Management of ischemic and hemorrhagic stroke
- Acute ischemic and hemorrhagic stroke require timely recognition, evaluation, and intervention to optimize patient outcomes.
- Priority care for acute ischemic stroke includes expedient evaluation and delivery of I.V. thrombolysis and/or mechanical thrombectomy, if eligible.
- Immediate priorities for hemorrhagic stroke include anticoagulation reversal and blood pressure and intracranial pressure control.
According to the American Heart Association (AHA), more than 795,000 Americans experience a stroke each year, and first-time strokes account for over three-quarters of them. An acute stroke, whether ischemic or hemorrhagic, is a clinical emergency that requires urgent intervention. Immediate priorities include timely recognition of stroke symptoms and expedient activation of systems of care. Bedside nurses play a primary role in activating these systems and providing coordinated stroke care throughout hospitalization.
Learning Objectives
- Describe key aspects of identifying stroke symptoms and activating clinical emergency systems.
- Discuss acute ischemic stroke nursing interventions and treatment priorities.
- Describe hemorrhagic stroke nursing interventions and treatment priorities (for both intracerebral hemorrhage and subarachnoid hemorrhage).
The author, planners, and peer reviewers of this CNE activity have disclosed no relevant financial relationships with any commercial companies pertaining to this activity, except for the following: Bethany Young, author for this nursing continuing professional education development activity, has received financial compensation from Medtronic as a speaker. All relevant financial relationships for this individual have been mitigated. See the last page of the article to learn how to earn CNE credit.
Expiration: 1/1/26
Acute ischemic stroke
The AHA reports that ischemic stroke accounts for 87% of all strokes. Causes include atherosclerosis (the most common) and thrombotic or embolic events. Common acute symptoms include facial drooping, speech difficulty, unilateral weakness or numbness, vision changes, difficulty ambulating, vertigo, and sudden decreased level of consciousness.
Initial actions
Restoring perfusion and optimizing neurologic outcomes require timely recognition of ischemic stroke symptoms and emergency response activation. Approximately 1.9 million neurons are lost during each minute of untreated large vessel stroke, highlighting the notion that “time is brain,” and every second counts during an acute ischemic stroke (AIS). In addition, time-limited AIS interventions (such as I.V. thrombolysis and endovascular thrombectomy) depend on knowing when the patient was last well. For these reasons, treat suspected stroke as a medical emergency that requires immediate activation of hospital-based emergency systems, whether in the emergency department or inpatient units. To facilitate expedient evaluation and care, ensure you’re familiar with your organization’s emergency stroke response protocols.
