A facility’s innovative program effectively reduced patient injury.
Takeaways:
- Learn how a nurse-led interprofessional innovation was designed at an organization that’s received Magnet® designation.
- Gain insight how use of empowerment structures by clinical bedside staff can reap sustained improvements in patient safety outcomes.
As patient care equipment and technology evolve, medical device-related hospital-acquired pressure injuries continue to capture national healthcare attention. These frequently preventable injuries, which develop during the application of a diagnostic or therapeutic device during hospitalization, can result in a stageable pressure injury, even deep tissue and mucosal. Injuries can develop anywhere on the patient’s body and depend on the device application. They’re frequently identified when the injury shape matches the device. (See Injury statistics.)
Injury statistics
Medical-device related hospital-acquired pressure injuries contribute to the overall pressure injury burden on the U.S. healthcare system.
- Black and colleagues (2010) reported that the use of each medical device amplifies the pressure injury risk by 2.4 times.
- According to the Agency for Healthcare Research and Quality, pressure injuries result in 60,000 deaths and up to $11.6 billion annually.
- According to an integrative review by Cavalcanti and Kamada, medical device pressure injury incidence in acute care ranges from 10.95% to 47% for adult populations.
- Baharestani and Quillen reported injury incidence as high as 52% in pediatric patients.
Some evidence-based interventions for preventing medical device injuries have been published; however, a practical and scientifically evaluated clinical protocol doesn’t yet exist. This lack of guidance poses a challenge for healthcare teams when developing comprehensive, individualized preventive care plans involving medical devices. Nurses must rely on innovative solutions endorsed by legacy practices or a combination of diverse published evidence. Healthcare organizations that have achieved American Nurses Credentialing Center (ANCC) Magnet® designation are well positioned to address these issues because of their cultures of exemplary professional practice (EPP), which foster critical thinking, problem-solving, and creative solutions.
This article profiles how one organization that’s received Magnet designation designed an innovative nurse-led and nurse-driven interprofessional protocol that resulted in injury reductions over 4 years.
Establishing a data-based patient safety problem
Baptist Health Lexington, a 434-licensed bed community hospital in central Kentucky, has achieved Magnet designation four times. The organization’s culture of nursing demonstrates EPP by its commitment to autonomous practice and sustained excellence in reported outcomes facilitated by the professional practice model (PPM). Since 2004, the two-tier shared governance empowerment model has been cultivating nurse engagement at all levels to address daily practice and patient- and family-centered care issues.
In early 2017, nurses from the wound, ostomy and continence (WOC) department and a doctorally prepared Magnet program coordinator and the nurse leader for the pressure injury program unveiled an opportunity to improve care related to medical devices. When reviewing the hospital-acquired pressure injury data for October–December 2016, the program coordinator identified an unusual escalation in the monthly incidence across all medical-surgical/telemetry (MST) units and intensive care units (ICU) among adult patients. During daily unit rounds, the coordinator presented the data and collaborated with four WOC nurses to review and categorize injuries by type and care setting. Data analysis revealed nearly 50% of all reported injuries involved a medical device.
Alarmed by the analysis, the coordinator and one of the WOC nurses conducted a critical literature review to explore the national incidence and availability of prevention measures. Findings from the review inspired an innovative proposal to improve the care delivery system using a high-level shared governance approach through the PPM.
Designing the innovation
From mid-to-late January 2017, the Magnet program coordinator guided the unit-level nursing workgroup in the design phase of their care innovation by selecting a few recently published findings with the strongest level of evidence. Through participation in the National Pressure Injury Advisory Panel (NPIAP), WOC nurses in the workgroup accessed and reviewed two best practices for preventing medical device pressure injuries on MST units and ICUs and incorporated evidence from a previously published institutional study endorsing a nurse-driven protocol for urinary catheters. During a series of discussions, consensus supported creating a two-part, four-item protocol. Using Watson’s theory of caring, nurses named the protocol C.A.R.E to help increase translation efficiency and keep it top-of-mind. The C.A.R.E. framework outlines four key nursing and interdisciplinary actions when implementing preventive care for patients with a medical device:
- Choose/verify size-appropriate devices and place on skin free from alterations in integrity
- Assess skin under the medical device during focused rounds
- Reposition and Reapply using protective padding
- Empowered to evaluate daily discontinuation.
