Vigilant monitoring will maximize outcomes.
- After shock is recognized and I.V. fluid resuscitation is initiated, vasopressors and inotropes are used as supportive therapies in critical care settings.
- Safe and effective titration of these pharmacologic agents requires that nurses understand shock pathophysiology and hemodynamic monitoring.
CNE
1.5 contact Hours
Learning Objectives
Attacking anterior-wall myocardial infarction in time
How to recognize and respond to hypovolemic shock
- Identify receptors related to cardiovascular physiology.
- Differentiate the effects of vasopressors and inotropes.
- Discuss nursing care of patients receiving vasopressors and inotropes in the ICU.
The authors and planners of this CNE activity have disclosed no relevant financial relationships with any commercial companies pertaining to this activity. See the last page of the article to learn how to earn CNE credit.
Expiration: 2/1/24
Shock requires early recognition and quick action to prevent organ failure. (See Types of shock.) After initial I.V. fluid resuscitation, pharmacologic agents—such as vasopressors and inotropes—are used in critical care settings as supportive therapies to improve myocardial contractility, heart rate, and vascular resistance in patients with low cardiac output.
When critical care nurses understand shock pathophysiology and hemodynamic monitoring, they can effectively and safely titrate (increase or decrease an infusion rate for therapeutic effect) these medications using the lowest possible dose to avoid adverse effects.
Cardiovascular physiology
Shock disrupts cardiovascular physiology, particularly blood pressure, so understanding normal function is important. Short-term blood pressure (BP) regulation is controlled by the autonomic nervous system (ANS) via baroreceptors in the aortic arch and carotid sinus. The ANS regulates the heart, secretory glands, and smooth muscles via activation of the parasympathetic and sympathetic systems. Body processes activate the ANS in response to information produced by internal and external stimuli, including blood pressure, body temperature, heart rate, respiratory rate, metabolism, urination, defecation, digestion, and sexual response via chemical and neurotransmitter signaling.
The sympathetic system prepares the body for stressful or emergent situations, resulting in increased heart rate and contractility, vasoconstriction, and norepinephrine and epinephrine release, which increase BP. The parasympathetic system conserves and restores body processes during normal conditions, resulting in reduced heart rate, cardiac output, and BP.
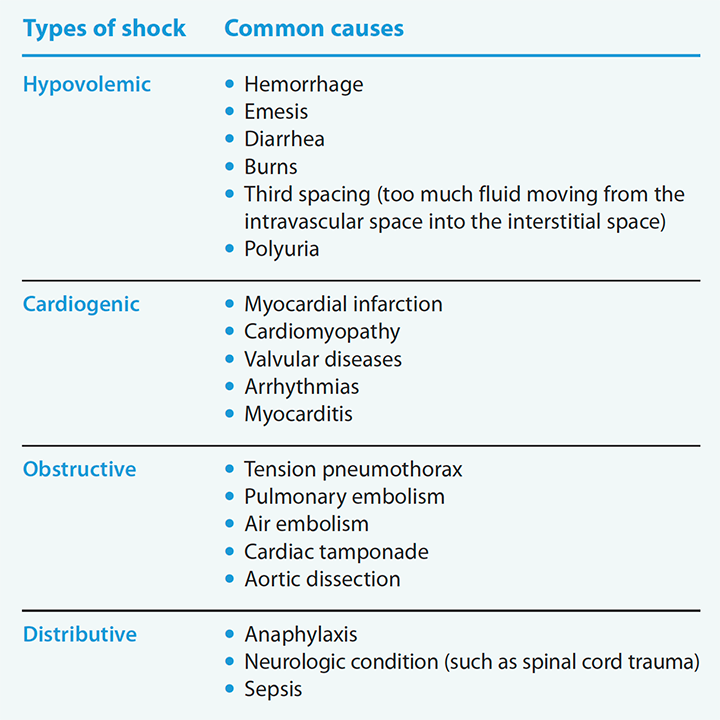
Types of shock
Shock is a decline in tissue perfusion and oxygen delivery, leading to cellular dysfunction and death. Organ hypoperfusion results from decreased circulating volume (hypovolemic shock), decreased cardiac output (cardiogenic and obstructive), and vasodilation (distributive).

