Assessment is key for optimal management.
Takeaways:
- An early symptom of Alzheimer’s disease is decline in cognition, most typically manifesting as short-term memory loss. Early symptoms of Lewy body dementia are characterized by cognitive, behavioral, movement, and autonomic changes.
- Common early signs of Lewy body dementia include visuospatial disturbances, vivid hallucinations, bradykinesia and rigidity and postural hypotension, loss of smell, and constipation.
- Strategies for managing symptoms in these two conditions vary.
- Screening and early dementia diagnosis provide opportunities to pursue health behaviors that can reduce risk, for earlier medication initiation to help moderate symptoms, to slow progression, and for long-term care planning.
CNE
1.75 contact Hours
Learning Objectives
- Compare Alzheimer’s disease and Lewy body dementia as to pathophysiology, signs, symptoms, and management.
- Describe how to screen for memory loss.
The authors and planners of this CNE activity have disclosed no relevant financial relationships with any commercial companies pertaining to this activity. See the last page of the article to learn how to earn CNE credit.
Expiration: 1/1/24
As the U.S. population ages, the risk for age-related illnesses such as Alzheimer’s disease and other types of dementia increases. According to Lang and colleagues, about 50% of individuals with dementia are undiagnosed. When dementia is diagnosed early, opportunities to address it increase. These opportunities include pursuing health behaviors that reduce progression, initiating medications earlier to help moderate symptoms and to potentially slow progression, as well as participating in research and advanced care planning. Nurses in all settings must increase their knowledge and understanding of dementia to ensure early detection.
What is dementia?
Dementia is any syndrome that manifests in three main sets of symptoms: debilitating cognitive decline, decreased independence in activities of daily living, and behavioral changes. Alzheimer’s disease (AD) is the most common type of dementia and is sometimes used incorrectly to include all forms of dementia. Another common type of neurodegenerative dementia is Lewy body dementia (LBD), which consists of two diseases: dementia with Lewy bodies and Parkinson’s disease dementia. Other causes of dementia include cerebrovascular disease and frontotemporal degeneration. In many cases, the presentation of dementia is characterized as “mixed,” meaning that more than one pathological process is at play. Frequently, mixed dementias involve both vascular dementia and a neurodegenerative disease. In this article, we review the core features and current management approaches for AD and LBD.
Alzheimer’s disease
According to the Alzheimer’s Association’s 2020 Alzheimer’s Disease Facts and Figures Report, more than 5 million Americans are living with AD, and that number is projected to reach 7 million by 2025. Traditionally viewed as a memory plus syndrome, AD dementia typically presents as a progressive episodic memory decline and impairment in a second cognitive domain such as executive function or language. Such decline may involve the inability to recall recently learned information, create new memories, or recognize familiar faces or objects. For example, someone with AD may repeat a question within minutes, or tell the same story they told earlier the same day. Other common features include forgetting important dates or events, misplacing items, word-finding difficulty, impaired judgment and decision making, and needing more help from family members to perform daily tasks.
As the disease progresses throughout the brain, symptoms become more severe and eventually encompass all major aspects of cognition and function. In addition, patients may exhibit significant behavioral changes, including wandering, irritability, agitation, aggression, paranoid delusions, and depression.
Pathophysiology
Alois Alzheimer is credited with the 1906 discovery of the neuropathological changes associated with AD. Specifically, abnormalities in two proteins, beta amyloid and tau, represent the neuropathological hallmarks of AD. Excess beta-amyloid proteins form plaques in the extracellular space and are thought to impair neuron-to-neuron communication. Tau neurofibrillary protein tangles cluster within neurons, which affects neuron health and causes cell death. The beta amyloid and tau changes lead to inflammation and brain atrophy. Recent evidence highlighted by the Alzheimer’s Association points to amyloid plaques and tau tangles depositing 20 years or more before the first AD symptoms appear and formal diagnosis.
Together, these cellular alterations lead to cognitive changes that manifest as memory loss, functional impairment, and behavioral symptoms. Previously, amyloid and tau accumulations were detectable only at autopsy, but they’re now detectable in positron emission tomography (PET) scans and their presence can be inferred from abnormal findings in cerebrospinal fluid (CSF).