The workgroup presented a draft of the innovation to several organization-level councils and committees to solicit feedback from key stakeholders and finalize the protocol. The nurses introduced a preview of the protocol during a monthly Skin, Wound & Assessment Team (SWAT) Force meeting. SWAT Force is an organization-wide taskforce of 36 clinical nurses and interprofessional care providers who serve as unit-level resources for assessing and preventing pressure injuries. In addition to conducting quarterly prevalence studies, the taskforce reviews pressure injury data and provides guidance regarding care changes.
After review and discussion, SWAT Force members unanimously agreed to adopt the C.A.R.E. protocol and forwarded the proposal to the nursing leadership council for feedback. The council, which includes inpatient and ambulatory nurses, autonomously drives improvements and provides formal recognition in professional nursing practice based on strategic priorities.
Participants from the taskforce and the leadership council provided recommendations for simplifying and finalizing the two-part protocol. Part I promotes clinical autonomy and standardized implementation of the four C.A.R.E. actions based on each patient’s total Braden Scale score. A score >18 triggers C.A.R.E. protocol implementation at least every 12 hours on MST units and every 4 hours in the ICU. A score ≤18 indicates implementation at least every 4 hours on MST units and every 2 hours in the ICU.
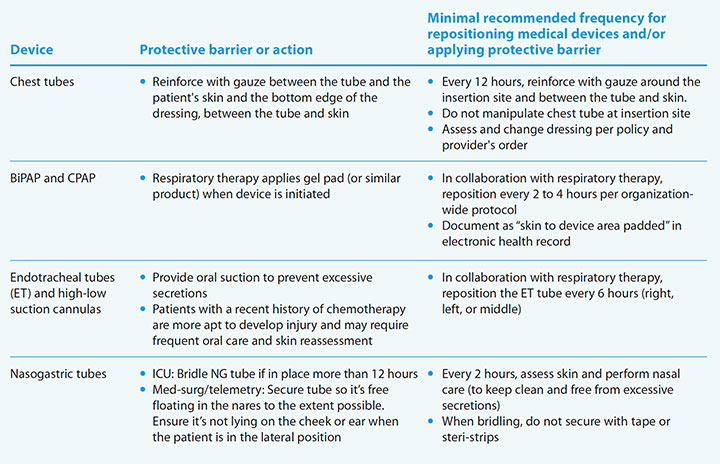
Part II, a 20-item list of high- and low-volume medical devices used across all MST units and ICUs, identifies the recommended minimum skin assessment frequency, protective barrier application, and documentation. (See Protection and assessment.)
Protection and assessment
Part II of the Baptist Health Lexington C.A.R.E. protocol provides protection guidance for specific devices. Below are examples of four devices and associated actions. Access the complete list at myamericannurse.com/?p=112429 .

Implementing the innovation
From March to May 2017, WOC nurses and the Magnet program coordinator collaborated with SWAT Force and the Magnet Nurse Champions (a nurse-led shared-governance council) to launch this innovation education. Over 3 months, clinical nurse super-users used a blended-learning methodology involving an online learning management system to provide an interactive 30-minute professional development activity for unit peers on qualifying inpatient units. Magnet Nurse Champions and SWAT Force members reinforced the activity with a four-page educational flyer during scheduled shifts and staff meetings. The flyer defined the four key actions illustrated by C.A.R.E., identified low- and high-volume medical devices used across patient care units, recommended timeframes for assessment and reassessment, and suggested protective barriers for each device. To validate competency, nurses completed a 20-item knowledge assessment administered via an online learning management system. Full protocol implementation began in late May 2017.
Measuring outcomes
Incidence and prevalence outcome measures were chosen to evaluate the protocol’s effectiveness. The mean monthly medical device pressure injury incidence decreased from 1.23 per 1,000 patient days (October–December 2016) to 0.20 (June 2017–July 2020), reflecting an overall 84% reduction over 4 years. Postintervention, 16 of 38 months reported no injuries across all MST units and ICUs with none reported in the past 7 months (January–July 2020). Quarterly prevalence studies revealed exemplary outcomes. Most qualifying inpatient units (n = 15) outperformed the national benchmark for 11 quarters postintervention, with eight quarters reflecting zero injuries.
C.A.R.E. innovation impact
The graphs illustrate the positive outcomes associated with Baptist Health Lexington’s C.A.R.E. protocol to prevent medical-device related pressure injuries.
