Keep patients at the center of decision making.
- Describe atrial fibrillation and symptoms associated with it.
- Describe management of atrial fibrillation.
- Describe how nurses can aid early recognition and treatment of atrial fibrillation to prevent adverse outcomes.
The authors and planners of this CNE activity have disclosed no relevant financial relationships with any commercial companies pertaining to this activity. See the last page of the article to learn how to earn CNE credit.
Expiration: 4/1/25
Takeaways:
- Atrial fibrillation is a complex medical diagnosis requiring a holistic and interprofessional approach to minimize symptoms, prevent adverse outcomes, and improve the patient’s quality of life.
- The treatment plan for a patient with atrial fibrillation focuses on thromboembolism and stroke prevention, heart rate control to minimize symptoms, and comorbidity optimization to reduce the duration of atrial fibrillation and improve the patient’s quality of life.
In 2021, between 2.7 and 6.1 million Americans experienced atrial fibrillation, which the European Society of Cardiology defines as a “supraventricular tachyarrhythmia with uncoordinated atrial electrical activation and consequently ineffective atrial contraction.” Underlying conditions (such as hypertension or heart failure) alter the conduction system, which can result in atrial fibrillation.
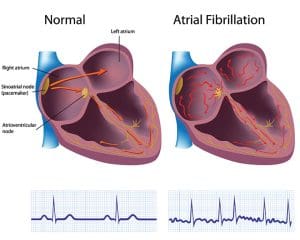
Normally, the sinoatrial (SA) node, located in the right atrium, generates the heart’s electrical impulse, which spreads via the conduction pathway to the right and left atria, causing them to contract. The electrical impulse continues down the conduction pathway past the atrioventricular (AV) node, initiating contraction of the ventricles. This movement of the electrical impulse allows for complete relaxation and contraction of the atria and ventricles to maintain adequate perfusion throughout the body.
In patients with atrial fibrillation, the SA node no longer initiates the electrical impulse. Instead, it originates outside the SA node, generated by the pulmonary veins in the left atrium. As a result, the atria contract in a disorganized fashion, causing them to quiver or fibrillate. When the disorganized signals transmit to the ventricles, they contract irregularly and sometimes quickly. Ineffective atria contraction causes blood to pool in the atria, increasing the patient’s risk for stroke. Ineffective atrial contraction results in a loss of atrial kick, decreasing cardiac output by 20%. Patients may experience syncope, chest pain, or pulmonary edema. (See Normal conduction vs. atrial fibrillation.)
Normal conduction vs. atrial fibrillation
Nurses can aid early recognition and treatment of atrial fibrillation to prevent adverse outcomes.

Atrial fibrillation diagnosis
Patients with atrial fibrillation may experience fatigue, dyspnea, dizziness, palpitations, and chest pain, or they may be asymptomatic. A provider diagnoses atrial fibrillation if a 12-lead ECG shows a narrow QRS complex, irregular R-R intervals, and no discernable P waves. (See Key characteristics.)
Key characteristics
Key characteristics of atrial fibrillation include no discernable P waves, a narrow QRS complex, and irregular R-R intervals.
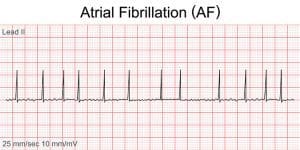

A 12-lead ECG of a patient in normal sinus rhythm will have a P wave before every QRS and regular R-R intervals. Atrial depolarization produces a P wave on a 12-lead ECG. P waves aren’t seen in patients with atrial fibrillation because the SA node no longer initiates the electrical impulse. The lack of one electrical impulse starting at the SA node results in multiple electrical impulses competing for an opportunity to travel through the AV node. Some electrical impulses will reach the AV node, but they will be fast and disorganized, resulting in irregular ventricle contraction. This produces a narrow QRS and an irregular R-R interval.
Based on atrial fibrillation episode duration, the condition is classified into five patterns:
- First diagnosed (new onset)—The patient has no prior history of atrial fibrillation.
- Paroxysmal—Atrial fibrillation terminates spontaneously or with intervention within 7 days of onset.
- Persistent—The episode lasts more than 7 days.
- Long-standing persistent—Atrial fibrillation continues for more than 12 months.
- Permanent—Despite interventions to restore normal sinus rhythm, the condition persists. At this stage, the patient and provider accept that sinus rhythm can’t be maintained and agree that no further attempts will be made to restore it.