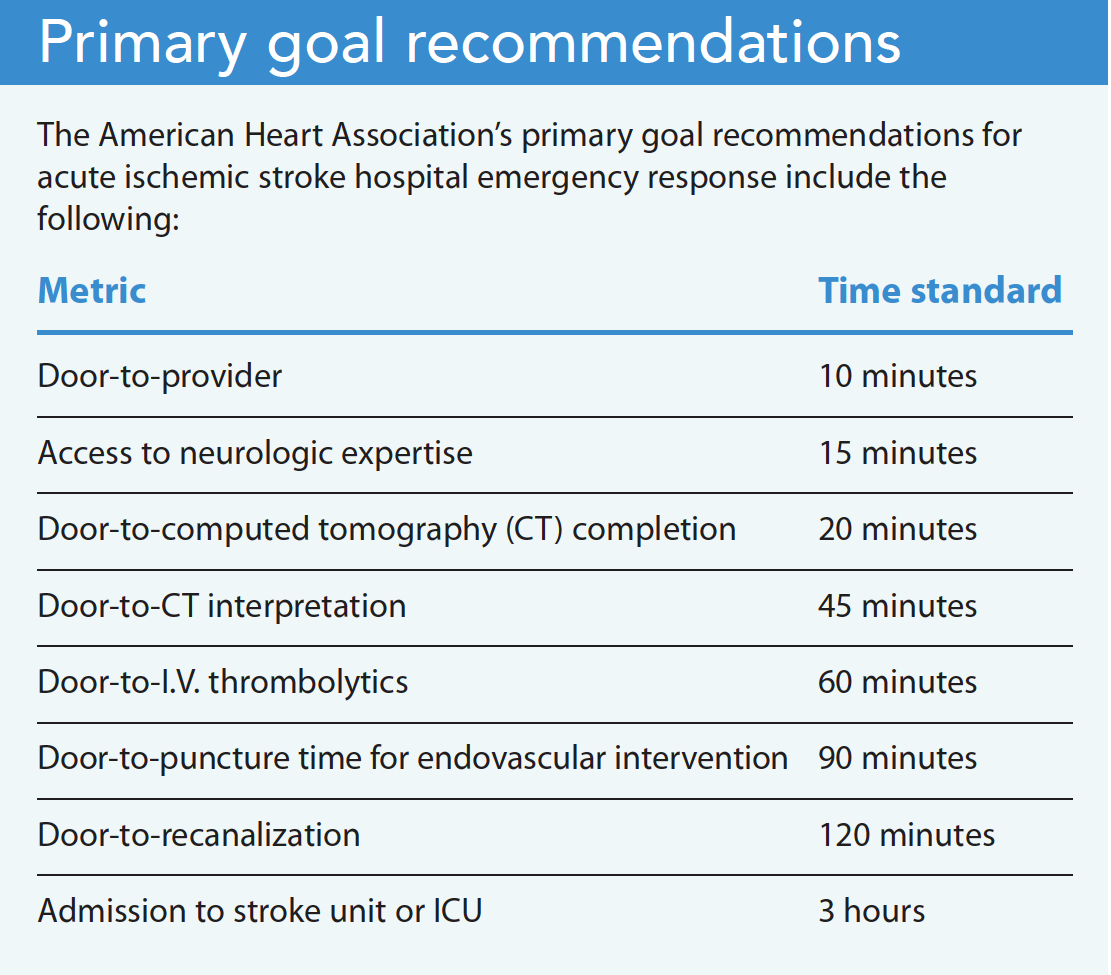
The AHA recommends clear targets to drive timely stroke care: neurologic evaluation, non-contrast head computed tomography (CT), and evaluation of eligibility for I.V. thrombolytic therapy. A standardized neurologic assessment tool, such as the National Institutes of Health Stroke Scale, can help quantify stroke symptom severity with reliable reproducibility. At the same time, continually assess the patient’s airway and cardiopulmonary stability. Nurses can provide supplemental oxygen to maintain SpO2 >94% and advocate for additional stabilizing support if needed.
If airway and hemodynamic status are stable, early priorities for suspected AIS focus on timely neuroimaging and determining treatment eligibility. To facilitate these priorities, immediate nursing actions include obtaining vital signs, capillary glucose level, and an accurate weight. Establish reliable I.V. access to obtain basic labs, accommodate I.V. contrast for a CT angiography, and for potential thrombolytic administration. The provider may order additional laboratory studies, such as complete blood count and coagulation studies, but these shouldn’t delay obtaining a CT. To determine the patient’s thrombolytic eligibility, note their history (including malignancy, recent surgery, or trauma), the timing of symptom onset, and any anticoagulating medications. (See Primary goal recommendations.)
Treatment priorities
Treatment priorities for AIS include I.V. thrombolysis and endovascular mechanical thrombectomy (MT).
I.V. thrombolysis.
Because ischemic and hemorrhagic stroke symptoms may be indistinguishable, a CT scan must be performed to rule out the presence of a hemorrhage before administering thrombolytics. However, because ischemic stroke is more prevalent than hemorrhagic stroke, the healthcare team should prepare to treat an ischemic stroke until proven otherwise. A CT angiogram also can be used to identify the presence of a large vessel occlusion, which might be a target for endovascular intervention. After neuroimaging, subsequent priorities include deciding whether to administer I.V. thrombolysis and assessing eligibility for endovascular thrombectomy.
The Food and Drug Administration has approved I.V. thrombolysis with alteplase (tPA) for up to 3 hours after AIS onset or when the patient was last known to be well; the AHA endorses it for up to 4.5 hours after onset in eligible patients (those with no hemorrhage on CT, coagulopathy, recent anticoagulation, or recent surgery). This practice aligns with many U.S.-based organizational practices. In the absence of absolute contraindications for thrombolysis, you can prepare the patient for thrombolytic administration by achieving blood pressure control below 185/110 mmHg. To encourage adequate brain perfusion and limit the risk of intracranial hemorrhage while also avoiding hypotension, maintain blood pressure below 180/105 mmHg for 24 hours after thrombolytic administration.
