Through early recognition and prompt treatment, you can help relieve patient pain and discomfort.
Key Takeaways
– Patients with chronic kidney disease, particularly those who have end-stage renal disease are at risk for secondary hyperparathyroidism.
– Management of secondary hyperparathyroidism requires interdisciplinary collaboration.
– Vitamin D analogs, phosphate binders, and calcimimetics are used in the treatment of secondary hyperparathyroidism.
Bob Adams, a 62-year-old African American man, has been receiving outpatient hemodialysis for 6 years. Two years ago, he began experiencing severe bone pain in both arms and legs. At that time, blood tests showed his serum parathryroid hormone (PTH) was 1,200 pg/mL, serum calcium was 9.3 mg/dL, and phosphorus was 6.7 mg/dL.
Current estimates indicate that about 10% of U.S. adults are affected by chronic kidney disease (CKD), which is defined by a glomerular filtration rate (GFR) of less than 60 mL/min for more than 3 months (normal GFR is 90 to 120 mL/min). End-stage renal disease (ESRD), which is categorized as stage 5 CKD, is defined by a GFR of less than 15 mL/min. (See On the rise.)

Complications of CKD and ESRD include cardiovascular diseases, anemia, osteoporosis, depression, and electrolyte imbalances. This article explores secondary hyperparathyroidism (SHPT), also referred to as renal hyperparathyroidism, a less familiar but common long-term consequence of CKD and ESRD.
Pathophysiology of SHPT
Understanding the pathophysiology of SHPT begins with an explanation of the parathyroid glands, two pairs of which are located on the back of each side of the thyroid. Their main function is secretion of PTH to regulate serum calcium levels, increasing when levels are low and decreasing when they’re high. They do this by producing calcium-sensing receptor (CaSR), a G protein that controls PTH levels. (See PTH explained.)
On the rise
| • On December 31, 2014, there were 678,383 prevalent cases of end-stage renal disease (ESRD), representing a 3.5% increase from the 655,435 cases in 2013; 63.1% of all prevalent ESRD patients were receiving hemodialysis therapy, 6.9% were being treated with peritoneal dialysis, and 29.6% had a functioning kidney transplant. • The number of ESRD incident cases plateaued in 2010, and the number of ESRD prevalent cases continues to rise by about 21,000 per year. • Compared to Whites, ESRD prevalence in 2014 was about 3.7 times greater in African Americans, 1.4 times greater in Native Americans, and 1.5 times greater in Asians. • Chronic kidney disease is growing most rapidly in people 65 and older. Source: United States Renal Data System 2016. |
Because patients with CKD convert less vitamin D into its active form, which is needed to absorb calcium in the gut, many experience hypocalcemia. This then stimulates PTH secretion, which promotes bone demineralization. Hyperphosphatemia in patients with CKD occurs when the bone releases phosphate and renal excretion decreases.
Stage 3 CKD is defined by an increase in PTH levels that develops when the GFR drops below 60 mL/min. As the GFR continues to decline, serum phosphorus levels rise and induce hypocalcemia by binding bioavailable calcium. Phosphate retention is thought to be the initiating factor for the development of SHPT. Consequently, overstimulation of the parathyroid glands leads to a further rise in PTH production and hyperplasia of the glands.
PTH explained
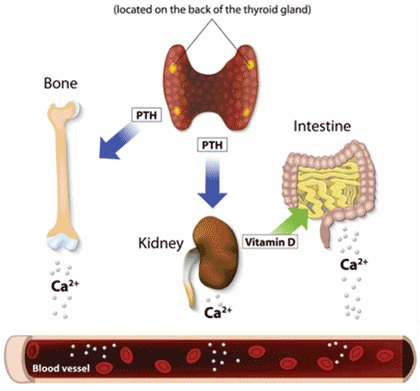
Parathyroid hormone (PTH), considered the most important regulator of calcium metabolism, functions through two primary processes (see image below):
– In the bones, PTH stimulates osteoclast formation and bone resorption (loss of substance) and inhibitsbone formation (removes calcium from bone), which leads to the release of calcium and phosphate into the blood.