The first recognizable symptomatic stage of dementia is called mild cognitive impairment (MCI), which is characterized by subjective complaints of cognitive changes and objective deficits that are detectable during cognitive testing; everyday functioning remains mostly normal. MCI was operationalized in sentinel articles by Petersen and is now widely regarded as a risk state for AD. In recently published practice guidelines, Peterson and colleagues note that people over age 65 with a diagnosis of MCI are three times more likely to develop AD. Up to 55% of those with MCI may revert to normal cognitive function over time. MCI also can be a precursor to other forms of dementia.
AD stages
AD stages are classified as mild (early stage), moderate (middle stage), and severe (late stage). The mild stage is characterized by readily detectable cognitive ability impairment with some impairment in daily function. Affected individuals may or may not be aware of the cognitive and functional deficits. Behavioral symptoms such as mood changes and apathy may begin in the mild stage.
In the moderate stage, cognitive and functional symptoms progress and individuals struggle to complete instrumental and basic activities of daily living (such as dressing, bathing, and sleeping) on their own. Behavioral symptoms may be more pronounced and can include delusions, agitation, hallucinations, and disinhibition. Ornstein and Gaugler found that these behavioral symptoms are associated with family caregiver depression and burden.
In the severe stage, people with AD may lose the ability to communicate, become unable to attend to activities of daily living (such as eating and bathing), and experience loss of awareness of their current surroundings. These individuals frequently require 24-hour care until death.
AD medications
Although current medications won’t slow or prevent AD onset and progression, two categories of medications have received Food and Drug Administration approval for treating symptoms. Information about management is presented by Austrom and other experts in the Dementia Care Practice Recommendations from the Alzheimer’s Association.
Cholinesterase inhibitors. Donepezil, rivastigmine, and galantamine are cholinesterase inhibitors approved to treat mild-to-moderate AD symptoms. Donepezil also is approved to treat severe AD symptoms. Cholinesterase inhibitors interfere with the breakdown of the neurotransmitter acetylcholine (which plays a key role in attention and memory) and help increase the brain’s circulating supply.
These medications are available as pills and liquids; donepezil is available as an orally dissolving tablet, and rivastigmine can be administered via a transdermal patch.
The most common side effects of cholinesterase inhibitors—diarrhea, nausea, vomiting, fatigue, loss of appetite and weight, and insomnia—are transient and subside within the first month of treatment. However, bradycardia and syncope may develop later in the course of treatment, and decisions about benefit vs. harm must be made in collaboration with the patient’s care team.
N-methyl-D-aspartate (NMDA) receptor antagonists. Memantine is the only drug in this class indicated for moderate or severe AD, either alone or in combination with a cholinesterase inhibitor. Memantine is neuroprotective but also may help diminish symptoms of agitation, aggression, and delusion. It works by blocking the binding of glutamate (a neurotransmitter that’s harmful when present in excess) to its receptor. A fixed combination of donepezil and memantine also is available but is used less frequently due to its high cost and inability to adjust the doses of its individual components.
Memantine is available in pill as both immediate and extended-release forms. It’s also available as a liquid.
The most common side effects of memantine—headache, confusion, dizziness, and constipation—are transient and occur during the initial titration.
Lewy body dementia
LBD affects over 1.4 million Americans. It’s more common in men than women and progresses more quickly than AD. LBD typically begins at age 50 and older.
LBD is an umbrella term that encompasses two related conditions: Parkinson’s disease dementia (which occurs when cognitive decline develops after the motor symptoms of Parkinson’s disease are established) and dementia with Lewy bodies (which occurs when cognitive symptoms begin before or concurrently with motor symptoms). According to Galvin, no unique sign or symptom distinguishes dementia with Lewy body from Parkinson’s dementia; however, time of onset frequently is used to determine which LBD subtype is present. If dementia occurs before or with the onset of motor symptoms associated with Parkinson’s disease, then dementia with Lewy body is suspected. If the dementia occurs 12 or more months after Parkinsonism, then Parkinson’s disease dementia is diagnosed. Although this 1-year rule continues to be debated, discerning if AD or LBD is present will help direct treatment. Additional neurologic testing can help to determine if dementia with Lewy body or Parkinson’s disease dementia is responsible for most of the symptoms. For example, the Lewy Body composite risk score can help detect LBD by focusing the clinical interview on common LBD symptoms (psychomotor slowing, hallucinations, rigidity).
