It’s lunchtime. Three of your patients are scheduled to receive rapid-acting insulin in addition to sliding-scale insulin.
- Mr. Jones, age 87, has type 2 diabetes. His blood glucose level is 223 mg/dL. He’s on a clear diet.
- Mrs. Smith, age 63, has type 1 diabetes, a serum creatinine level of 1.6 mg/dL, an inconsistent appetite, and widely varying blood glucose levels. Her current blood glucose level is 105 mg/dL.
- Mr. Brown, age 58, has pneumonia, type 2 diabetes, and obesity; he’s receiving corticosteroids. He eats everything on his tray and asks for snacks. His lunchtime blood glucose level is 152 mg/dL. By the time you’re able to administer his insulin, he has eaten half his lunch tray.
Which patient should receive insulin as scheduled? Should any of them not receive it? Should any receive scheduled insulin plus the sliding-scale dose? What should you do if one of them has a normal blood glucose level? Are any at risk for hypoglycemia? What could happen if they eat before you can administer insulin?
These are questions you might ask yourself every day but rarely have the time or resources to get the answer. Yet to make safe clinical decisions, you need the required knowledge base, because insulin is strongly linked to medication errors and adverse drug events (ADEs). The American Hospital Association deems coordination of meals and insulin a top priority for reducing in-hospital ADEs.
The variety of insulin types and their wide-ranging pharmacokinetic properties further complicate insulin use in the hospital. What’s more, much variation exists in the insulin regimens used to meet blood glucose goals recommended by regulatory bodies and professional organizations, such as The Joint Commission and the American Diabetes Association.
Basal-prandial insulin therapy
One insulin regimen involves basal-prandial insulin therapy. This therapy became popular after publication of the RABBIT 2 trial in 2007, which compared stand-alone, sliding-scale insulin therapy with a basal-prandial insulin regimen. It found that the latter decreased mortality and complications without significantly increasing hypoglycemia occurrences.
Basal-prandial insulin therapy has three components:
- long-acting insulin given once or twice daily
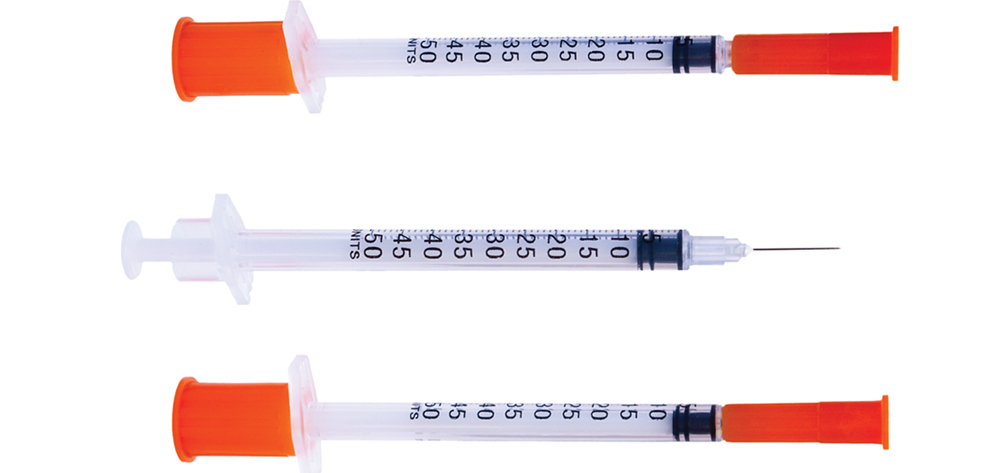
- rapid-acting insulin given in prescribed doses with meals
- correction insulin given with meals and at bedtime. (See Comparing short- and rapid-acting insulin.)
Basal-prandial regimens may involve more insulin than you’re used to giving with traditional sliding-scale and stand-alone regimens. This may make you hesitate, particularly at mealtimes, when you may be giving higher doses of rapid-acting insulin. A clear understanding of the pharmacologic principles of basal-prandial insulin and how it’s prescribed will boost your confidence.
