Do you know the magic number?
Takeaways:
- Follow your organization’s policies and procedures for interpreting TB skin test results and stay up-to-date on the most recent guidelines and recommendations.
- Accurate interpretation depends on various risk factors; no single measurement is always indicative of TB.
- Measure the induration only, not the redness of the tuberculin skin test reaction.
As required for volunteer work at a local hospital, Monica, a 28-year-old woman, arrives as a new patient at a small community clinic to request a tuberculosis (TB) skin test. When the nurse asks Monica if she’s ever had a positive TB test, she says that she hasn’t. The nurse administers the purified protein derivative (PPD) vaccine to the patient’s left forearm and advises her to return in 48 to 72 hours to read the results. When the patient returns 2 days later, the nurse notes a 7-mm induration and no redness. He documents the results as negative.
About 3 months later Monica returns to the clinic with a cough and night sweats, which she says have worsened over the past 2 months. The provider obtains a complete history. The patient has hypertension, diabetes, and is HIV positive. A chest X-ray ordered by the provider indicates that Monica may have TB. A sputum culture confirms the diagnosis. Everyone Monica has encountered (at the hospital where she volunteers, at the clinic, and in her home and community) must be tested and receive ongoing follow-up and care.
Re-imagining nursing’s social contract with the public
A guide for adhesive removal: Principles, practice, and products
Color awareness: A must for patient assessment
Could this situation have been avoided when Monica received her TB skin test? Did the nurse make an error when he interpreted the 7-mm induration as negative? Does a magic number exist that nurses can rely on as the minimum induration for a positive result?
Nurses’ role in TB testing
All nurses learn how to administer and interpret TB skin tests and that a result of ≥10 mm induration is positive. The test can be given as a one- or two-step screening. In the one-step protocol, the nurse administers the PPD vaccine intradermally to the patients’ forearm and interprets the result within 48 to 72 hours. If the results aren’t read within that timeframe, the result becomes invalid and the test must be repeated. In the two-step protocol, a negative result after 48 to 72 hours requires a repeat test within 1 to 4 weeks. This protocol reduces the likelihood of a boosted reaction to subsequent TB skin tests in patients with no previous history of TB. Nurses must be accurate when interpreting TB skin test results to identify those patients who may need further diagnostic testing and follow up. Inaccurate readings may increase the number of TB cases worldwide. (See About TB.)
About TB