Pathophysiology
About 80% of patients with LBD will have co-existing AD pathology (amyloid plaques and tau tangles). However, the signature pathology is a Lewy body composed of abnormal intraneuronal aggregations of the presynaptic protein alpha-synuclein. Friederich Lewy first described Lewy bodies in 1912, but alpha synuclein wasn’t described until the late 1990s.
Symptoms
How Lewy bodies accumulate in the brain may predict its clinical presentation. If they begin accumulating in the brain stem, the result may be autonomic and motor symptoms; Lewy bodies beginning in the neocortex may lead to cognitive or behavioral symptoms. Symptom presentation is classified into four domains: cognition, behavior, movement, and autonomic function. (See LBD vs. AD symptoms.) A consortium of experts led by McKeith established that the diagnosis of LBD is made in the presence of dementia combined with at least two core criteria: parkinsonism, cognitive fluctuations, visual hallucinations, and rapid eye movement sleep behavior disorder (RBD).
LBD vs. AD symptoms
The table compares selected Lewy body dementia (LBD) symptoms and common features of Alzheimer’s disease (AD) in four domains.
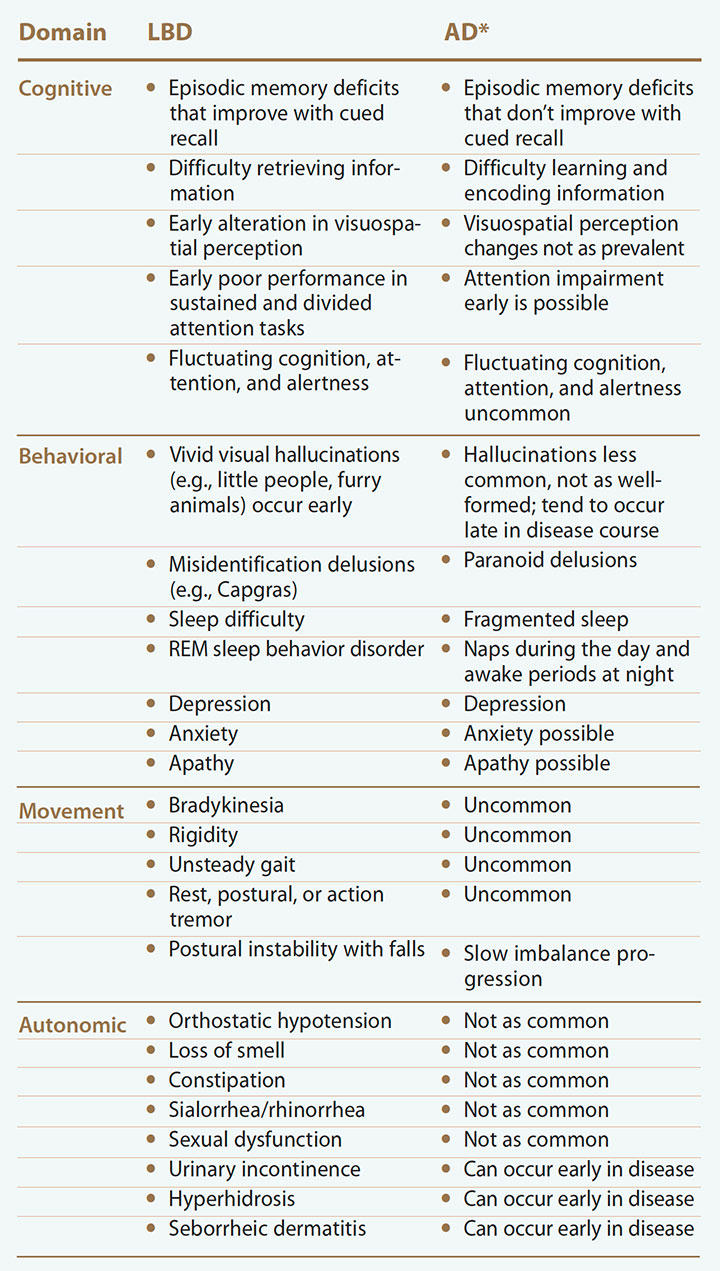

*Without influence of medication effects or other underlying illness
Cognitive symptoms. Fluctuating cognition includes seemingly spontaneous changes in attention and alertness, staring spells, on/off periods of lethargy, and incoherent thought that can change from minute to minute, hour to hour, or day to day. Cognition fluctuations can be assessed using tools such as the Mayo Fluctuations Questionnaire, the Clinical Assessment of Fluctuation, and the One-day Fluctuation Assessment Scale.