Unlike traditional sliding-scale regimens, which are reactive, basal-prandial regimens address the patient’s insulin requirements proactively. With these regimens, dosages are calculated based on the patient’s weight and estimated insulin sensitivity. Defined as the patient’s expected response to 1 unit of insulin, insulin sensitivity can vary widely among patients and depends on several factors. For instance, patients with renal failure, advanced age, and type 1 diabetes tend to be more insulin-sensitive. In contrast, those with obesity, type 2 diabetes, or infections and those receiving steroids tend to be more insulin-resistant.
Once the patient’s insulin sensitivity is determined, a sensitivity factor is selected and multiplied by the patient’s weight in kg; the result is the total daily dosage of insulin. Half of the total dosage is given as basal insulin and the remainder is divided by three and given with meals. (See Calculating basal-prandial insulin.)
Each part of the basal-bolus regimen serves a specific purpose:
- Long-acting insulin meets basal insulin requirements and is designed to match the liver’s continuous glucose output.
- The prandial rapid-acting insulin component covers the carbohydrate bolus that the patient eats at each meal.
- The correction coverage addresses blood glucose levels outside the target range and is dosed on a sliding scale based on blood glucose levels. (See Generic correctional insulin scale.)
Mealtime insulin and food intake
Mealtime boluses of rapid-acting insulin should be given with 30 to 60 g of carbohydrates. But few patients count carbohydrates in the hospital. So how do you know how much carbohydrate a patient consumes? Typically, hospital patients on a diabetic diet receive 1,800 calories per day. On an 1,800-calorie diet tray, the carbohydrate portion of one meal is about 60 to 75 g. That means the patient must eat about 50% of the tray consistently to receive the prescribed prandial boluses. A patient like Mrs. Smith, with an inconsistent appetite, normal blood glucose level, and poor renal function, needs to be evaluated at each meal to determine how much insulin to give. If she eats a full meal, you may administer a full prandial dose; if she eats less than 50% of her meal, call the physician for clarification. Ideally, patients like her should have standing orders on how to proceed when they eat less than 50% of a meal (if such orders aren’t part of the facility’s basal-bolus order set). You might suggest that the physician address the variable prandial doses by writing a standing order to cover future meals so you don’t have to call him or her.
If you administer insulin to patients receiving basal-prandial insulin, consider the type of diet they’re on. Here are some examples:
- Patients receiving nothing by mouth (NPO) shouldn’t receive prandial boluses.
- Those on clear liquid diets don’t consume enough carbohydrate to warrant prandial insulin administration. Typically, they’re on these diets to rest the gut therapeutically—for instance, because of a poor appetite or nutritional absorption problems.
If your patient is NPO or on a clear liquid diet and has an order for prandial insulin boluses, clarify the order with the attending physician. Take, for instance, Mr. Jones—the 87-year-old on a clear liquid diet whose blood glucose level is 223 mg/dL. He needs insulin to reduce his blood glucose to a normal level to avoid further hyperglycemia, but not so much insulin that hypoglycemia occurs. So you need to withhold prandial insulin. Call the attending physician to clarify the correctional insulin dose.
Mealtime blood glucose levels and insulin administration
Although you’ll need to assess nutritional intake at each meal for mealtime boluses, you should give correctional insulin as indicated regardless of diet type, appetite, and overall intake. Correctional insulin aims to correct the blood glucose level based on the premeal glucose level. Ideally, measure blood glucose as close to mealtime and insulin administration as possible. This helps ensure that the insulin dose you give is appropriate for the patient’s current blood glucose level to prevent over- or underdosing, which could lead to hyper- or hypoglycemia.
You may be concerned (legitimately so) about giving insulin when a patient is NPO. Many hospitals have adopted NPO correction scales. Typically, these scales provide reduced insulin coverage and begin covering blood glucose at a much higher level. This level depends on target blood glucose goals set by the hospital. If your hospital doesn’t have an NPO sliding scale, review the patient’s blood glucose levels with the attending physician to determine if he or she should receive insulin while NPO.
Although you may feel comfortable giving insulin when the patient’s blood glucose level is elevated, you may have concerns about giving scheduled insulin doses when the glucose level is normal. Remember—the purpose of prandial insulin is to cover the carbohydrate consumed in a meal, so you should give prandial insulin boluses even if the blood glucose level is 70 to 140 mg/dL, as with Mrs. Smith. Because she has type 1 diabetes and doesn’t produce insulin, she must receive exogenous insulin even when her blood glucose level is normal to avoid diabetic ketoacidosis. But if a patient has a blood glucose level below 70 mg/dL, treat the blood glucose according to your hospital’s hypoglycemia protocol and notify the attending physician. As part of the hypoglycemia notification process, review all premeal blood glucose levels and scheduled prandial insulin doses.
