Take a crash course in extracorporeal membrane oxygenation.
Takeaways:
- Extracorporeal membrane oxygenation (ECMO), a form of cardiopulmonary bypass, is a life-saving treatment for many patients, including those with COVID-19.
- Types of ECMO include venoarterial (cannula placed peripherally or centrally in vein) and venovenous (peripheral cannulation).
- Patients attached to ECMO require compassionate care.
Extracorporeal membrane oxygenation (ECMO) is a life-saving treatment for many patients, including those with COVID-19. Here’s what you need to know to better care for these patients.
What is ECMO?
ECMO, a form of partial cardiopulmonary bypass, provides either venoarterial (VA) or venovenous (VV) support. The ECMO circuit pumps venous blood outside of the body to an oxygenator, which removes carbon dioxide and adds oxygen before returning the blood to an artery or vein. VA configurations provide rest for the heart and lungs by returning oxygenated blood under pressure to an artery. VV configurations provide pulmonary support only, returning oxygenated blood to a vein.
How is a patient placed on ECMO?
Several cannulation methods, including peripheral and central, can be used to place a patient on ECMO.
Peripheral cannulation methods include the following:
- Seldinger technique. This technique safely acquires vascular access by puncturing a vessel with a hollow needle and inserting a guidewire into the lumen. The vessel is then serially expanded with large-bore dilators until the lumen has an entry large enough to accommodate the sheath or cannula without excessive force.
- Surgical open cutdown
- Open cutdown Seldinger (semi-Seldinger)
- Open cutdown with end-to-side Dacron graft.
Central cannulation requires a sternotomy or a thoracotomy and typically is used when a patient can’t be weaned off of cardiopulmonary bypass after cardiac surgery. In this case, the cannulas connect directly to the VA ECMO circuit. With central cannulation, a venous cannula is placed in the right atrium, and an arterial cannula in the ascending aorta. The larger diameter cannulas and decreased resistance permit higher flow.
Newer ECMO circuits are smaller and more portable, allowing successful deployment of the procedure in the field or outpatient settings with a properly trained staff. Previous bulky ECMO systems prevented these options.
What are the different types of ECMO?
VA ECMO (See Types of ECMO, A.) extracts deoxygenated blood from a cannula placed peripherally or centrally in a vein, pumps the blood through the circuit with an oxygenator, and returns the oxygenated blood to the arterial system. Typically, the femoral or internal jugular vein is used for the inflow (drainage) cannula, and the femoral, axillary, or carotid artery is used for the outflow (return) cannula.
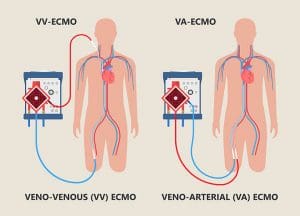

VV ECMO (See Types of ECMO, B.) requires peripheral cannulation. The standard two-cannula approach is achieved with a femoral cannula positioned in the inferior vena cava, which drains deoxygenated blood into the ECMO circuit. A separate short cannula placed in the superior vena cava infuses oxygenated blood from the circuit into the patient. Alternatively, a dual-lumen cannula drains deoxygenated blood from both the superior and inferior vena cava simultaneously and reinfuses oxygenated blood in the right atrium at the level of the tricuspid valve.
More complex cannulation options not discussed in this article are available. In addition, a patient can be placed on ECMO without being intubated and sedated, although it’s relatively uncommon.
How does the ECMO circuit operate?
All ECMO circuits have five major components: inflow (drainage) cannula, pump, oxygenator with a heat exchanger, blender, and outflow (return) cannula. (See ECMO components.)
ECMO components
Extracorporeal membrane oxygenation (ECMO) comprises five components.
- Inflow (drainage) cannula: Drains venous blood into the ECMO circuit.
- Pump: Moves blood through the system.
- Oxygenator: This semipermeable membrane system functions similarly to lungs by exchanging oxygen and carbon dioxide. The surrounding heat exchanger allows the clinician to control the temperature of the blood circulating throughout the body. The oxygenator consists of many low-resistance, hollow, non-microporous fibers that allow for gas exchange across a semipermeable membrane.
- Return cannula: Returns newly oxygenated blood to the systemic circulation.
- Blender: Allows a specific percentage of oxygen vs. room air components (carbon dioxide and nitrogen).