Similar to people with AD, those with LBD may have memory impairment. However, according to Galvin, one difference is that patients with LBD sometimes benefit from cued recall (using cues to jog memory when free recall fails). In addition to memory, prominent symptoms in attention, executive function, and visual perception occur early in the course of LBD. Although no single clinical indicator of LBD exists, careful early assessment for visuospatial impairment can help differentiate LBD from other dementias.
Behavioral symptoms. Prominent visual hallucinations are a key distinguishing hallmark of LBD. When present, they typically occur early in the course of the disease. The hallucinations are well formed and typically of small or dysmorphic people, children, or animals. Auditory or olfactory hallucinations also may occur but are less common. Delusions frequently occur in LBD. These delusions include Capgras syndrome, where the person believes that a family member or friend has been replaced by an identical imposter.
One of the most overlooked but earliest symptoms of LBD is sleep difficulty and RBD, which is characterized by acting out dreams during sleep through violent movements, talking, and falling out of bed. Other common symptoms are excessive daytime sleepiness (2 or more hours of sleepiness daily), restless leg syndrome (walking or movement is needed to help relieve uncomfortable leg sensations), and increased discomfort at rest.
People with LBD, similar to those with AD, frequently experience depression, exhibited by feelings of worthlessness, sadness, and trouble eating or sleeping. Other behavior and mood changes include apathy or lack of desire to participate in previously enjoyed activities, agitation (which manifests as restlessness and unexplained irritation), intense levels of anxiety (which manifests as anger or fear when a loved one isn’t present), and fear about a future event.
Movement disorder. Many patients with LBD experience Parkinsonism symptoms; by definition, all patients with the Parkinson’s disease dementia form of LBD experience them. Early signs may include changes in facial expression, hand posture, walking, balance, and handwriting. LBD motor symptoms include bradykinesia (slowness in initiating or carrying out a movement), rigidity (stiffness), and noticeable gait change such as shuffling (festination). Unstable posture (such as stooping over) and balance problems that lead to increased falls also frequently are associated with LBD. Other common symptoms include diminished facial expressions, difficulty swallowing, and tremor or shaking at rest.
Autonomic/constitutional changes. Lewy body deposits affecting the autonomic nervous system produce autonomic or constitutional symptoms—changes in bodily operations that patients have no intentional control over. Consequently, those with LBD may have orthostatic hypotension, anosmia (loss of smell), and frequent constipation. Other commonly observed symptoms include hyperhidrosis (excessive sweating), sialorrhea (excessive salivation), seborrheic dermatitis (itchy rash with flaky skin), rhinorrhea (runny nose), sexual dysfunction, urinary incontinence, and heat and cold insensitivity. Consistent nursing support is essential to effectively manage these complex symptoms.
LBD medications
If a patient with LBD is misdiagnosed as having a psychiatric illness because of symptoms such as hallucinations and illogical thinking, providers may prescribe medications that are detrimental. For example, carbidopa/levodopa, although helpful for patients with Parkinson’s disease, may exacerbate hallucinations in those with LBD. Antipsychotic medications may relieve hallucinations but also could exacerbate rigidity and motor slowness.
No medications have been approved in the United States to specifically treat LBD, but rivastigmine does have approval to treat symptoms of mild-to-moderate Parkinson’s disease dementia. Treatment should be based on the four LBD domains; however, all treatment is off-label. (See LBD pharmacologic treatment options.) To treat cognitive symptoms, cholinesterase inhibitors and NDMA receptor antagonists used in AD frequently are prescribed for LBD. Cholinesterase inhibitors may show a more robust initial response in LBD compared with AD. For motor symptoms, carbidopa/levodopa may help reduce Parkinsonian symptoms such as slow gait and muscle rigidity. Unfortunately, these medications may increase confusion and hallucinations so titration should be performed slowly.
LBD pharmacologic treatment options
No pharmacologic treatments have been approved to treat Lewy body dementia (LBD) in the United States. However, the following medications are used off-label to treat LBD symptoms.