Recognizing nurses, disseminating outcomes
The C.A.R.E. protocol received recognition at the local, state, and international levels. Organization administration recognized WOC nurses and the Magnet program coordinator via emails distributed organization-wide and during shared governance council meetings. In addition, the administration allocated money for formal presentations at local and national conferences. In December 2019, the Magnet Recognition Program distinguished the nursing workgroup with two exemplars validated during an onsite survey: nurses using the PPM to promote patient care outcome improvements and outperformance of a nationally benchmarked, nurse-sensitive indicator over eight quarters. In July 2020, WOC nurses and the Magnet program coordinator disseminated outcomes in a poster presentation during a system-level, virtual forum highlighting safety and quality innovations. Over the past 3 years, C.A.R.E. received formal international recognition as the workgroup shared the protocol in seven poster and podium presentations during the annual ANCC Pathway to Excellence® (2018–2021), Magnet (2019–2021), and NPIAP 2020 conferences.
Inspiring innovation
C.A.R.E. continues to inspire innovations and new knowledge development within the organization. Currently, a WOC nurse is leading the development of a similar innovation (C.R.A.D.L.E.) to address medical device pressure injuries in the neonatal population. A two-part, system-level quantitative research study is being led by the nursing workgroup to identify pressure injury risk factors and test a risk assessment instrument that includes a focus on medical devices. These high-level scholarly activities enable bedside nursing professionals to generate measurable benefits and exemplify distinction with cutting-edge, evidence-based care. AN
The authors work at Baptist Health Lexington in Lexington, Kentucky. C. Preston Lewis is executive director of the orthopedics, wound ostomy, and continence, and Magnet Recognition Program. Kristene E. Colcord is a wound, ostomy, and continence nurse. Ashley Peterson is the Magnet Program coordinator. Charmaine Pfister, Mary Ellen Robertson, and Aaron Slyh are wound, ostomy, and continence nurses. Brittany Smoot is a professional development specialist. Kathy Tussey is executive director of administrative services. Heather Whalen is a wound, ostomy, and continence nurse.
References
Agency for Healthcare Research and Quality. Preventing pressure ulcers in hospitals: A toolkit for improving quality of care. October 2014. https://www.ahrq.gov/patient-safety/settings/hospital/resource/pressureulcer/tool/index.html
Ahtiala MH, Soppi ET, Wiksten A, Koskela H, Grönlund JA. Occurrence of pressure ulcers and risk factors in a mixed medical-surgical ICU—A cohort study. J Intensive Care Soc. 2014;15(4):340-3. doi:10.1177/175114371401500415
Baharestani M, Quillen JH. Medical device related pressure injuries: the hidden epidemic across the lifespan. Presentation on behalf of National Pressure Ulcer Advisory Panel. February 28, 2013.
Black J, Alves P, Brindle CT, et al. Use of wound dressings to enhance prevention of pressure ulcers caused by medical devices. Int Wound J. 2015;12(3):322-7. doi:10.1111/iwj.12111
Black JM, Cuddigan JE, Walko MA, Didier LA, Lander MJ, Kelpe MR. Medical device related pressure ulcers in hospitalized patients. Int Wound J. 2010;7(5):358-65. doi: 10.1111/j.1742-481X.2010.00699.x
Cavalcanti EdO, Kamada I. Medical-device-related pressure injury on adults: An integrative review. Text & Context Nursing. 2020;29:e20180371. doi:10.1590/1980-265x-tce-2018-0371
European Pressure Ulcer Advisory Panel, National Pressure Injury Advisory Panel, Pan Pacific Pressure Injury Alliance. Prevention and treatment of pressure ulcers/injuries: Clinical practice guideline. 2019. internationalguideline.com
The Joint Commission. Managing medical device-related pressure injuries. Quick Safety 43. 2018. jointcommission.org/-/media/tjc/newsletters/qs_43_med_dev_pressure_injuries_7_17_18_final2pdf.pdf
National Pressure Injury Advisory Panel. Pressure injury stages. 2016. npiap.com/page/PressureInjuryStages
National Pressure Injury Advisory Panel. Best Practices for Prevention of Medical Device-Related Pressure Injuries. February 2020. cdn.ymaws.com/npiap.com/resource/resmgr/mdpi-poster2020.pdf
Prevention Plus Home of the Braden Scale. Bibliography. http://www.bradenscale.com/bibliography.htm
Watson J. Nursing: The Philosophy and Science of Caring. Boulder, CO: University Press of Colorado; 2008.
Wound, Ostomy and Continence Nurses Society. WOCN Society Position Paper: Avoidable Versus Unavoidable Pressure Ulcers (Injuries). 2017. cdn.ymaws.com/member.wocn.org/
resource/resmgr/document_library/Avoidable_vs._Unavoidable_Pr.pdf

















1 Comment.
very informative.