A cannula placed in a vein drains deoxygenated blood from a patient, which is then drawn into the ECMO circuit by the centrifugal-flow pump. The blood passes through a membrane oxygenator that removes carbon dioxide and adds oxygen before delivering the oxygenated blood to a cannula placed in either an artery or a vein.
How does gas exchange occur in the oxygenator?
A semipermeable membrane divides the oxygenator into a blood compartment and a gas compartment. The pump pushes venous blood into the oxygenator, where gas exchange occurs across the membrane as blood interacts with “fresh” gas. The patient’s blood flow rate (liters per minute, or how much blood will come into contact with the oxygenator each minute) through the ECMO circuit is determined by adjusting the centrifugal pump’s rotations per minute.
The blender allows for adjustment of the fraction of delivered oxygen in the sweep gas. Sweep gas rates (liters per minute) are proportionate to blood flow rates. An increase in sweep gas increases carbon dioxide elimination.
Based on diffusion principles, gas exchange occurs across the semipermeable membrane: oxygen infuses into the patient’s blood and carbon dioxide is swept off through the blender. The system returns oxygenated blood to either an artery or a vein, depending on the patient’s ECMO configuration.
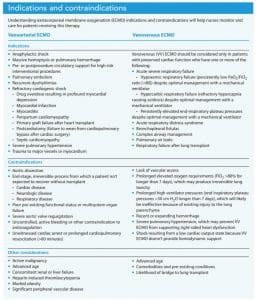
What are indications and contraindications for ECMO?
Indications and contraindications for VA and VV ECMO differ. For example, VV ECMO should only be considered when patients have preserved cardiac function, whereas VA ECMO is indicated in patients experiencing refractory cardiogenic shock (including myocardial infarction and myocarditis). (See Indications and contraindications.)

Can patients on ECMO ambulate?
Patients with certain ECMO configurations can move from the bed to the chair, use equipment such as the seated leg press machine, and ambulate. However, the interprofessional team must first evaluate the patient to determine the clinical appropriateness to engage in rehabilitative therapy. Because ECMO cannulas are wire-reinforced, they’re sturdy.
Some patients on ECMO might be required to demonstrate their strength by performing pre-designated physical activities to increase the likelihood that they’ll be approved for the transplant wait list. Many patients on VV ECMO who are candidates for lung transplantation have a dual-lumen cannula inserted into the internal jugular vein, which serves as the patient’s only cannulation site and enables patient mobility.
How long can a patient be on ECMO?
No absolute maximum or minimum time frame exists for ECMO. Therapy length is an interprofessional decision made on a case-to-case basis; however, according to the Extracorporeal Life Support Organization, the average length for VA ECMO is 5 to 10 days and 10 to 14 days for VV. Some patients may require therapy for more than 30 days. The team should consider the proposed patient outcomes before implementing ECMO. The therapy must be incorporated into a larger clinical plan of care and should serve as a bridge to recovery or long-term treatment such as implantable ventricular assist devices or transplant.
After a patient is placed on ECMO, they should be allowed to experience the therapy’s benefits for at least 24 hours before discussing discontinuation.
What are some of the major complications of ECMO?
ECMO complications include limb ischemia, Harlequin syndrome, hematologic issues, sequestered medications, and absence of pulsatility.
Limb ischemia
Access issues usually occur during cannula placement when ECMO therapy is initiated. Although these issues most frequently occur with arterial cannulas, they also may be encountered with venous cannulas. Clinicians must understand the size of the vessel being cannulated, but emergent placement may prevent careful investigation to guide decisions. As a result, ischemia distal to the large cannulas may cause thrombosis and gangrene. Best practice calls for using a small reperfusion cannula to bypass the vessel area occupied by the ECMO cannula. The nurse must assess all cannulated extremities every hour (more often as needed) to evaluate for signs of ischemia, compartment syndrome, and vascular injury.
Harlequin syndrome
(North–South syndrome)
In patients peripherally cannulated for VA ECMO, oxygenated blood is returned in a retrograde fashion up the aorta. This forms a mixing cloud between blood returned by the ECMO circuit and blood ejected by the left ventricle. Blood from the ECMO circuit is richly oxygenated, but oxygenation of the native circulation depends on pulmonary gas exchange, which frequently is severely impaired. During the early stages of cardiogenic shock, the mixing cloud will occur at the aortic valve because of the patient’s severely impaired native cardiac output. If the left ventricle recovers, the location of the mixing cloud will move distally. If native pulmonary function remains impaired, poorly oxygenated blood will start to perfuse the upper body and coronary arteries. As deoxygenated blood is pushed through the left ventricle, hypoxemia results in the upper parts of the body. Nurses must assess the patient for cyanosis and implement lung optimization strategies, including protective mechanical ventilation and medication as prescribed.