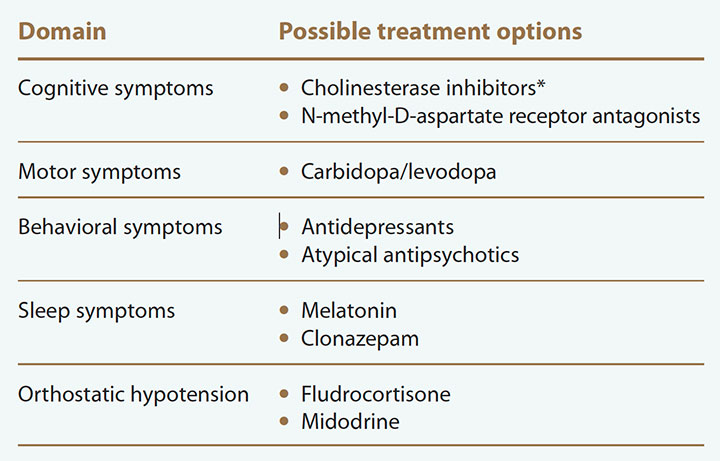

*Donepezil has been approved to treat LBD in Japan and the Philippines, and rivastigmine has been approved to treat Parkinson’s disease dementia in the United States and Europe.
No medications have been approved to manage LBD behavioral symptoms, and many common psychotropic medications can have deleterious side effects in patients with LBD. In general, all classic neuroleptic medications, such as haloperidol, should be avoided. When possible, nonpharmacologic approaches should be attempted to alleviate these symptoms. (To learn more about nonpharmacologic strategies, visit myamericannurse.com/?p=69690.) If necessary, careful use of atypical antipsychotic medications (such as quetiapine or pimavanserin) can be tried but require close monitoring. Attention to physical ailments such as constipation and pain should be the first priority because they can trigger behavioral changes. Mood symptoms are common in patients with LBD. Selective serotonin reuptake inhibitors, which help increase the amount of serotonin in the brain, appear to reduce irritability, agitation, and depression. RBD frequently is treated with melatonin or clonazepam.
Nonpharmacologic management of dementia
Applying Florence Nightingale’s environmental theory is particularly useful when supporting patients with dementia. Environmental interventions differ depending on the dementia stage and, in some instances, the etiology of the impairment.
- In early stage Alzheimer’s disease, clearly displayed reminders and notes can help an individual maximize independence and personal safety, but written cues may be less effective over time.
- If a patient with Lewy body dementia (LBD) is experiencing rapid eye movement sleep behavior disorder, the bedroom environment may need to be modified to prevent patient or sleep partner injury.
- In general, eliminating distracting noises, reducing clutter in the home, and talking in soothing tones are key strategies across all levels of dementia severity.
- Nightingale’s admonition to avoid “chattering advice” is helpful, as is refraining from quizzing or correcting a person with any dementia; these confrontational techniques may trigger or exacerbate agitation.
- Recognizing and validating the concerns of the person with dementia is important to reducing tension and frustration.
- Maintaining a daily routine as much as possible, keeping tasks simple, and providing instructions, one step at a time, can help diminish the frequency of dementia-related outbursts.
Alsace and colleagues demonstrated that behavioral symptoms frequently signal unmet needs in the person with dementia at any stage of the illness. Understanding the “need-driven behavior” is a gold standard for providing effective nursing care for individuals exhibiting dementia behaviors.
Research
Research indicates that nonpharmacologic symptom management for any type of dementia is at least as important as medications.
- Park and colleagues report that emerging strategies—such as chair yoga, exercise, and music—have shown significant improvement in patient mood and behaviors.
- In a critical review of publications spanning a decade, Cammisuli and colleagues found that behavioral interventions can be categorized as
- holistic techniques—reminiscence therapy and compensation techniques (such as color-coded reminders)
- brief psychotherapy—person-centered and solution-based
- cognitive stimulation therapy—spaced retrieval (providing answers to repetitive questions to increase retention and recall of meaningful items) and cognitive stimulation games
- alternative methods—checklists and memory notebooks.
- An extensive review of literature published from 2000 to 2018 by Zucchella and colleagues found that holistic interventions—such as reminiscence therapy, cognitive stimulation therapy, and validation therapy—in achieving person-centered care have been successful, although more research is needed. Other nonpharmacologic interventions cited as achieving some success based on the study were music therapy, exercise, aromatherapy, touch therapy, bright light therapy, and therapy combinations.