I.V. thrombolytic complications include major intracranial or systemic bleeding, angioedema, oozing from gums and venipuncture sites, hematuria, and hemoptysis. To help detect these complications, conduct stringent vital sign checks and neurologic assessments within the first 24 hours after thrombolytic administration (every 15 minutes for the first 2 hours, every 30 minutes for the next 6 hours, then hourly for 16 hours). If major bleeding occurs during tPA administration, stop the infusion, notify the provider, and prepare to obtain an emergent CT. Maintain supportive care (cardiovascular support, airway management, intracranial pressure [ICP] management) and prepare to administer cryoprecipitate and other antifibrinolytics or adjunctive agents, such as thawed or fresh frozen plasma and prothrombin complex concentrate.
Endovascular MT.
Endovascular MT, with or without I.V. thrombolysis, is an additional standard of care for AIS resulting from large vessel occlusion. Patients with a favorable ratio of salvageable brain tissue may qualify for MT up to 24 hours after the onset of stroke symptoms vs. the 3 to 4.5 hour window for I.V. thrombolysis. This is determined through radiographic assessment of early infarction volume and volume of at-risk but salvageable tissue. Hospitals capable of caring for patients with stroke can’t necessarily provide all types of interventions. If a hospital can’t perform MTs, patients may require transport to another center. Coordination within stroke systems of care and with local emergency response services helps ensure patients have access to the appropriate treatment center as quickly as possible.
In addition to careful neurologic assessment after an MT, care requires frequent vital sign checks, puncture site care, and neurovascular assessment. Complications include vessel dissection; extension of infarcted tissue, leading to cerebral edema and increased ICP, if the vessel isn’t recanalized; intracranial hemorrhage; and access site complications (retroperitoneal bleed, pseudoaneurysm, distal ischemia). Frequently assess for and immediately report pain, swelling, bruising, erythema, bleeding, hematoma, pulsatile mass, or drainage at the puncture site to ensure complication detection and intervention.
Additional nursing care
Patients with AIS have complex, multisystem needs that inform nursing priorities throughout hospitalization. Ongoing neurologic assessment and early recognition of new or recurrent deterioration can help minimize further complications. In the first 3 to 5 days after stroke, monitor for neurologic changes indicative of cerebral edema, particularly in patients with a large hemispheric stroke for whom early recanalization wasn’t successful. The provider should consider immediate brain imaging in response to any acute neurologic change. Cerebral edema should be medically managed and may require neurosurgical intervention.
Up to 65% of patients experience some degree of swallowing dysfunction (dysphagia) after stroke. Dysphagia contributes significantly to post-stroke morbidity and mortality resulting from aspiration, pneumonia, and malnutrition. Consider performing bedside dysphagia screening (and in some cases formal evaluation) before any oral intake (including medications). If the patient experiences difficulty swallowing during screening, a speech-language pathologist should perform a complete swallowing assessment within 24 to 48 hours of hospital admission, and the healthcare team should make a decision about feeding within 72 hours of admission.
Blood pressure management is crucial in stroke patients. Excessive hypertension in patients with AIS can lead to intracerebral hemorrhage (ICH) and end-organ damage. Conversely, hypotension can lead to a lack of collateral perfusion and infarct extension. To keep blood pressure below 180/105 mmHg, administer short-acting antihypertensive agents (such as labetalol, nicardipine, and clevidipine) as ordered. Continue cardiac monitoring for at least 24 hours post-stroke to observe for any dysrhythmias.
Additional supportive elements of post-stroke care include reducing fever burden and maintaining glycemic control. Perform an infection workup and administer treatment, as ordered. Administer antipyretic medications and apply cooling measures and devices for fever management. Hyperglycemia in the first 24 hours post-AIS is associated with poor outcomes, so preserve blood glucose between 140 and 180 mg/dL.
Patients who’ve experienced a stroke may be at increased risk for immobility, skin breakdown, and bowel or bladder dysfunction. They’re also at increased risk for venous thromboembolism (VTE) for about 4 months after an acute stroke, so they’ll require pharmacologic and mechanical VTE prophylaxis and early mobilization. Adjunctive post-discharge treatments may include lipid-lowering agents, glucose management, antidepressants, and antiepileptics.