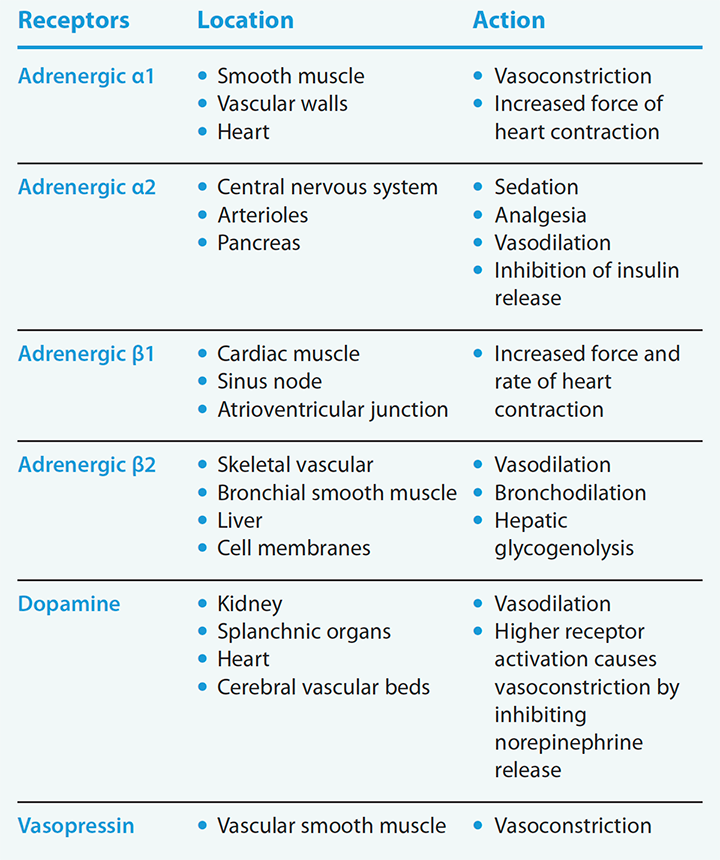
Receptors
Adrenergic receptors (which affect the sympathetic nervous system) within the heart and vascular system facilitate the hemodynamic effects of vasopressors via the interaction of catecholamines and synthetic medications such as norepinephrine and epinephrine. Adrenergic receptors are classified as alpha (a1, a2) or beta (b1, b2), and their location determines their physiologic response. Other receptors are vasopressin (V1 and V2; this article focuses on V1) and dopamine. (See Receptor physiology.)
Receptor physiology
Adrenergic (alpha and beta), dopamine, and vasopressin receptors exert actions on specific organs.

Adrenergic receptors. a1 receptors are located within the veins, arterioles, and many capillary beds. When stimulated, they produce vasoconstriction. a2 receptors also are located on the nerve terminals of the peripheral nervous system and function to inhibit transmitter release.
Beta receptors are found on the arterioles of the heart, smooth muscle tissues, and the lungs where they bind to circulating norepinephrine and epinephrine. b1, the dominant receptor found in the heart and kidney, enhances myocardial contractility and relaxation when it’s activated. It also increases heart rate and cardiac output. Systemic vasodilation may offset increased cardiac output, resulting in decreased arterial pressures. b1 receptor activation in the kidneys induces the release of renin into the blood, promoting angiotensin synthesis and elevating BP. b2 receptors, which are located in the arterioles of the heart, smooth muscle tissues, and lungs, help mediate many essential processes. Although b2 receptors cause vasodilation, their effect on cardiac output is less than that of b1. In addition, b2 receptors have a significant impact on smooth muscles in the airway and cause bronchodilation.