Atrial fibrillation management
Atrial fibrillation, a complex medical diagnosis, places a heavy burden on the patient and the healthcare team. It requires a holistic, interprofessional approach to minimize symptoms, prevent adverse outcomes, and improve the patient’s quality of life. The plan of care focuses on comorbidity management to reduce the duration of atrial fibrillation and prevent reoccurrence, heart rate control to minimize symptoms, and thromboembolism and stroke prevention.
Comorbidities
Great strides in atrial fibrillation management have occurred, but its incidence continues to increase due to the growing prevalence of obesity and the population’s advancing age. The development of a single risk factor, such as obesity, increases the risk for developing other modifiable risk factors such as hypertension, obstructive sleep apnea, and diabetes. Lifestyle and risk factor management play an important role in the patient’s plan of care. (See Nonmodifiable risk factors.)
Nonmodifiable risk factors
Nonmodifiable risk factors associated with atrial fibrillation include the following:
Age
Although atrial fibrillation can develop at any age, it occurs most frequently in those 55 years and older.
Genetics
Patients with a family history of atrial fibrillation have a 40% increased risk for developing the condition.
Ethnicity
Individuals of European descent are most likely to develop atrial fibrillation. According to the European Society of Cardiology, the lifetime risk for this population is 1 in 3 individuals.
Gender
Both men and women are at risk for developing atrial fibrillation; however, the prevalence is higher in men. Women are more likely to experience complications of atrial fibrillation.
Obesity
Obesity (body mass index ≥30 kg/m2) increases the likelihood of developing atrial fibrillation as well as other risk factors. Physical inactivity, frequently a precursor to obesity, triggers systemic inflammation and elevated sympathetic tone, which then trigger structural and electrical changes in the heart. Over time, the left atrium and left ventricle enlarge, forcing the heart to work harder. As a result, patients may experience atrial fibrillation in addition to other complications such as hypertension and heart failure.
Encourage patients with obesity to develop a weight loss plan. According to Chung and colleagues, the American Heart Association recommends a 10% reduction in weight to decrease the incidence of atrial fibrillation. Sustaining weight loss is highly associated with maintaining normal sinus rhythm and minimizing the symptom burden associated with other risk factors. Fluctuations in losing and gaining weight are associated with recurrent atrial fibrillation episodes. To help achieve and maintain weight loss, encourage patients to participate in 150 minutes per week of moderate-intensity exercise or 75 minutes per week of vigorous-intensity aerobic exercise. Explain that participation in regular physical activity may help improve quality of life and minimize atrial fibrillation symptoms.
Hypertension
Hypertension (systolic blood pressure ≥140 mmHg and/or diastolic blood pressure ≥90 mmHg) is linked to obesity and diabetes, as well as unhealthy lifestyle habits that increase a patient’s risk for atrial fibrillation.
If the patient has hypertension and atrial fibrillation, or has not yet been diagnosed with atrial fibrillation but has hypertension in addition to other risk factors, teach the patient about limiting salt intake, choosing fresh fruits and vegetables over fast or processed food, and consuming caffeine and alcohol in moderation. Encourage them to participate in regular physical activity, including aerobic exercise and strength training 2 to 3 days per week, and encourage them to minimize stress.
These lifestyle modifications help decrease the risk of developing atrial fibrillation and minimize the burden of the condition. In addition to providing patient education, collaborate with the provider to establish a blood pressure goal for the patient and administer medications as ordered to achieve and maintain that goal.
Obstructive sleep apnea
Obstructive sleep apnea and atrial fibrillation are more prevalent in men than women and in patients with advanced age, hypertension, obesity, and heart failure. Although obstructive sleep apnea and atrial fibrillation frequently are associated with one another solely because of their shared risk factors, data support decreased incidence of atrial fibrillation in patients with obstructive sleep apnea who receive continuous positive airway pressure (CPAP) treatment.
Assess the patient for a history of obstructive sleep apnea. If they’ve never been diagnosed with the condition, ask if they have symptoms, such as excessive daytime sleepiness. Notify the provider if the patient reports a history or current symptoms of obstructive sleep apnea. Treatment with CPAP in patients with atrial fibrillation helps achieve rhythm control. Educate the patient about lifestyle modifications, such as participating in regular physical activity and eating fresh fruits and vegetables, to minimize obstructive sleep apnea and atrial fibrillation symptoms.
Diabetes
Diabetes is an independent risk factor for atrial fibrillation, which is more prevalent in patients with long-standing diabetes and poor glycemic control. Glucose intolerance and insulin resistance contribute to structural and electrical changes within the heart, contributing to the development of atrial fibrillation.