Hematologic issues
Because ECMO involves continuous flow through plastic tubing as well as an oxygenator with a large surface area, many patients experience hematologic issues. For example, clotting can occur in the circuit and may manifest as cerebrovascular, renal, or splenic infarcts. In addition, bleeding can occur at the insertion sites, especially if insertion was traumatic. Anticoagulation can be achieved with short-acting agents such as heparin or direct thrombin inhibitors. Nurses should closely monitor patients for bleeding and clotting and take corrective measures as ordered.
Sequestered medications
Some medications may become sequestered in the circuit if they stick to the tubing. As a result, patients may require higher doses to achieve therapeutic benefits. Nurses should consult with the provider about possible dosage adjustments if the patient doesn’t seem to be responding to medications as expected.
Absence of pulsatility
The continuous flow centrifugal pump and retrograde arterial flow of VA ECMO may increase left ventricular afterload. Retrograde flow impedes distinct opening and closing of the aortic valve. Absence of pulsatility is especially common in patients with severe cardiogenic shock. When monitoring a patient on VA ECMO, nurses may observe an arterial line absent of pulsatility as evidenced by a flat waveform tracing without the presence of a dicrotic notch.
Absent or low pulsatility indicates decreased left ventricular stroke volume, leading to blood stasis and an increased risk of thrombus formation in the ventricle and around the aortic valve. Higher pulsatility indicates potential myocardial recovery during VA ECMO support. Nurses should monitor arterial waveforms for lack of pulsatility as an indication of a closed aortic valve and worsening myocardial function. This knowledge may help guide the team when prescribing I.V. positive inotropes or additional ventricular support along with concomitant intra-aortic balloon pump.
What’s the most important thing to remember when caring for a patient on ECMO?
The interprofessional team must remember that despite the daunting appearance of the ECMO circuit, the patient attached to it requires compassionate care. Never refer to the patient as the device. For example, don’t say, “the ECMO in bed nine.” Rather, remember that the patient in bed nine requires ECMO therapy. Everyone on the team, including nurses, must preserve the patient’s dignity and humanity during an exceptionally vulnerable time.
Rachael Alexis Jividen is a clinical nurse specialist at the Cleveland Clinic Heart, Vascular & Thoracic Institute in Cleveland, Ohio.
References
Asber SR, Shanahan KP, Lussier L, et al. Nursing management of patients requiring acute mechanical circulatory support devices. Crit Care Nurse. 2020;40(1):e1-11. doi:10.4037/ccn2020764
Baran DA. Extracorporeal membrane oxygenation and the critical cardiac patient. Curr Transplant Rep. 2017;4(3):218-25. doi:10.1007/s40472-017-0158-5
Chung M, Shiloh AL, Carlese A. Monitoring of the adult patient on venoarterial extracorporeal membrane oxygenation. ScientificWorldJournal. 2014;393258. doi:10.1155/2014/393258
Han JJ, Swain JD. The perfect ECMO candidate. J Am Coll Cardiol. 2018;71(10):1178-82. doi:10.1016/j.jacc.2018.02.001
Krishnan S. Venovenous extracorporeal membrane oxygenation for lung failure. Consult QD. January 7, 2019.
Pavlushkov E, Berman M, Valchanov K. Cannulation techniques for extracorporeal life support. Ann Transl Med. 2017;5(4):70. doi:10.21037/atm.2016.11.47
Pujara D, Sandoval E, Simpson L, Mallidi HR, Singh SK. The state of the art in extracorporeal membrane oxygenation. Semin Thorac Cardiovasc Surg. 2015;27(1):17-23. doi:10.1053/j.semtcvs.2015.02.004
Su Y, Liu K, Zheng J, et al. Hemodynamic monitoring in patients with venoarterial extracorporeal membrane oxygenation. Ann Transl Med. 2020;8(12):792. doi:10.21037/atm.2020.03.186
Wilson J, Fisher R, Caetano F, et al. Managing Harlequin syndrome in VA-ECMO—Do not forget the right ventricle. Perfusion. 2021;2676591211020895. doi:10.1177/02676591211020895