AIS, a life-altering diagnosis, requires early and frequent communication with patients and families about causes and risk factors, health maintenance, medications, and lifestyle adaptation. Nearly a quarter of patients with AIS will experience a subsequent stroke, making early targeted discussions about ongoing individual risk factors and stroke prevention measures crucial.
Hemorrhagic stroke
Hemorrhagic stroke (intracerebral hemorrhage [ICH] and subarachnoid hemorrhage [SAH]) accounts for 13% of stroke cases. ICH occurs when a cerebral blood vessel bursts, causing blood to leak into surrounding brain tissue. This accounts for about 10% of all hemorrhagic strokes. SAH results from blood leaking into the subarachnoid space (the space between the two outer layers of the brain that houses cerebrospinal fluid [CSF]). Rupture of an intracranial aneurysm or other cerebrovascular abnormalities can cause an SAH. Although hemorrhagic stroke is far less common than ischemic stroke, both ICH and SAH carry high morbidity and mortality risk and represent serious medical emergencies.
ICH
ICH can result from a number of causes, including chronic hypertension, cerebral amyloid angiopathy, coagulopathies, vascular malformations, or sympathomimetic drug use. ICH also may result from bleeding into brain tissue after ischemic stroke, a complication known as hemorrhagic transformation, Bleeding into brain tissue compresses surrounding structures, and rapid hematoma formation increases ICP. Definitive ICH diagnosis requires neuroimaging because of AIS symptom similarity. However, severe headache, quickly progressing neurologic findings, acute and severe hypertension, and decreased level of consciousness are more common in ICH than AIS.
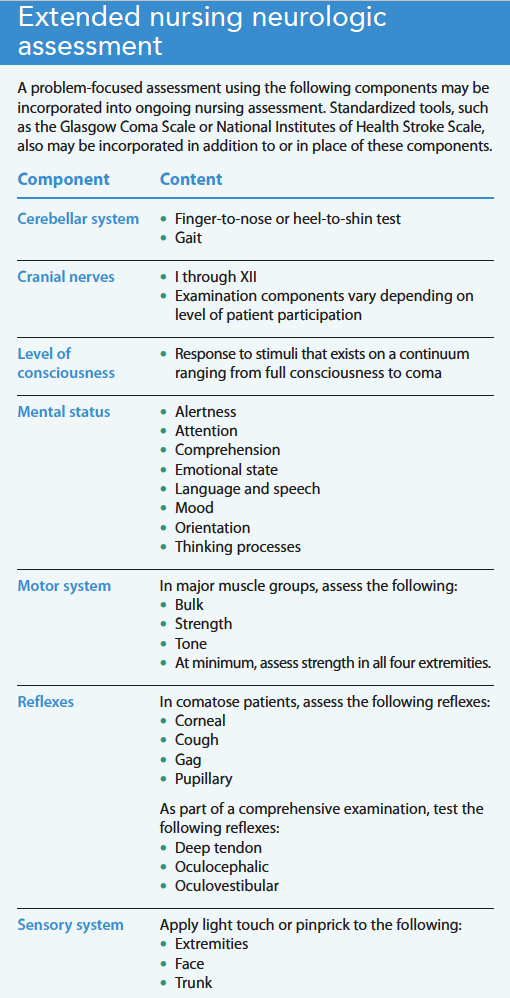

Initial actions. Increased risk for rapid and progressive decreased consciousness requires immediate airway and circulatory stabilization. Rapid sequence intubation should occur without delay if needed to establish normal ventilation. Simultaneously, obtain an initial neurologic assessment, including, at a minimum, pupillary reactivity and Glasgow Coma Scale score. The next immediate action is a non-contrast head CT for timely diagnosis. (See Extended nursing neurologic assessment.)
Treatment priorities. After ICH diagnosis confirmation, the following treatment priorities occur quickly and in tandem: blood pressure management, coagulopathy correction, neurosurgical consultation (including for external ventricular drainage [EVD] placement for ICP management and monitoring).
Many patients with ICH experience hypertension. Convention dictates lowering blood pressure to prevent re-bleeding and hematoma expansion, but precise blood pressure management strategies remain controversial. Two randomized trials have demonstrated safety of acutely lowering systolic blood pressure to less than 140 mmHg after ICH, although exact targets that optimize overall patient outcomes remain unclear. Therefore, AHA Guidelines recommend a somewhat tailored approach to blood pressure lowering based on the patient’s presenting blood pressure, comorbidities, and hypertension chronicity, with the guiding principle that blood pressure shouldn’t remain extremely elevated. Antihypertensive therapy should be initiated quickly, ideally with the use of titratable I.V. infusions.