Educate the patient about diabetes management and lifestyle modifications. Encourage the patient to follow their recommended nutrition plan to promote adequate glycemic control and body weight goal maintenance, and promote physical activity and smoking cessation. As ordered by the provider, monitor the patient’s blood glucose levels and administer insulin or other oral medications. Achieving blood sugar control may help minimize the risk and burden of atrial fibrillation.
Smoking
Smoking tobacco is associated with increased risk for atrial fibrillation and the inability to maintain normal sinus rhythm after ablation. Smoking triggers vasoconstriction within the coronary arteries, which leads to increased workload of the heart. As the heart works harder, the muscle will grow and adverse events such as hypertension, heart failure, and atrial fibrillation will result. Provide the patient with smoking cessation information and referrals.
Alcohol consumption
Alcohol intake may trigger atrial fibrillation and decrease the efficacy of pharmacologic or procedural treatment for atrial fibrillation. Encourage male patients with atrial fibrillation to consume no more than two alcoholic drinks per day; female patients should consume no more than one-and-a-half drinks per day.
Caffeine intake
Some data show that caffeine may induce palpitations, a symptom of atrial fibrillation, while other research indicates that caffeine may reduce the incidence of atrial fibrillation. Insufficient data exist to support reducing caffeine intake to prevent the condition. Encourage patients to consume caffeine in moderation, and notify the provider if palpitations become more severe.
Surgery
Postoperative atrial fibrillation occurs in 20% to 50% of patients after cardiac surgery. Most episodes are self-terminating, but postoperative atrial fibrillation can lead to persistent atrial fibrillation. To prevent postoperative atrial fibrillation, monitor the patient’s electrolytes and oxygenation status. Assess the patient’s level of pain and provide pharmacologic and nonpharmacologic interventions, such as repositioning and dimming the lights to create a calming environment. Monitor the patient’s heart rate and rhythm and administer beta-blockers as ordered.
Heart rate and rhythm control to minimize symptoms
Identifying and managing risk factors serves as only one component of the plan of care for a patient with atrial fibrillation. Another element consists of controlling symptoms with rate or rhythm control. Before initiating therapy, ask the patient about their goals and preferences. Rhythm or rate control strives to alleviate symptoms and prevent complications, allowing the patient to reach their desired quality of life.
Rate control
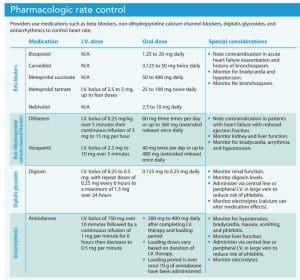
Rate control uses pharmacologic interventions to achieve a target heart rate. Most providers aim for a sustained heart rate less than 110 beats per minute. The medications of choice include beta blockers, non-dihydropyridine calcium channel blockers, digitalis glycosides (digoxin), and antiarrhythmics (amiodarone). The provider selects medications after careful evaluation of the patient’s symptoms, ejection fraction, and hemodynamics.
Beta blockers provide first-line therapy for patients with no comorbidities, hypertension, heart failure with preserved ejection fraction (HFpEF), or heart failure with reduced ejection fraction (HFrEF) but are contraindicated in patients with acute heart failure exacerbation and a history of bronchospasm. In an acute setting, beta blockers’ rapid onset makes them preferrable to digoxin. Beta blockers can be combined with digoxin or amiodarone if the patient’s resting heart rate stays above 110 beats per minute or the patient experiences worsening symptoms.
Non-dihydropyridine calcium channel blockers (diltiazem or verapamil) have a rapid onset and are the treatment of choice for patients with chronic obstructive pulmonary disease and asthma. They also can be used as first-line treatment for patients with hypertension or HFpEF. Both diltiazem and verapamil are contraindicated in patients with HFrEF. They can be combined with digoxin if one alone does not achieve a heart rate less than 110 beats per minute or the patient experiences worsening symptoms.
Digoxin is used in lower doses or in combination with beta blockers, diltiazem, or verapamil to achieve rate control. It’s ineffective in patients with increased sympathetic drive and has been associated with mortality in patients with atrial fibrillation.
Amiodarone is used in combination with beta blockers and digoxin to achieve rate control in patients with HFrEF.