Nurses have an obligation to inform the public that the phrase “clinical studies,” which frequently appears on nutritional supplements claiming to treat or delay memory impairment, is not the same as “rigorous scientific studies showing safe and effective use in humans.” Refer patients and families to research reports from the Global Council on Brain Health (aarp.org/health/brain-health/global-council- on-brain-health/). The reports highlight findings that the only behaviors proven to delay dementia onset are consistent exercise, improved nutrition, socialization, and chronic disease management.
Treating autonomic symptoms is challenging and requires a holistic nursing approach, including identifying therapies targeted to each symptom. For example, constipation and diaphoresis in the context of orthostatic hypotension require comfort measures combined with safety strategies. Effective nursing care will make a critical difference in preventing or mitigating the physical health challenges associated with dementia syndromes.
Screening for memory loss
Because of the importance of early detection, cognitive screening is now a required component of the Medicare/Medicaid annual wellness visit. Several measures are available that nurses can use to briefly assess for risk of cognitive decline. For example, the Quick Dementia Rating Scale (QDRS) is a 5-minute informant interview that addresses ten cognitive domains: memory and recall, orientation, decision-making and problem-solving abilities, activities outside the home, function at home and hobbies, toileting and personal hygiene, behavior and personality changes, language and communication abilities, mood, and attention and concentration. The QDRS format helps to avoid problems such as patient anxiety with cognitive testing, which can lead to refusing to be tested. The scale also can be completed by family members. Scores of the 30-item scale range from 0 (no impairment in any domain) to 30 (indicating severe cognitive impairment). The QDRS has strong reliability and validity when correlated with other common cognitive assessment tools, such as the Clinical Dementia Rating scale (r=0.80, P <.001) and the Mini-Mental State Exam (r=-.599, P <.001); the Mini-Mental State Exam requires payment for use. In addition, the QDRS provides enough information to stage dementia severity, and it can be used by clinicians to determine the need for more formal testing.
The Montreal Cognitive Assessment (MoCA), developed by Nasreddine and colleagues, is a performance-based assessment that addresses attention, memory, concentration, orientation, visuoconstruction skills, and language. Julayanont and colleagues’ MoCA-B is geared toward individuals with health literacy and education levels below 8th grade. The MoCA is available in multiple languages, including Spanish, Chinese, and Hindi and also as a telephone Mini-MoCA, which was designed for use during periods of quarantine. These assessments are available at MoCAtest.org. Galvin’s Lewy Body Composite Risk Score assesses LBD risk and can differentiate LBD from AD with high accuracy, validity, and reliability. (See Lewy body composite risk score assessment.)
Lewy body composite risk score assessment
The Lewy body composite risk score is an accurate, valid, and reliable tool for differentiating between Lewy body dementia and Alzheimer’s disease.


Reprinted with Permission from Dr. James E. Galvin. Galvin JE. Improving the clinical detection of Lewy body dementia with the Lewy body composite risk score. Alzheimers Dement (Amst). 2015;1(3):316-24.
The science behind dementia detection continues to change rapidly. Patients identified with dementia ideally should receive brain imaging (non-contrast computed tomography or magnetic resonance imaging) to rule out other causes. PET and CSF biomarkers can be used to diagnose AD by testing for the presence of amyloid plaque and tau proteins in the brain or CSF. These tests are expensive.
Dopamine transporter imaging using single PET scans can be used to diagnose Parkinson’s disease. Blood tests aren’t yet available for these dementias, but advances are being made. For example, Thijssen and colleagues tested for the presence of pTau181 protein in the blood plasma of 400 individuals and were successful in distinguishing those who were healthy from those with AD pathology. These findings were corroborated with two established biomarkers (presence of pTau181 in spinal fluid and amyloid protein in PET scans) being used in AD research.
After screening
If screening indicates that a patient is at risk for cognitive decline, they should be referred to a board-certified advanced gerontological nurse practitioner, advanced practice registered nurse gerontological specialist, neurologist, or other provider comfortable with diagnosing and treating cognitive impairment.