Dopamine receptors. Site-specific dopamine receptors also exert cardiovascular actions. Small doses of dopamine result in vasodilation. As activation increases, vasoconstriction occurs via stimulation of a1 receptors and inhibition of norepinephrine release. Dopamine receptor activation within the kidney vasculature leads to enhanced renal perfusion.
Vasopressin receptors. Arginine vasopressin, an antidiuretic hormone, is synthesized in the hypothalamus and controls the body’s osmotic balance, regulates BP, and influences water reabsorption in the kidneys. V1 receptors are found in vascular smooth muscles. Their stimulation results in vasoconstriction, primarily in the splanchnic, renal, and hepatic arteriole vasculature. Hypovolemia and decreased arterial blood volume stimulate vasopressin release via baroreceptors in the carotid sinuses and aortic arch, resulting in increased water retention that causes vascular smooth muscle constriction.
Supportive pharmacologic therapies
Vasopressors and inotropes are commonly used in the ICU to manage shock and support the cardiovascular system.
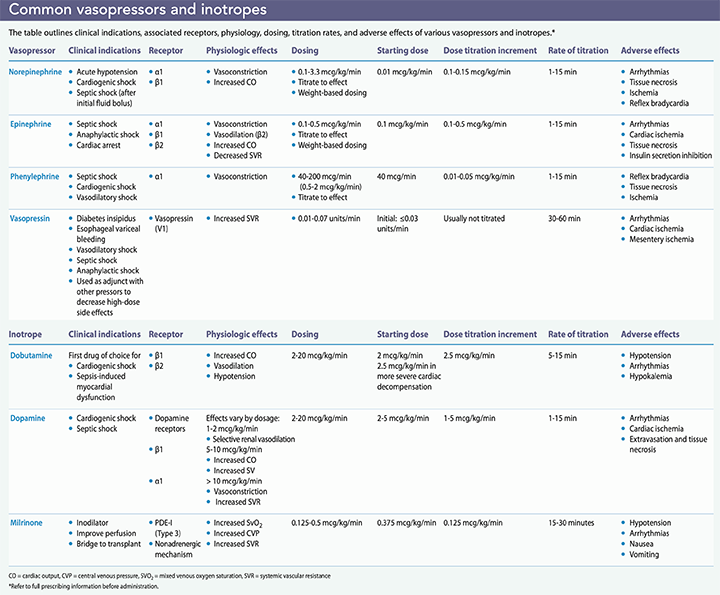
Vasopressors. Vasopressors are I.V. medications that produce arteriole vasoconstriction via positive inotropic or chronotropic effects that lead to increased systemic vascular resistance and BP. Common vasopressors are norepinephrine, epinephrine, phenylephrine, and vasopressin. Indications include a decrease in systolic pressure of more than 30 mmHg from baseline or mean arterial pressure (MAP) less than 60 mmHg resulting in end-organ dysfunction. (MAP, the average pressure in the arteries during one cardiac cycle, is the best indicator of organ perfusion.)
Vasopressors work on specific adrenergic receptors by activating endogenous or synthetic hormones to restore normal arterial pressure and organ perfusion during times of physical or emotional stress.
Inotropes. Inotropes, which are crucial to resuscitating patients with cardiogenic shock and heart failure, can exude positive or negative effects on the heart. Positive inotropic agents, which include dopamine and dobutamine, increase myocardial contractility, boosting cardiac output. These agents have been used to treat heart failure, but variability in conditions and heart rate may alter their effects. Adverse reactions associated with positive inotropic agents include arrhythmias, induced myocardial ischemia, and hypotension. Negative inotropic agents, which include beta blockers and calcium blockers (such as metoprolol, diltiazem, and verapamil), decrease the heart’s workload by reducing the force of muscle contraction; they are outside the scope of this article.
Hemodynamic monitoring and medication administration
Hemodynamic monitoring involves using invasive and noninvasive methods to gather information about a critically ill patient’s cardiovascular functioning for diagnosis and treatment guidance. Hemodynamic instability can be the result of hypovolemia or cardiac or vasomotor dysfunction, which can lead to organ dysfunction, multi-organ failure, or death. Noninvasive monitoring includes manual BP, electrocardiogram, temperature, respiratory rate, pulse oximetry, and urine output.