Rhythm control
Rate control frequently is sufficient to alleviate symptoms of atrial fibrillation; however, comorbidities and persistent symptoms may require additional therapy. Rhythm control, which is indicated for patients with symptomatic atrial fibrillation and a poor quality of life, aims to restore and maintain normal sinus rhythm and alleviate symptoms. Rhythm control incorporates rate control in combination with electrical and pharmacologic cardioversion and catheter ablation, which are indicated in emergent and non-emergent situations to restore normal sinus rhythm.
Before cardioversion, therapeutic anticoagulation must be achieved. If that’s not possible, the provider will order a transesophageal echocardiogram to assess for atrial thrombi. Pharmacologic and electrical cardioversion are contraindicated in patients with known atrial thrombus.
Electrical cardioversion involves placing electrode pads in the anterior–posterior position and delivering a synchronized electrical shock. Patients undergoing an electrical cardioversion are sedated with I.V. midazolam and propofol or etomidate. Before the procedure, confirm patent I.V. access and that the patient has had nothing by mouth for the previous 6 hours, as recommended by the Association of periOperative Registered Nurses, and monitor the patient’s blood pressure, heart rate, and oxygenation status during and after the procedure. Burns may occur on the patient’s skin after the procedure. Perform skin assessments and provide interventions to alleviate discomfort.
Pharmacologic cardioversion using antiarrhythmic medications such as amiodarone, flecainide, propafenone, dronedarone, or sotalol, is most effective in patients who have developed atrial fibrillation within the preceding 7 days. During pharmacologic cardioversion, monitor the patient’s heart rate, blood pressure, and potassium levels, and perform 12-lead ECGs to assess for QT prolongation. Electrical and pharmacologic cardioversion are contraindicated in patients with digitalis toxicity, multifocal atrial tachycardia, and suboptimal anticoagulation.
Catheter ablation is indicated for patients with symptomatic persistent or paroxysmal atrial fibrillation despite pharmacologic attempts to restore normal sinus rhythm. The procedure involves placing a catheter into the heart via the femoral vein. The catheter releases radiofrequency energy, targeting the pulmonary veins in the left atrium, to produce areas of small thermal myocardial injury that ablates the abnormal electrical current. Catheter ablation is the best treatment option for patients who can’t tolerate antiarrhythmic medications; however, it’s not always successful. Approximately 20% of patients who undergo catheter ablation will experience atrial fibrillation after the procedure, requiring another ablation.
As an alternative to catheter ablation, surgical ablation (maze procedure) uses a sternotomy or mini thoracotomy approach to restore normal sinus rhythm by creating scar tissue via cryothermy or radiofrequency energy to block the abnormal electrical impulses that cause atrial fibrillation. Surgical ablation is indicated for patients with symptomatic drug-refractory atrial fibrillation and a failed catheter ablation in the past or for patients undergoing surgery for other structural diseases such as mitral valve disease.
Thromboembolism and stroke prevention
Patients with atrial fibrillation are at high risk for thromboembolism and stroke. Anticoagulation is an essential therapy for all patients with atrial fibrillation unless it’s contraindicated (active bleeding, severe thrombocytopenia, intracranial hemorrhage) or the patient declines treatment.
Before initiating therapy, the provider calculates the patient’s risk for stroke using the CHA2DS2-VASc scoring system and discusses risks and benefits of treatment with the patient. The system provides a score based on the presence of heart failure, hypertension, age between 65 and 74 years or 75 years or older, diabetes, stroke, vascular disease, and gender. Anticoagulation is recommended for patients with a score of at least 2 who have no contraindications.
Patients may be reluctant to accept anticoagulation therapy because of the required frequent blood draws and lifestyle modifications. However, non-vitamin K antagonist oral anticoagulants (apixaban, dabigatran, edoxaban, and rivaroxaban), the first-line therapy for oral anticoagulation, don’t require frequent blood draws. Warfarin, a vitamin K antagonist, is the preferred oral anticoagulant for patients with atrial fibrillation and prosthetic mechanical heart valves. The therapeutic level of warfarin requires regular monitoring using the international normalized ratio (INR), which can be modified by consuming foods containing vitamin K.
Educate patients about the importance of consistent vitamin K intake and not binging on foods (including, broccoli, Brussel’s sprouts, and green leafy vegetables such as spinach and kale) that contain it. Regardless of the anticoagulant prescribed, teach all patients about bleeding risk. Instruct them to use an electric razor and a soft toothbrush to minimize bleeding, and encourage them to take extra precautions to maintain safety, prevent falls, and remain free of injury.
