukoencephalopathy: A 25-year retrospective cohort study. Neurol Neuroimmunol Neuroinflamm. 2019;6(6):e618. doi:10.1212/nxi.0000000000000618
Berger JR. Classifying PML risk with disease modifying therapies. Mult Scler Relat Disord. 2017;12:59-63. doi:10.1016/j.msard.2017.01.006
Ciciriello S, Johnston RV, Osborne RH, et al. Multimedia educational interventions for consumers about prescribed and over-the-counter medications. Cochrane Database Syst Rev. 2013;4:CD008416. doi:10.1002/14651858.cd008416.pub2
Clifford DB, Nath A, Cinque P, et al. A study of mefloquine treatment for progressive multifocal leukoencephalopathy: Results and exploration of predictors of PML outcomes. J NeuroVirol. 2013;19(4):351-8. doi:10.1007/s13365-013-0173-y
Cortese I, Muranski P, Enose-Akahata Y, et al. Pembrolizumab treatment for progressive multifocal leukoencephalopathy. N Engl J Med. 2019;380(17):1597-1605. doi:10.1056/nejmoa1815039
Du Pasquier RA. Pembrolizumab as a treatment for PML? Neurol Neuroimmunol Neuroinflamm. 2019;6(6):e629. doi:10.1212/nxi.0000000000000629
Dunham SR, Schmidt R, Clifford DB. Treatment of progressive multifocal leukoencephalopathy using immune restoration. Neurotherapeutics. 2020;17(3):955-65. doi:10.1007/s13311-020-00848-z
Garry T, Kelly P. MS Research Update. Multiple Sclerosis Association of America. 2020.
Grebenciucova E, Berger JR. Progressive multifocal leukoencephalopathy. Neurol Clin. 2018;36(4):739-50. doi:10.1016/j.ncl.2018.06.002
Grimley MS, Clous G, Mims A, et al. Virus-specific T cells (VSTs) therapy for progressive multifocal leukoencephalopathy (PML)—A novel therapy to combat a fatal disease. Biol Blood Marrow Transplant. 2020;26(3):S339-40. doi:10.1016/j.bbmt.2019.12.371
Jamilloux Y, Kerever S, Ferry T, Broussolle C, Honnorat J, Sève P. Treatment of progressive multifocal leukoencephalopathy with mirtazapine. Clin Drug Investig. 2016;36(10):783-9. doi:10.1007/s40261-016-0433-8
Kapadia RK, Ney D. Stabilization of progressive multifocal leukoencephalopathy after pembrolizumab treatment. Neurohospitalist. 2020;10(3):238-9. doi:10.1177/1941874420902872
Küpper C, Heinrich J, Kamm K, Bücklein V, Rothenfusser S, Straube A. Pembrolizumab for progressive multifocal leukoencephalopathy due to primary immunodeficiency. Neurol Neuroimmunol Neuroinflamm. 2019;6(6):e628. doi:10.1212/nxi.0000000000000628
Landi D, De Rossi N, Zagaglia S, et al. No evidence of beneficial effects of plasmapheresis in natalizumab-associated PML. Neurology. 2017;88(12):1144-52. doi:10.1212/wnl.0000000000003740
Maas RPPWM, Muller-Hansma AHG, Esselink RAJ, et al. Drug-associated progressive multifocal leukoencephalopathy: A clinical, radiological, and cerebrospinal fluid analysis of 326 cases. J Neurol. 2016;263(10):2004-21. doi:10.1007/s00415-016-8217-x
Major EO, Yousry TA, Clifford DB. Pathogenesis of progressive multifocal leukoencephalopathy and risks associated with treatments for multiple sclerosis: A decade of lessons learned. Lancet Neurol. 2018;17(5):467-80. doi:10.1016/s1474-4422(18)30040-1
Muftuoglu M, Olson A, Marin D, et al. Allogeneic BK virus–specific T cells for progressive multifocal leukoencephalopathy. New Engl J Med. 2018;379(15):1443-51. doi:10.1056/nejmoa1801540
National Institute of Neurological Disorders and Stroke. Progressive multifocal leukoencephalopathy information page. July 29, 2019.
Pavlovic D, Patel MA, Patera AC, Peterson I. T cell deficiencies as a common risk factor for drug associated progressive multifocal leukoencephalopathy. Immunobiology. 2018;223(6–7):508-17. doi:10.1016/j.imbio.2018.01.002
Pawlitzki M, Schneider-Hohendorf T, Rolfes L, et al. Ineffective treatment of PML with pembrolizumab. Neurol Neuroimmunol Neuroinflamm. 2019;6(6):e627. doi:10.1212/nxi.0000000000000627
Rath L, Vijiaratnam N, Skibina O. Assessing understanding of individual risk and symptoms of progressive multifocal leukoencephalopathy in patients prescribed natalizumab for multiple sclerosis. Intern Med J. 2017;47(2):194-9. doi:10.1111/imj.13318
Scarpazza C, Prosperini L, De Rossi N, et al. To do or not to do? Plasma exchange and timing of steroid administration in progressive multifocal leukoencephalopathy. Ann Neurol. 2017;82(5):697-705. doi:10.1002/ana.25070
Skelton SL, Waterman AD, Davis LA, Peipert JD, Fish AF. Applying best practices to designing patient education for patients with end-stage renal disease pursuing kidney transplant. Prog Transplant. 2015;25(1):77-84. doi:10.7182/pit2015415
Stefoski D, Balabanov R, Waheed R, Ko M, Koralnik IJ, Morales FS. Treatment of natalizumab‐associated PML with filgrastim. Ann Clin Transl Neurol. 2019;6(5):923-31. doi:10.1002/acn3.776
Volk T, Warnatz K, Marks R, et al. Pembrolizumab for treatment of progressive multifocal leukoencephalopathy in primary immunodeficiency and/or hematologic malignancy: A case series of five patients. J Neurol. 2021;269(2):973-81. doi: 10.1007/s00415-021-10682-8