Invasive monitoring includes inserting an arterial line for continuous BP measurement and arterial blood gas analysis, and central venous catheterization or pulmonary artery catheters (PACs) for advanced hemodynamic monitoring to guide fluid resuscitation and the need for vasopressors or inotropic infusions.
Both invasive and noninvasive monitoring are helpful for assessing patients’ responses to medications, so that titration is effective. Here is a closer look at invasive techniques used for monitoring and medication administration.
Arterial catheterization
Vasopressor and inotropic drugs act quickly, so invasive BP monitoring, typically by arterial catheterization, may be required to safely titrate vasopressor and inotrope infusions. Arterial catheterization also provides access for diagnostic laboratory blood testing.
A trained provider places the arterial line using sterile technique. Radial and femoral sites are the most common, but other locations include the brachial, axillary, and dorsalis pedis artery. As the critical care nurse, you’re responsible for arterial transducer setup (including leveling) line patency, hemodynamic response assessment using arterial waveform interpretation, and insertion site care. Transducer setup requires tight tubing and catheter connections, a pressure bag set at 300 mmHg to maintain system patency, and a transducer leveled to the phlebostatic axis. The phlebostatic axis is located at the convergence of a vertical line drawn from the fourth intercostal space to the right of the sternum midway from the anterior to the posterior chest. Mark the site to ensure consistency for accurate pressure readings. Transducer leveling should be performed every shift or whenever a data discrepancy occurs.
The arterial line is a continuous measurement of heart rate, BP, systemic perfusion, MAP, pulse pressure, and pulse pressure variation. Arterial waveforms represent left ventricular pressure during systole and consist of three components: systolic phase (steep rise in arterial pressure from the lowest point to the peak), dicrotic notch (closure of the aortic valve), and the diastolic phase (lowest point).
The Square Wave Test ensures accuracy of the hemodynamic values. It’s performed by opening the continuous flush valve to a high-pressure signal, invoking transducer vibration from a “fast flush.” If the waveform is adequately dampened, only two oscillations follow the flush with a subsequent clear arterial waveform and a visible dicrotic notch.
Waveform dampening also can result from kinked or obstructed cannulas, loose connections, and air bubbles within the tubing or transducer, resulting in false low or high readings. Inaccurate and misinterpreted waveforms can affect infusion titrations and clinical endpoints.
Arterial line complications include severe blood loss, local or systemic infections, arterial thrombosis, air embolism, and accidental medication injection. (See Arterial line complications and nursing considerations.)
Arterial line complications and nursing considerations
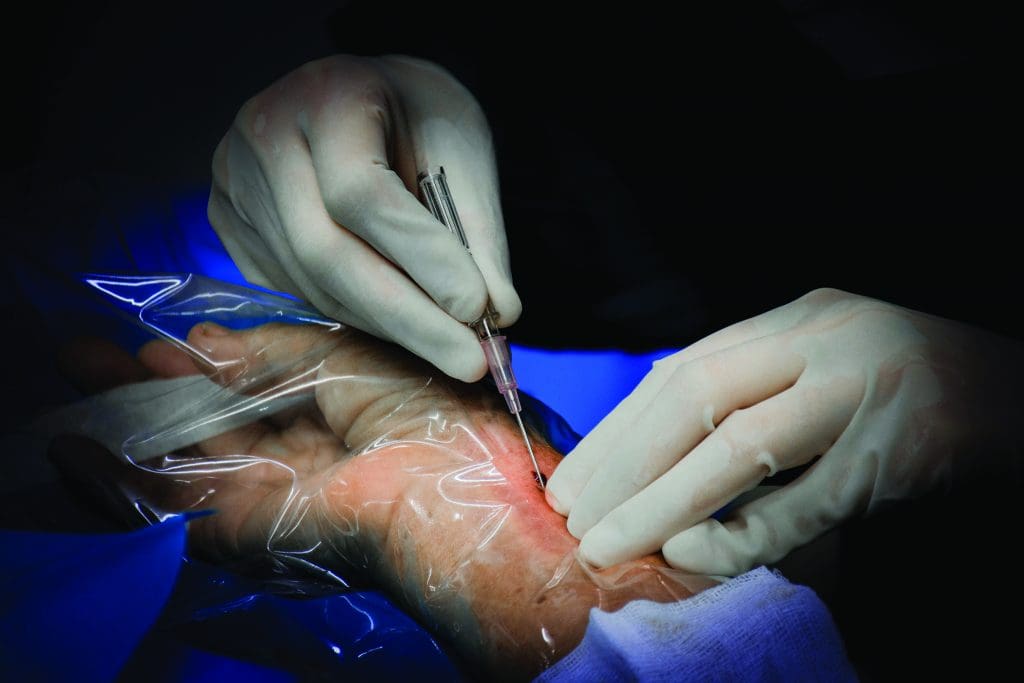

- Loose connections can lead to massive blood loss if undetected. Check for a dampened waveform or unusually low pressure reading, which may indicate a loose connection.