Coagulopathy correction requires immediate serum coagulation studies. Coagulopathies occur as a result of underlying medical conditions or secondary to antithrombotic medication regimens. Because not all anticoagulants result in abnormal partial thromboplastin time or international normalized ratio, the healthcare team must obtain a reliable medical history with a home medication list. Familiarize yourself with your organization’s anticoagulation reversal protocols; administer appropriate reversal agents as ordered and as quickly as possible. Many antithrombotic medications, with various mechanisms of action, exist. Determining the appropriate reversal agent depends on the prescribed anticoagulant and organization formulary. Readily available anticoagulation reversal protocols can aid timely therapeutic goals.
Surgical options for most patients with ICH remain limited in their ability to improve long-term mortality or functional outcome. However, it’s still reasonable to obtain early neurosurgical consultation for these patients. Minimally invasive surgical hematoma evacuation may offer promise for reducing mortality after supratentorial ICH. Given the physical space constraints of the posterior fossa, the AHA and the American Stroke Association recommend early surgical clot evacuation for patients with cerebellar hemorrhage and neurologic deterioration, brainstem compression, or ventricular obstruction. Overall, surgical treatment for spontaneous ICH remains limited and individualized according to hematoma size, age, timing, baseline clinical severity, and other clinical factors. EVD placement for patients with acute hydrocephalus also may require neurosurgical services.
Additional nursing care.
Patients with ICH frequently experience neurologic and medical instability. Ideally, they’re monitored in an ICU staffed by physicians and nurses with neuroscience expertise. Detection of neurologic deterioration and other adverse events, such as seizure or ICP elevations, requires ongoing diligent neurologic assessment. Clinical seizures require appropriate treatment; electroencephalography can help rule out subclinical seizures in patients who exhibit unexplained coma. Glycemic control, fever avoidance, and VTE prevention should continue throughout hospitalization.
Nurses provide a constant and consistent bedside presence and therefore play a critical role in patient and family education about treatment modalities and secondary ICH prevention measures. Patients with ICH also may require transport between hospitals and experience various transitions in levels of care throughout hospitalization. Apply your nursing communication skills during these transitions to help ensure safe transport and facilitate well-coordinated, evidence-based care.
SAH
Nontraumatic SAH accounts for approximately 3% of all strokes, most of which result from ruptured intracranial aneurysms. Major risk factors for aneurysmal SAH (aSAH) include hypertension, cigarette smoking, binge alcohol drinking, and drug and stimulant use. This type of stroke frequently occurs in individuals between the ages of 35 and 60. aSAH is often heralded by a sudden, severe headache, frequently characterized as the “worst headache of my life,” and sometimes accompanied by vomiting, neck stiffness or pain, or loss of consciousness. Although aSAH most frequently presents as a sudden “thunderclap” headache, 10% to 40% of patients experience a more mild, preceding headache several weeks before a sentinel headache. Many patients experience altered consciousness or focal neurologic signs after aSAH, but some don’t, which can lead to misdiagnosis. Atypical headache characterization, therapeutic response to analgesics, or normal mental status don’t necessarily rule out aSAH.
A thorough history and clinical examination can aid aSAH diagnosis. A non-contrast head CT may reveal blood in the subarachnoid space; however, the CT may appear normal if the hemorrhage is too small to detect or if enough time has lapsed since the hemorrhage occurred and normal CSF circulation has cleared the blood. Patients with suspected aSAH but negative or equivocal CT scans should undergo lumbar puncture to assess for the presence of red blood cells in the CSF. Additional angiography after aSAH diagnosis can confirm the source of aneurysmal bleeding and assist surgical planning.