These providers will consider other causes of cognitive decline, such as negative medication effects; trauma; depression; or vitamin B-12, folate, or thyroid deficiency. Symptom-directed treatment is more effective the earlier it’s initiated in the disease process. Education to adapt healthy behaviors may potentially delay a patient’s decline, and participation in research studies is an option for those in the early stages of dementia. (See Modifying behaviors to decrease dementia risk.)
Modifying behaviors to decrease dementia risk
In the past, it was believed that nothing could be done to delay the onset of dementia or slow its progress. However, research on lifestyle modifications that may diminish risk factors for dementia is growing. Recommended strategies that may reduce Lewy body dementia risk include
- refraining from smoking
- maintaining a healthy diet
- getting adequate sleep
- participating in cognitively stimulating activities
- interacting with others socially
- exercising.
Edwards and colleagues identified several medical (obesity, diabetes, hypertension, elevated cholesterol, depression) and lifestyle (dietary fat and cholesterol consumption, smoking, lack of physical activity) Alzheimer’s disease (AD) risk factors that may be modifiable with health-promoting interventions. In August 2020, Livingston and colleagues added other risk factors to this list—traumatic brain injury, pollution, and excessive alcohol consumption. They reported that all of the modifiable risk factors account for about 40% of worldwide dementias that could be theoretically prevented or delayed.
Recent evidence from Haslam and colleagues’ work with the Framingham Heart Study about the impact of sugary drink consumption indicates that even a small but consistent change in diet, such as eliminating sugar-sweetened soft drinks, may help reduce dementia risk. Improving nutritional habits to include more fruits and vegetables, beans, nuts, whole grains, olive oil, and fish (the MIND [Mediterranean-DASH Intervention for Neurodegenerative Delay] diet) should be encouraged to promote brain health. Also, promising new biomarkers may aid in early dementia detection.
Ease the burden
As the population ages, nurses need to learn more about AD and LBD. Distinguishing between these two conditions is critical to ensure proper treatment and symptom management. Regular screening and early dementia detection offer opportunities for nurses and families to partner in research and symptom management and ease illness burden. AN
Lisa Kirk Wiese is an associate professor of nursing at the Florida Atlantic University C. E. Lynn College of Nursing in Boca Raton. Jennifer Lingler is professor and vice chair for research at the Health and Community Systems University of Pittsburgh School of Nursing in Pittsburgh, Pennsylvania. Allison Lindauer is an assistant professor of neurology at the School of Medicine Oregon Health and Science University and the Layton Aging and Alzheimer’s Disease Center director of outreach, recruitment, and education in Portland.
To view a list of references, visit myamericannurse.com/?p=69690.
References
Algase DL, Beck C, Kolanowski A, et al. Need-driven dementia-compromised behavior: An alternative view of disruptive behavior. Am J Alzheimers Dis. 1996;11(6):10-9.
Alzheimer’s Association. 2020 Alzheimer’s Disease Facts and Figures Report. alz.org/media/Documents/alzheimers-facts-and-figures.pdf
Alzheimer’s Association. Dementia care practice recommendations. 2020. alz.org/professionals/professional-providers/dementia_care_practice_recommendations
Austrom MG, Boustani M, LaMantia MA. Ongoing medical management to maximize health and well-being for persons living with dementia. Gerontologist. 2018;58(suppl 1):S48-57.
Cammisuli DM, Danti S, Bosinelli F, Cipriani G. Non-pharmacological interventions for people with Alzheimer’s disease: A critical review of the scientific literature from the last ten years. Eur Geriatr Med. 2016;7(1):57-64.
Daviglus ML, Bell CC, Berrettini W, et al. NIH state-of-the-science conference statement: Preventing Alzheimer’s disease and cognitive decline. NIH Consens State Sci Statements. 2010;27(4):1-30.
de Oliveira AM, Radanovic M, de Mello PCH, et al. Nonpharmacological interventions to reduce behavioral and psychological symptoms of dementia: A systematic review. BioMed Res Int. 2015;2015:218980.
Edwards GA III, Gamez N, Escobedo G Jr, Calderon O, Moreno-Gonzalez I. Modifiable risk factors for Alzheimer’s disease. Front Aging Neurosci. 2019;11:146.
Ferman TJ, Smith GE, Boeve BF, et al. DLB fluctuations: Specific features that reliably differentiate DLB from AD and normal aging. Neurology. 2004;62(2):181-7.