According to the World Health Organization, tuberculosis (TB) is the 13th leading cause of death worldwide and the second leading infectious killer after COVID-19.
- Worldwide, in 2020, an estimated 10 million cases of TB were diagnosed and 1.5 million people died from the disease.
- The incidence of TB in the United States decreased from 9,105 in 2017 to 7,860 in 2021.
- In 2020, 43 states reported incidences at or below the national average of 2.2 cases per 100,000 persons, six states reported incidences that exceeded the national average, and only one state reported no TB cases.
- The 2020 data reveal a significant decrease in the number of reported TB cases compared to previous years. This decline is believed to be impacted by COVID-19 in many ways, including underdiagnosis and a true reduction in the disease.
Nurse knowledge
Although we can’t prevent some TB infections, many cases occur because patients go undiagnosed as a result of false-negative skin tests and then unknowingly spread TB to others. False-negative tests can result from improper PPD vaccine storage, improper administration, and improper interpretation of results due to lack of knowledge or inexperience. (See False-negatives.)
False-negatives
Many sources of false-negative tuberculosis (TB) skin tests are correctable. Nurse knowledge of the following causes can help prevent care delays and ensure adequate treatment:
- Delayed administration after drawing up purified protein derivative (PPD)
- Improper PPD vaccine storage (prolonged exposure to light or heat)
- Incorrect administration (not administered intradermally)
- Measurement error
- Misinterpretation of TB skin test reaction
- Recent live virus administration (within 4 to 6 weeks of receiving TB vaccine)
- Vaccine contamination
Source: Centers for Disease Control and Prevention 2020
As a family practice RN, I’m asked by many licensed vocational nurses (LVNs) to verify their TB test result measurements and interpretations. The frequency of these requests prompted me to follow up on their skin test knowledge and their familiarity with the organization’s interpretation policy. The family medicine department conducted a brief survey with eight family medicine LVNs, eight pediatric LVNs, and four RNs. We asked: “When reading a TB skin test, what would you interpret and document as a positive result?” and “Are you aware of the organizational policy for interpreting TB skin test results?”
Half of the family medicine LVNs responded incorrectly to the first question. Answers included “any millimeters of induration” and “0.5 mm induration.” The other half gave partially correct answers, with responses ranging from 9 mm to 10 mm induration with and without erythema. Of the eight pediatric LVNs, 25% responded that they were unsure, 50% responded inaccurately (results ranged from 5 mm to 20 mm induration), and 25% answered correctly. Of the four RNs surveyed, only half responded correctly. None of the participants were aware of the organization’s policy for interpreting TB skin test results, which had been updated recently.
Interpreting results
Do you know the magic number for interpreting a TB skin test as positive? If not, don’t worry. No such thing actually exits. Instead, nurses should consider three different numbers based on a patient’s medical history and TB risk factors. For example, a patient with a history of HIV (as in the case with Monica) should be considered to have a positive TB skin test with a measurement of ≥5 mm induration. Patients in hospitals or nursing homes, children under 4 years of age, and patients who use injection drugs with ≥10 mm induration should be considered positive. Anyone 4 years and older with a low risk for TB infection is considered positive with ≥15 mm induration. (See Accurate interpretation.)
Accurate interpretation
Accurate interpretation of tuberculosis (TB) screening (interferon-gamma release or skin test) requires knowledge of the patient’s medical history and risk factors. Depending on this information, different induration measurements may indicate a positive result.
≥5mm
- Patients with HIV
- Those who’ve had recent contact with someone who has TB
- Patients with fibrotic changes on a chest X-ray consistent with old TB
- Organ transplant recipients
- Patients who are immunosuppressed for reasons other than organ transplant (for example, those taking the equivalent of >15 mg/day of prednisone for 1 month or longer, those taking TNF-α antagonists)
≥10 mm
- Recent immigrants (<5 years) from countries with a high prevalence of TB
- Patients who use injection drugs
- Residents and employees of high-risk group housing settings (correctional facilities, nursing homes, homeless shelters, hospitals, and other healthcare facilities)
- Mycobacteriology laboratory personnel
- Children under 5 years of age and children and adolescents exposed to adults in high-risk categories
- People with a low body weight (<90% of ideal body weight).
≥15 mm
- Individuals with no known TB risk factors
Source: Centers for Disease Control and Prevention 2020
Screening options
In addition to administering TB skin tests, you can screen patients for TB with an interferon gamma-release assay (IGRA). Perform this screening independent of a skin test. Because of limited data, IGRA isn’t recommended for children under 5, patients recently exposed to TB, and patients who are immunocompromised.
Advantages of IGRA include completed testing in one patient visit, results within 24 hours, no boosted response in subsequent testing, and no altered results in patients who’ve previously received the bacille Calmette-Guerin (BCG) vaccine. The BCG vaccine can cause false-positive TB skin test results, but its reactivity typically decreases over time. If a TB skin test is considered positive 5 years or more after the patient received the BCG vaccine, results likely are due to TB infection and require further diagnostic evaluation.
Screening education and recommendations
Education and competency validation can help ensure you stay up-to-date with new TB skin test interpretation. Results of my short survey indicated the need for staff education. After discussions with the leadership team, education and training are now provided to current and incoming staff to increase accuracy of TB skin test interpretations and decrease the risks of false-negatives.
Safe patient care requires that all nurses be aware of their organization’s policies and procedures by reviewing them periodically and staying current with new guidelines. For example, the 2005 Centers for Disease Control and Prevention (CDC) guidelines recommended that all U.S. healthcare personnel working in medium-risk settings with the potential for ongoing TB transmission receive baseline screening and annual testing. The most recent updates from 2019 include the following:
- Baseline TB screening with symptom evaluation and an individual risk assessment.
- TB skin test or IGRA screening for anyone without a documented history of TB.
- In the absence of ongoing transmission or a known exposure, no requirement for routine serial testing at any interval after baseline.
- If not contraindicated, encourage treatment for all healthcare workers with latent TB.
- Annual symptom screening for untreated healthcare personnel with latent TB.
- Annual TB education for all healthcare workers.
Keys to accurate interpretation
When interpreting TB skin test results, follow your organization’s policies and procedures, which should contain the most recent CDC guidelines. Consider the patient’s medical history and TB risk factors to determine whether they’ll be positive at 5 mm, 10 mm, or 15 mm induration.
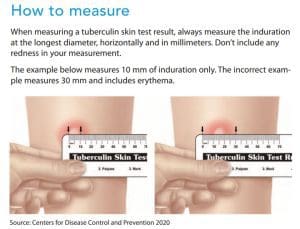
To ensure accuracy, perform interpretations in adequate lighting and mark the areas of induration. Then measure its longest diameter horizontally. Don’t measure any redness, which may indicate an allergic or PPD vaccine reaction and doesn’t have interpretive value. Always document results in millimeters, and repeat the test if you suspect a false-positive or false-negative. (See How to measure.)

If you interpret the test as positive for TB, refer the patient for a chest X-ray and further diagnostic testing based on your organization’s policies and procedures. Don’t be afraid to ask for help, verification, or clarification. Remember, you’re the driver of your education and knowledge—stay updated.
Shanna Jackson is a nurse practitioner at Kaiser Permanente Medical Center in Riverside, California.
References:
Centers for Disease Control and Prevention. Interferon gamma-release assays (IGRAs) – Blood tests for TB infection. May 4, 2016.
Centers for Disease Control and Prevention. Resources for TB Programs. May 11, 2016.
Centers for Disease Control and Prevention. Targeted tuberculosis testing and interpreting tuberculin skin test results. August 31, 2016.
Centers for Disease Control and Prevention. TB incidence in the United States, 1953-2020. October 5, 2021.
Centers for Disease Control and Prevention. Trends in tuberculosis, 2020. October 12, 2021.
Centers for Disease Control and Prevention. Tuberculin skin testing. November 20, 2020.
Centers for Disease Control and Prevention. Tuberculosis: Data and Statistics. March 24, 2022.
Sosa LE, Njie GJ, Lobato MN, et al. Tuberculosis screening, testing, and treatment of U.S. health care personnel: Recommendations from the National Tuberculosis Controllers Association and CDC, 2019. MMWR Morb Mortal Wkly Rep. 2019;68(19):439-43. doi:10.15585/mmwr.mm6819a3
World Health Organization. Tuberculosis. October 14, 2021.


















2 Comments. Leave new
Wow, very insightful write up on Tuberculin skin test interpretations. Thank you.
Insightful, thank you