In patients who can’t tolerate long-term anticoagulation, left atrial appendage obliteration may be implemented for stroke prevention. The left atrial appendage, the primary source of thrombus formation in the left atrium, can be surgically removed during other cardiac procedures (such as cardiac surgery) or occluded using a percutaneously inserted device to prevent thrombus formation.
Complications


As symptoms worsen and complications develop, the patient may begin to experience decreased quality of life because they can’t participate in the activities they enjoy. About 16% to 20% of patients with atrial fibrillation will experience depression due to severe symptoms, decreased quality of life, and medication side effects.
Patient-centered care
Atrial fibrillation is a complex condition associated with a large symptom burden and complications. Although prompt recognition and treatment are crucial, the patient must remain at the center of decision making. Involving the patient in the plan-of-care discussion and providing them with appropriate educational materials may increase their willingness to make lifestyle modifications and adhere to the prescribed treatment plan. By addressing comorbidities and providing heart rate and rhythm control and anticoagulation therapy, patients with atrial fibrillation can achieve an optimal quality of life.
Kayla Little is a clinical nurse specialist in the cardiovascular medicine step-down, vascular surgery step-down, and heart and lung transplant step-down units at Cleveland Clinic in Cleveland, Ohio.
References
Adiyaman A, Buist TJ, Beukema RJ, et al. Randomized controlled trial of surgical versus catheter ablation for paroxysmal and early persistent atrial fibrillation. Circ Arrythm Electrophysiol. 2018;11(10):e006182. doi: 10.1161/CIRCEP.118.006182
American Diabetes Association. 5. Lifestyle management: Standards of Medical Care in Diabetes – 2019. Diabetes Care. 2019;42(suppl1):S46-60. doi: 10.2337/dc19-S005
American Heart Association. Get with the Guidelines® AFib.
Association of periOperative Registered Nurses. Procedures at a glance: Cardioversion.
Centers for Disease Control and Prevention. Atrial fibrillation. September 27, 2021.
Centers for Disease Control and Prevention. Stroke: Signs and symptoms. August 28, 2020.
Chung MK, Eckhardt LL, Chen LY, et al. Lifestyle and risk factor modification for reduction of atrial fibrillation: A scientific statement from the American Heart Association. Circulation. 2020;141(16):e750-72. doi: 10.1161/CIR.0000000000000748
Collado FMS, Lama von Buchwald CM, Anderson CK, et al. Left atrial appendage occlusion for stroke prevention in nonvalvular atrial fibrillation. J Am Heart Assoc. 2021;10(21):e022274. doi: 10.1161/JAHA.121.022274
Huang H, Wang Q, Xu J, Wu Y, Xu C. Comparison of catheter and surgical ablation of atrial fibrillation: A systematic review and meta-analysis of randomized trials. J Thorac Cardiovasc Surg. 2020;S0022-5223(20). doi: 10.1016/j.jtcvs.2020.04.154
Gutierrez C, Blanchard DG. Diagnosis and treatment of atrial fibrillation. Am Fam Physician. 2016;94(6):442-52.
Hindricks G, Potpara T, Dagres N, et al. 2020 ESC guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS): The Task Force for the diagnosis and management of atrial fibrillation of the European Society of Cardiology (ESC) Developed with the special contribution of the European Heart Rhythm Association (EHRA) of the ESC. Eur Heart J. 2021;42(5):373-498. doi: 10.1093/eurheartj/ehaa612
January CT, Wann LS, Calkins H, et al. 2019 AHA/ACC/HRS focused update of the 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: A report of the American College of Cardiology/American Heart Association task force on clinical practice guidelines and the heart rhythm society in collaboration with the Society of Thoracic Surgeons. Circulation. 2019;140(2):e125-51. doi: 10.1161/CIR.0000000000000665
Staerk L, Sherer JA, Ko D, Benjamin EJ, Helm RH. Atrial fibrillation: Epidemiology, pathophysiology, and clinical outcomes. Circ Res. 2017;120(9):1501-17. doi: 10.1161/CIRCRESAHA.117.309732
Unger T, Borghi C, Charchar F, et al. 2020 International Society of Hypertension global hypertension practice guidelines. Hypertension. 2020;75(6):1334-57. doi: 10.1161/HYPERTENSIONAHA.120.15026.

















3 Comments.
I am a nursing professor, I plan on using it for my students! It is an easy informative read. Thanks and well done!
article informative and nice review for cardiac
Well done article, very informative