Initial actions. Top priorities in the initial care of a patient with aSAH include airway and hemodynamic assessment and stabilization. Immediately connect the patient to cardiac telemetry monitoring and obtain a 12-lead electrocardiogram (ECG). Send a full laboratory panel, as ordered, including coagulation studies and cardiac enzymes. Cardiopulmonary injury exhibited by elevated troponin, ECG changes, cardiac arrhythmias, and pulmonary edema are common after aSAH. Catecholamine excess may result in cardiomyopathy and left ventricular failure; acute brain injury may cause neurogenic pulmonary edema. These factors, in combination with compromised neurologic status, may necessitate endotracheal intubation with mechanical ventilation and additional pharmacologic hemodynamic support. Optimal patient outcomes rely on conscientious nursing assessment and cardiopulmonary stability maintenance until definitive treatment initiation.
Treatment priorities. Initial treatment priorities after aSAH focus on preventing aneurysm re-rupture, managing active life-threatening conditions, and facilitating early, definitive aneurysm treatment. Anticoagulation reversal may help reduce the risk of aneurysm re-rupture and prevent neurosurgical bleeding complications. Careful blood pressure management requires avoiding hypotension to maintain cerebral perfusion while simultaneously preventing severe hypertension that could lead to re-bleeding. Short-acting, titratable I.V. antihypertensive medications can help maintain a systolic blood pressure below 160 mmHg or mean arterial pressure below 110 mmHg before aneurysm securement.
Early hydrocephalus, leading to elevated ICP, is common after aSAH. Placing an EVD can address symptomatic hydrocephalus. Nurses competent in EVD management should oversee this device, as over-drainage before aneurysm securement may increase aneurysm re-rupture risk. Ultimately, establishing an aneurysm securement plan, either via surgical clipping or endovascular occlusion, can help to definitively manage the source of bleeding and prevent re-occurrence.
After aneurysm securement, careful ongoing nursing neurologic assessments can aid identification of delayed cerebral ischemia (DCI). Angiographic narrowing of intracranial blood vessels (vasospasm) occurs in 30% to 70% of patients after aSAH, typically beginning about 3 days post-bleed and peaking around days 7 to 10. Some patients will experience ischemic, focal neurologic deficits. In addition to serial neurologic assessments, blood pressure management, euvolemia maintenance, oral or enteral administration of nimodipine (a calcium channel blocker), transcranial Doppler ultrasonography, and, if needed, intra-arterial vasospasm intervention, aid detection, prevention, and treatment of cerebral hypoperfusion and DCI.
Additional nursing care. Patients with aSAH need intensive nursing care. They often require long hospital stays due to the protracted nature of cerebral vasospasm and DCI after aneurysm rupture. In addition to managing hemodynamic care and diligently and continuously assessing neurologic conditions throughout hospitalization, nurses evaluate and manage pain. Severe headache, common after aSAH, necessitates a multimodal pain management strategy using the least-sedating and lowest effective analgesic dosages to maintain a reliable neurologic exam. Clustering nursing care (turning, suctioning, bathing) and maintaining 30-degree head-of-bed elevation can help prevent unnecessary ICP elevation.
As with all patients who’ve experienced a stroke, the risk of dysphagia requires conducting a swallowing assessment before oral intake. If the patient is stable, mobilize them early after aneurysm securement. In addition, maintain euvolemia. Both hyper- and hypovolemia after aSAH can result in poor outcomes. Care of patients with aSAH is lengthy and complex. Nurses play a critical role in optimizing patient outcomes across the continuum of care via ongoing detailed neurologic assessment and standardized management.
Optimize outcomes
Acute stroke, a medical emergency, requires urgent attention and well-coordinated care. Nurses are vital to the initial and ongoing care of these patients. Recognition, initial actions, and ongoing priorities throughout hospitalization differ among various stroke types. Familiarization with national standards and organizational protocols can help you advocate for patients and optimize outcomes.
The authors work in the neurocritical care unit at the Hospital of the University of Pennsylvania in Philadelphia. Megan Burns and Lauren Pomager are RNs and Bethany C. Young is a clinical nurse specialist.
American Nurse Journal. 2023; 18(1). Doi: 10.51256/ANJ012306
Key words: acute stroke, ischemic stroke, intracerebral hemorrhage, subarachnoid hemorrhage
References
Albers GW, Marks MP, Kemp S et al. Thrombectomy for stroke at 6 to 16 hours with selection by perfusion imaging. N Engl J Med. 2018;378(8):708-18. doi:10.1056/NEJMoa1713973
Diringer MN, Edwards DF. Admission to a neurologic/neurosurgical intensive care unit is associated with reduced mortality rate after intracerebral hemorrhage. Crit Care Med. 2001;29(3):635-40. doi:10.1097/00003246-200103000-00031
Greenberg SM, Ziai WC, Cardonnier C, et al. 2022 Guideline for the management of spontaneous intracerebral hemorrhage: A guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2022(7);53:e282-e361.