- Catheters placed in the radial artery can damage the radial nerve. To prevent injury, place an arm board to facilitate proper alignment.
- Hand ischemia or digital embolization may occur with lines placed in the radial artery. These complications may be caused by thrombosis, so don’t force flush the line, which may prove fatal.
Central venous catheterization
Central venous catherization (CVC) allows for central venous access using one of three options: the internal jugular vein, common femoral vein, or subclavian vein. CVC is used for multiple infusions, if peripheral access can’t be obtained, and to administer medications that may be caustic to tissues when given via peripherally inserted catheters.
Vasoactive infusions through a peripherally inserted venous catheter may cause severe tissue injury and necrosis with infiltration. Controversy exists over the absolute need for a central venous catheter instead of a peripherally inserted one to administer vasopressor and inotrope infusions. Considerations include drug concentration, infusion rate, and duration. However, the absence of central access shouldn’t hinder the initiation of these drugs, as failing to initiate therapy promptly could result in detrimental effects to the patient. If central access isn’t available, initiate the infusion at the lowest rate and concentration necessary to achieve directed hemodynamic goals, and carefully monitor the peripheral site.
If infiltration is suspected, immediately discontinue the infusion, aspirate any residual fluid from the catheter, irrigate the line with saline, elevate the affected area, apply warm or cold compresses per protocol, and notify the pharmacy and provider. Pharmacologic treatment for infiltration of vasopressors and inotropes includes subcutaneous administration of 5 to 10 mg of phentolamine mesylate in 10 mL of saline around the infiltration within 12 hours of extravasation to prevent local ischemia.
To prevent intravascular catheter-related infections, the Centers for Disease Control and Prevention recommends using aseptic technique when changing dressings and prompt removal when the catheter is no longer needed.
Pulmonary artery catheterization
PAC (Swan-Ganz catheter) has been the gold standard for cardiac output monitoring for nearly 50 years, primarily in cardiac surgery patients. The PAC is inserted through a central vein (femoral, jugular, antecubital, or brachial) and advanced through the right side of the heart to the pulmonary artery. It’s used to measure intrathoracic intravascular pressures in the right atrium (central venous pressure), pulmonary artery, and pulmonary veins (pulmonary wedge pressure). A PAC is particularly helpful in patients with right ventricular dysfunction or pulmonary arterial hypertension.
Routine PAC site care requires following organization policy for sterile dressing changes and documenting catheter placement and signs and symptoms of site infection. The catheter’s sterile sleeve integrity must be maintained to allow catheter movement without contamination. In addition to site care, monitor for dysrhythmias, check waveform and hemodynamic values, oversee patient activity and positioning, and remove the catheter as ordered.