Coordinating meals and insulin
Coordinating insulin administration with meals can be a daunting task. Mealtimes are often the busiest times of a nurse’s day. You may have other medications to give and other tasks to do. But timing insulin administration with the first bite of food can reduce the risk of periprandial hypoglycemia and subsequent blood glucose variability. To avoid variability, administer mealtime boluses within 15 minutes before or after the first bite.
Prandial insulin doses are given as rapid-acting insulin. To understand the rationale for the administration times, you must be familiar with the pharmacokinetics of rapid-acting insulin. It’s an analog insulin, meaning it’s chemically engineered to be absorbed more rapidly in the subcutaneous tissue and behave more like endogenous insulin than regular insulin. When we eat, our bodies begin producing insulin within 5 to 15 minutes of the first bite. Within 1 to 2 hours, endogenous insulin and postprandial glucose reach peak concentrations; within 3 to 4 hours, they return to baseline. Similarly, rapid-acting insulin has an onset of 5 to 15 minutes, a peak time of 1 to 2 hours, and a duration of 3 to 4 hours. You must give it within 5 to 15 minutes of the first bite to match the peak postprandial blood glucose level.
An advantage of rapid-acting insulin over regular insulin as a mealtime insulin is that it can be given before or after the first bite. This offers some scheduling flexibility and the ability to assess how much the patient eats before giving insulin. In Mr. Brown’s case, he has eaten part of his meal before you arrive with his insulin dose. Some nurses may be tempted to withhold his insulin for fear of inducing hypoglycemia, but withholding this dose would put Mr. Brown in danger of hyperglycemia. Rapid-acting insulin analogs can be given safely up to 15 minutes after the first bite, avoiding hypoglycemia.
You may not always know how a patient will respond to a given insulin dose, as with patients who have poor renal function or complex diabetes states (“brittle” diabetes). This can be challenging at mealtimes, when many factors determine patient response, including the insulin type, purpose of insulin, current blood glucose level, disease state, renal function, and nutritional status.
Answers to the questions you may have about giving insulin at mealtimes may not always be straightforward. Mrs. Smith, for instance, has a long history of type 1 diabetes and a serum creatinine level of 1.6 mg/dL. Because of her poor nutritional status, impaired renal function, and diabetes state, her blood glucose response to insulin is less predictable. She needs close evaluation for each mealtime insulin dose. If you think a dose may need to be omitted or changed, consider all relevant factors to determine the proper course of action, and make recommendations to the attending physician.
Don’t take insulin therapy for granted
Some nurses may take insulin administration for granted because they perform it every day. But oversimplifying this task can put patients at risk for adverse outcomes, such as hyper- or hypoglycemia. Insulin administration involves a complex decision-making process, and clinicians need to collect and evaluate a great deal of data to reduce the risk of adverse outcomes. By considering all relevant patient data, you can reduce the likelihood of an insulin-related adverse outcome.
Selected references
American Diabetes Association. Standards of medical care in diabetes—2013. Diabetes Care. 2013;36 Suppl 1:S11-66.
Cobry E, McFann K, Messer L, et al. Timing of meal insulin boluses to achieve optimal postprandial glycemic control in patients with type 1 diabetes. Diabetes Technol Ther. 2010;12(3):173-7.
Freeland B, Penprase BB, Anthony M. Nursing practice patterns: timing of insulin administration and glucose monitoring in the hospital. Diabetes Educ. 2011;37(3):357-62.
Freeman JS. Insulin analog therapy: improving the match with physiologic insulin secretion. J Am Osteopath Assoc. 2009:109(1):26-36.
Umpierrez GE, Smiley D, Zisman A, et al. Randomized study of basal-bolus insulin therapy in the inpatient management of patients with type 2 diabetes (RABBIT 2 Trial). Diabetes Care. 2007;30(9):2181-6.
Julie S. Lampe is a diabetes clinical nurse specialist at the Orlando Regional Medical Center, Orlando Health, in Orlando, Florida. (Names in scenarios are fictitious.)