Hanley DF, Thompson RE, Rosenblum M, et al. Efficacy and safety of minimally invasive surgery with thrombolysis in intracerebral haemorrhage evacuation (MISTIE III): A randomised, controlled, open-label, blinded endpoint phase 3 trial. Lancet. 2019;393(10175):1021-32. doi:10.1016/S0140-6736(19)30195-3
Hickey JV. The Clinical Practice of Neurological and Neurosurgical Nursing. 8th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2019.
Hinduja A, Ahmed S. Emergency neurological life support: Acute ischemic stroke. Neurocritical Care Society. 2021. cdn.fs.pathlms.com/FQvmt72lQ5yAHwB18KRJ
Mainali S, Appavu B, John S. Emergency neurological life support: subarachnoid hemorrhage. Neurocritical Care Society. 2021. cdn.fs.pathlms.com/v5aXbWvRRSFu4fCXIpwD
Martino R, Foley N, Bhogal S, Diamant N, Speechley M, Teasell R. Dysphagia after stroke: Incidence, diagnosis, and pulmonary complications. Stroke. 2005;36(12):2756-63. doi:10.1161/01.STR.0000190056.76543.eb
Mendelow AD, Gregson BA, Rowan EN, et al. Early surgery versus initial conservative treatment in patients with spontaneous supratentorial lobar intracerebral haematomas (STICH II): A randomised trial. Lancet. 2013;382(9890):397-408. doi:10.1016/S0140-6736(13)60986-1
Nogueira RG, Jadhav AP, Haussen DC, et al. Thrombectomy 6 to 24 hours after stroke with a mismatch between deficit and infarct. N Engl J Med. 2018;378(1):11-21. doi:10.1056/NEJMoa1706442
Powers WJ, Rabinstein AA, Ackerson T, et al. Guidelines for the early management of patients with acute ischemic stroke: 2019 Update to the 2018 guidelines for the early management of acute ischemic stroke: A guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2019;50(12):e344-418. doi:10.1161/STR.0000000000000211
Rogers ML, Fox E, Abdelhak T, et al. Care of the patient with acute ischemic stroke (endovascular/intensive care unit-postinterventional therapy): Update to 2009 comprehensive nursing care scientific statement: A scientific statement from the American Heart Association. Stroke. 2021;52:e198-210. doi:10.1161/STR.0000000000000358
Saver JL. Time is brain—Quantified. Stroke. 2006;37(1):263-6. doi:10.1161/01.STR.0000196957.55928.ab
Singh V, Williamson C, Erklauer J. Emergency neurological life support: Intracerebral hemorrhage. Neurocritical Care Society. 2021. cdn.fs.pathlms.com/WXWvfqCSTqsac32EjJDP
Tsao CW, Aday AW, Almarzooq ZI, et al. Heart disease and stroke statistics—2022 update: A report from the American Heart Association. Circulation. 2022;145(8):e153–639. doi:10.1161/CIR.0000000000001052
Venema E, Groot AE, Lingsma HF, et al. Effect of interhospital transfer on endovascular treatment for acute ischemic stroke. Stroke. 2019;50(4):923-30. doi:10.1161/STROKEAHA.118.024091
Vermeulen MJ, Schull MJ. Missed diagnosis of subarachnoid hemorrhage in the emergency department. Stroke. 2007;38(4):1216-21. doi:10.1161/01.STR.0000259661.05525.9a
Virani SS, Alonso A, Benjamin EJ, et al. Heart disease and stroke statistics—2020 update: A report from the American Heart Association. Circulation. 2020;141(9):e139-596. doi:10.1161/CIR.0000000000000757
Yang P, Zhang Y, Zhang L, et al. Endovascular thrombectomy with or without intravenous alteplase in acute stroke. N Engl J Med. 2020;382(21):1981-93. doi:10.1056/NEJMoa2001123
















2 Comments. Leave new
Article was concise but covered valuable information to apply to patient care and, if needed, care to family and/or friends.
Thank you for your feedback!