Assessing patients with a PAC is essential for preventing and identifying potential complications, including balloon rupture, dysrhythmias, air embolism, pulmonary thromboembolism, pulmonary artery rupture, and infarction. Review daily chest imaging to check catheter position, and zero and level transducers after repositioning. Spontaneous balloon wedging is life-threatening and requires immediate intervention. Ensure the wedging syringe isn’t full of air or accidentally inflated and that the balloon is deflated. (See PAC ports.)
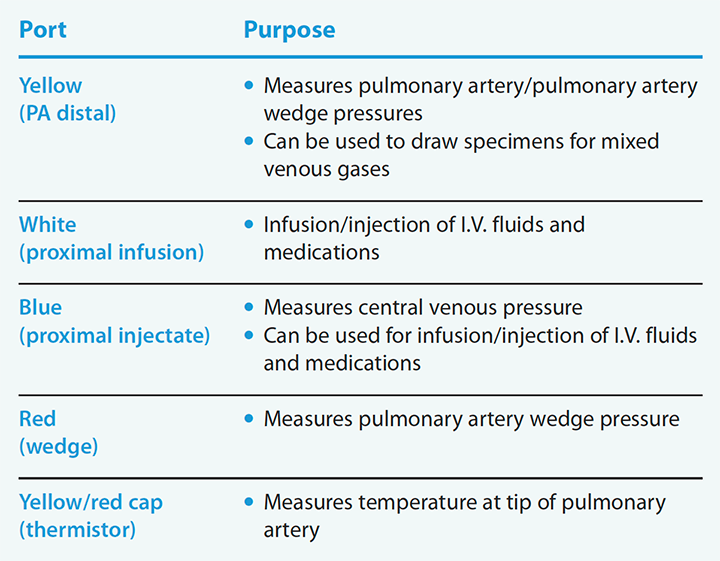
PAC ports
Critical care nurses should be familiar with the pulmonary artery catheter (PAC) port colors and their associated purposes.

Vasopressor and inotropic infusion titration
When the initial vasopressor or inotrope has been selected based on the underlying cause of shock, titrate the dose to achieve sufficient blood pressure or end-organ perfusion indicated by the MAP, as ordered by the provider. The most common target for vasopressor and inotropic titration is a MAP value of 60 mmHg to 65 mmHg.
Continuous vasopressor/inotropic infusions must be administered via infusion pump to prevent interrupted flow, unexpected changes in flow rates, and undesired boluses. Vasopressors have a short half-life and duration of action, so they require continuous infusion and rapid titration, usually every 5 to 15 minutes. You want to find a balance between hemodynamic stability and adequate perfusion while using the minimum dose needed. When hemodynamic stability has been achieved, begin weaning vasopressors to facilitate the lowest dose required to maintain the desired effects.
Your familiarity with organizational and unit titration policies will help ensure patient safety and reduce medication error and adverse event risks. Smart pumps can help reduce medication errors, but you must calculate and verify infusions before administration and confirm minimum and maximum doses.

Knowledge and competency
Hemodynamic monitoring and vasopressor and inotrope administration and titration are essential to ensuring sufficient tissue oxygenation and end-organ perfusion. Your knowledge of anatomy, physiology, and pharmacology, along with competent assessment skills, can ensure a safe outcome for your patients. AN
To view a list of references, visit myamericannurse.com/?p=71893.
Sonya M. Grigsby is a critical care advanced practice RN at Christus Mother Frances Hospital in Tyler, Texas.
References
Cardenas-Garcia J, Schaub KF, Belchikov YG, Narasimhan M, Koenig SJ, Mayo PH. Safety of peripheral intravenous administration of vasoactive medication. J Hosp Med. 2015;10(9):581-5.
Centers for Disease Control and Prevention. Summary of recommendations: Guidelines for the prevention of intravascular catheter-related infections. February 2017. cdc.gov/infectioncontrol/guidelines/bsi/recommendations.html
Chawla SPS, Kuar S. Vasopressors and inotropes in shock: Which one to choose? Sch J App Med Sci. 2015;3(2G):1027-34.