Galvin JE. Improving the clinical detection of Lewy body dementia with the Lewy body composite risk score. Alzheimers Dement (Amst). 2015;1(3):316-24.
Galvin JE. Lewy body dementia. Practical Neurology. June 2019. practicalneurology.com/articles/2019-june/lewy-body-dementia-1
Galvin JE. The Quick Dementia Rating System (QDRS): A rapid dementia staging tool. Alzheimers Dement (Amst). 2015;1(2):249-59.
Haslam DE, Peloso GM, Herman MA, et al. Beverage consumption and longitudinal changes in lipoprotein concentrations and incident dyslipidemia in US adults: The Framingham Heart Study. J Am Heart Assoc. 2020;9(5):e014083.
Julayanont P, Tangwongchai S, Hemrungrojn S, et al. The Monteral Cognitive Assessment–Basic: A screening tool for mild cognitive impairment in illiterate and low-educated elderly adults. J Am Geriatr Soc. 2015;63(12):2550-4.
Karantzoulis S, Galvin JE. Update on dementia with Lewy bodies. Curr Transl Geriatr Exp Gerontol Rep. 2013;2(3):196-204.
Lang L, Clifford A, Wei L, et al. Prevalence and determinants of undetected dementia in the community: A systematic literature review and a meta-analysis. BMJ Open. 2017;7:e011146.
Livingston G, Huntley J, Sommerland A, et al. Dementia prevention, intervention, and care: 2020 report of the Lancet Commission. Lancet. 2020;396(10248):413-46.
McKeith IG, Boeve BF, Dickson DW, et al. Diagnosis and management of dementia with Lewy bodies: Fourth consensus report of the DLB Consortium. Neurology. 2017;89(1):88-100.
Nasreddine ZS, Phillips NA, Bédirian V, et al. The Montreal Cognitive Assessment, MoCA: A brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005; 53(4):695-9.
Ornstein K, Gaugler JE. The problem with “problem behaviors”: A systematic review of the association between individual patient behavioral and psychological symptoms and caregiver depression and burden within the dementia patient–caregiver dyad. Int Psychogeriatr. 2012;24(10):1536-52.
Park J, Howard H, Tolea MI, Galvin JE. Perceived benefits of using nonpharmacological interventions in older adults With Alzheimer’s disease or dementia with Lewy bodies. J Gerontol Nurs. 2020;46(1):37-46.
Petersen RC. Mild cognitive impairment as a diagnostic entity. J Intern Med. 2004;256(3):183-94.
Petersen RC, Lopez O, Armstrong MJ, et al. Practice guideline update summary: Mild cognitive impairment. Neurology. 2018;90(3):126-35.
Petersen RC, Smith GE, Waring SC, Ivnik RJ, Tangalos EG, Kokmen E. Mild cognitive impairment: Clinical characterization and outcome. Arch Neurol. 1999;56(3):303-8.
Taylor CA, Bouldin ED, McGuire LC. Subjective cognitive decline among adults aged ≥45 years—United States, 2015–2016. MMWR Morb Mortal Wkly Rep. 2018;67(27):753-7.
Thijssen EH, La Joie R, Wolf A, et al. Diagnostic value of plasma phosphorylated tau181 in Alzheimer’s disease and frontotemporal lobar degeneration. Nat Med. 2020;26(3):387-97.
United States Census Bureau. 2017 national projection census tables: Main series. census.gov/data/tables/2017/demo/popproj/2017-summary-tables.html
Walker MP, Ayre GA, Cummings JL, et al. The clinician assessment of fluctuation and the one day fluctuation assessment scale. Two methods to assess fluctuating confusion in dementia. Br J Psychiatry. 2000;177:252-6
Zucchella C, Sinforiani E, Tamburin S, et al. The multidisciplinary approach to Alzheimer’s disease and dementia. A narrative review of non-pharmacological treatment. Front Neurol. 2018;9:1058.



















2 Comments.
As a fellow nurse I found this article clear and informative (as expected from nurses). Having been evaluated by neurologists and by psychosocial testing, I have been primarily diagnosed as PTSD and Major Depression. WRONG! This article will be marked with my experienced symptoms and discussed with my physicians.
Thank you for possibly giving me hope for a proper diagnosis, follow-up and a research study.
Very interesting and informative article. Well written to inform without excessive, unnecessary information.