– In the kidney, PTH activates 1-α-hydroxylase, which catalyzes the conversion of nonactive 25-hydroxy (25-OH) vitamin D to activated 1,25 dihydroxy (1,25-OH) vitamin D. This leads to increased absorption of calcium and phosphorus in the GI tract. Additionally, PTH increases reabsorption of calcium and decreases reab- sorption of phosphorus in the kidney tubules.
Parathyroid Glands

Patients with SHPT whose glands are hyperplastic have been noted to have a negative regulation (down regulation) by the CaSR. In other words, the parathyroid glands aren’t sensing calcium and are secreting more PTH, which leads to more bone resorption. Tissue hyperplasia leads to an on-and-off activation of the parathyroid glands, and over time, they begin to develop monoclonal nodules. With this underlying pathogenesis, SHPT leads to a host of signs and symptoms and complications resulting from osteodystrophy, cardiovascular changes, and GI abnormalities.
Clinical presentation and complications
Patients with SHPT may present with cardiac arrhythmias, gastric reflux, and bone diseases. High PTH effects on the heart, especially in patients receiving hemodialysis (who also have hyperkalemia), include chest discomfort and cardiac arrhythmias, such as atrial fibrillation and tachycardia. GI problems include reflux and dyspepsia. Some patients also may develop extra-skeletal calcification, calciphylaxis (vascular calcification and thrombosis), debilitating bone disease, and refractory pruritus.
The most prominent symptom is bone pain. (See Bone disorders.) Some patients with PTH above 1,000 pg/mL (normal is 10 to 65 pg/mL [or ng/L]), have difficulty walking. Research has shown that high levels of PTH interfere with the free flow of blood in the bone.
The combination of these manifestations may result in hospitalization with poor outcome because of, for example, serious injuries from falls or life-threatening arrhythmias. High PTH level is an independent risk factor for all-cause and cardiovascular mortality.
Diagnosis and management
Current guidelines recommend screening patients with stage 3 CKD (GFR < 60 mL/min) for SHPT. This includes monitoring serum calcium, phosphorous, and PTH levels. The treatment of SHPT consists of a low-phosphorus diet, phosphate binders, vitamin D derivatives, calcimimetics, and, in some cases, parathyroidectomy.
Oral or I.V. vitamin D is the treatment of choice for patients whose PTH is above 150 pg/mL. In cases where the patient’s phosphorus is high (above 6 mg/dL) despite nutritional modification (for example, low-phosphorus diet), a calcimimetic, such as cinacalcet, is recommended. (See Commonly prescribed medications.)
The latest guidelines from the Kidney Disease Outcomes Quality Initiative recommends keeping PTH between 150 and 300 pg/mL to avoid complete suppression of the osteoclasts and prevent adynamic bone disease. When pharmacologic treatment fails, parathyroidectomy may be considered as the last treatment of choice.
Bone Disorders
| Renal osteodystrophy refers to a group of bone disorders caused by dysregulation of mineral metabolism in patients with chronic kidney disease, including osteomalacia, adynamic bone disease, and osteitis fibrosa cystica. The natural balance between bone formation and resorption is altered, resulting in extremely high or low bone turnover and is typically associated with bone and joint pain, bone deformation, and fractures. Osteitis fibrosa cystica is a high turnover bone disease that stems from elevated parathyroid hormone, which stimulates osteoclast activity, leading to resorption of calcified bone, replaced by fibrous tissue. |
Key considerations for nurses include fall risk, fracture, pain, and immobility. Instruct the patient and family to take steps to reduce or eliminate fall risks in the home, including removing throw rugs and installing handrails in the shower. In addition, talk to the patient about maintaining his or her health by balancing rest and exercise, adhering to dietary modifications, and following the prescribed medication regimen. Nurses are pivotal to boosting a patient’s resiliency through motivational interviewing and therapeutic communication.
Early recognition, prompt treatment
With Mr. Adams’ input, his interdisciplinary team—which included a nephrologist, his primary care provider, a nephrology nurse practitioner, dialysis nurses, a dietician, a social worker, and a dialysis technician—decided to address his worsening SHPT. Mr. Adams’ main concern was his chronic bone pain. His provider started him on 30 mg of cinacalcet every day; vitamin D was not initiated because of his high phosphorus level. Within 2 weeks of taking cinacalcet, Mr. Adams’ PTH dropped to 1,034 pg/mL. Because he didn’t report any adverse reactions and experienced almost no side effects, the dose was increased to 60 mg per day with meals. A month later, Mr. Adams’ PTH level decreased to 756 pg/mL, and within 3 months it was down to 300 pg/mL. Mr. Adams continued to take his cinacalcet, which was decreased to 30 mg per day. Currently, his PTH ranges between 145 and 200 pg/mL, and he’s pain free except for intermittent leg discomfort.