Cuzzo B, Padala SA, Lappin, SL. Physiology, vasopressin. StatPearls. August 29, 2020. ncbi.nlm.nih.gov/books/NBK526069
Huygh J, Peeters Y, Bernards J, Malbrain MLNG. Hemodynamic monitoring in the critically ill: An overview of current cardiac output monitoring methods. F1000Research. 2016;5:F1000.
Jacq G, Gritti K, Carré C, et al. Modalities of invasive arterial pressure monitoring in critically ill patients: A prospective observational study. Medicine (Baltimore). 2015;94(39):e1557.
Kitch BB. A guide to autonomic pharmacology: Understanding neurotransmitters and their receptors can help you choose the right medication. EMS World. 2017;46(1):30-9.
Lewis T, Merchan C, Altshuler D, Papadopoulos J. Safety of the peripheral administration of vasopressor agents. J Intensive Care Med. 2019;34(1):26-33.
Loubani OM, Green RS. A systematic review of extravasation and local tissue injury from administration of vasopressors through peripheral intravenous catheters and central venous catheters. J Crit Care. 2015;30(3):653.e9-17.
Low P. Overview of the autonomic nervous system. MERCK Manual. April 2020. merckmanuals.com/home/brain,-spinal-cord,-and-nerve-disorders/autonomic-nervous-system-disorders/overview-of-the-autonomic-nervous-system
Manaker S. Use of vasopressors and inotropes. UpToDate. July 3, 2020. uptodate.com/contents/use-of-vasopressors-and-inotropes?search=vasopressors%20and%20inotropes&source=search_result&selectedTitle=1~150&usage_type=default&display_rank=1
Ogle S, Kulungowski AM. Arterial line placement technique. 2020. emedicine.medscape.com/article/1999586-technique#c6.
Paik JJ, Hirpara R, Heller JA, Hummers LK, Wigley FM, Shah AA. Thrombotic complications after radial arterial line placement in systemic sclerosis: A case series. Semin Arthritis Rheum. 2016;46(2):196-9.
Plum M, Moukhachen O. Alternative pharmacological management of vasopressor extravasation in the absence of phentolamine. P T. 2017;42(9):581-92.
Procter LD. Shock. Merck Manual. October 2020. merckmanuals.com/professional/critical-care-medicine/shock-and-fluid-resuscitation/shock
Rodriguez Ziccardi M, Khalid N. Pulmonary artery catheterization. StatPearls. September 20, 2020. ncbi.nlm.nih.gov/books/NBK482170
Rook WH, Turner JD, Clutton-Brock TH. Analysis of damping characteristics of arterial catheter blood pressure monitoring in a large intensive care unit. South Afr J Crit Care. 2017;33(1):8-11.
Scheeren TWL, Ramsay MAE. New developments in hemodynamic monitoring. J Cardiothorac Vasc Anesth. 2019;33(Suppl 1):S67-72.
Souza VN, dos Santos AC, de Lima Vesco N, Lima ACS, dos Santos RC, Florencio RS. Pulmonary artery catheter: Nursing care related to postoperative cardiac transplantation patients. J Nurs UFPE. 2017;11(5):1769-75. doi: 10.5205/reuol.11077-98857-1-SM.1105201703
Teboul JL, Saugel B, Cecconi M, et al. Less invasive hemodynamic monitoring in critically ill patients. Intensive Care Med. 2016;42(9):1350-9.
Theodore AC, Clermont G, Dalton A. Intra-arterial catheterization for invasive monitoring: Indications, insertion techniques, and interpretation. UpToDate. August 18, 2020. uptodate.com/contents/indications-interpretation-and-techniques-for-arterial-catheterization-for-invasive-monitoring
Waghmare LS, Srivatava TK. Conceptualizing physiology of arterial blood pressure regulation through the logic model. Adv Physiol Educ. 2016;40(4):477-9.