SHPT can be a debilitating complication of CKD and ESRD. By being alert to the possibility of SHPT, nurses can promote early recognition and prompt treatment to alleviate the patient’s pain and discomfort.
Commonly prescribed medications
| Drug | Mechanism of action | Usual adult dose | Common side effects | Nursing considerations | ||||
| Vitamin D analogs (e.g., calcitriol, doxercalciferol, ergocalciferol) | Suppresses high PTH levels and enhances intestinal absorption of calcium | Calcitriol: Doxercalciferol: Ergocalciferol: 50,000 units PO once per week | Constipation Dizziness Edema Headache Hypercalcemia Hyperphosphatemia Nausea/vomiting Somnolence | Ergocalciferol is used only in stages 3 and 4 CKD if the 25-hydroxyvitamin D level is <30 pg/mL | ||||
| Phosphate binders (e.g., calcium acetate, sevelamer HCL, or sevelamer carbonate) | Binds to dietary phosphorus through ion exchange | Calcium acetate: 3-6 g/day Sevelamer HCL or sevelamer carbonate: 4.8-9.6 g/day | Abdominal pain Constipation Diarrhea Dyspepsia Flatulence Hypercalcemia Hypomagnesemia Metabolic acidosis Nausea/vomiting | Take with meals | ||||
| Calcimimetics (e.g., cinacalcet) | Increases sensitivity of calcium-sensing receptors in the parathyroid glands, suppressing PTH secretion Indicated only for hemodialysis patients with PTH levels | Cinacalcet: 30-180 mg/day | Cough Diarrhea GI symptoms Hyperkalemia Hypocalcemia Nausea/vomiting Prolonged QT interval | Hold if calcium level is below 8.4 mg/dL May be used in combination with vitamin D | ||||
| ||||||||
PTH = parathyroid hormone, CKD = chronic kidney disease
Fidelindo Lim is a clinical assistant professor at New York University, Rory Meyers College of Nursing in New York City. Vivian Gasu works at Mount Sinai Beth Israel in the Nephrology & Hypertension Division, Irving Place Dialysis Unit in New York City.
Selected references
Carter T, Ratnam S. Calciphylaxis: A devastating complication of derangements of calcium-phosphorus metabolism—A case report and review of the literature. Nephrol Nurs J. 2013;40(5):431-5.
Centers for Disease Control and Prevention. National chronic kidney disease fact sheet, 2014.
International Society of Nephrology. KDIGO 2012 clinical practice guideline for the evaluation and management of chronic kidney disease. 2013.
Lewis SL, Dirksen SR, Heitkemper MM, Bucher L. Medical-Surgical Nursing: Assessment and Management of Clinical Problems. 19th ed. St. Louis, MO: Elsevier; 2017
National Institutes of Health. National Institute of Diabetes and Digestive and Kidney Diseases. Kidney disease statistics for the United States. 2016.
National Kidney Foundation. Glomerular filtration rate (GFR). 2015.
Nickolas TL, Jamal SA. Bone kidney interactions. Rev Endocr Metab Disord. 2015;16(2):157-63.
Nordholm A, Rix M, Olgaard K, Lewin E. Parathyroid hormone-related peptide plasma concentrations in patients on hemodialysis. Scand J Clin Lab Invest. 2014;74(3):206-12.
Ritter CS, Slatopolsky E. Phosphate toxicity in CKD: The killer among us. Clin J Am Soc Nephrol.2016;11(6):1088-100.
Sumida K, Nakamura M, Ubara Y, et al. Cinacalcet upregulates calcium-sensing receptors of parathyroid glands in hemodialysis patients. Am J Nephrol. 2013;37(5):405-12.
United States Renal Data System. Chapter 1: Incidence, prevalence, patient characteristics, and treatment modalities. 2016.


















