Standardized care and compassion are key to successful treatment.
Takeaways
- Evidence-based standardized protocols are needed to provide the best treatment for the mother-infant dyad and improve outcomes for infants neonatal abstinence syndrome (NAS) and mothers with substance use disorders.
- Nurses must increase their awareness, establish trust, and support mothers who frequently feel judged and stigmatized because of drug use.
- An infant’s recovery from NAS depends on positive attachment to a mother who is able to respond effectively to her infant’s needs.
- To improve outcomes for infants with NAS and the mothers of these infants, a broader healthcare perspective is needed that recognizes that NAS isn’t an acute medical condition but requires a multidisciplinary team care approach.
From the Delivery Room to Discharge: A Guide for Pulse Oximetry and Care
The opioid crisis and the matter of consent
- Identify manifestations of neonatal abstinence syndrome (NAS).
- Describe the treatment of NAS.
- Discuss how nurses can address challenges of NAS.
The authors and planners of this CNE activity have disclosed no relevant financial relationships with any commercial companies pertaining to this activity. See the last page of the article to learn how to earn CNE credit.
Expiration: 1/1/23
Opioid use disorder in the United States is a public health emergency not only for adults, but also for newborns. Infants exposed to opioids in utero and who exhibit withdrawal signs after birth are diagnosed with neonatal abstinence syndrome (NAS). According to Winkelman and colleagues, NAS incidence has quadrupled within the last decade; as of 2014, the incidence was approximately 14.4 cases per 1,000 births. More recent reports indicate that the incidence continues to rise, suggesting rates as high as 20 cases per 1,000 live births. Throughout the United States, at least one newborn is affected by NAS every 15 minutes.
The standard of care for newborns with NAS includes an environment that supports physiologic stability by offering comforting interventions such as holding, swaddling, and decreasing excessive stimulation. Infants who don’t respond to this care may require pharmacologic treatment, most commonly with morphine or methadone. Pharmacologic intervention is associated with significantly longer lengths of stay, increased hospital stay costs, and the potential for mother and baby separation.
Clinical effects
Newborns are incapable of experiencing the psychological or relational components that define addiction in adults, so they shouldn’t be described as addicted. Maternal use of various drugs (opioids, barbiturates, benzodiazepines, nicotine) individually or combined can lead to NAS; however, clinical NAS most commonly results from in utero opioid exposure. Anywhere from 55% to 94% of infants who are exposed to opioids in utero develop NAS.
NAS commonly affects the neurologic, respiratory, and GI systems. (See NAS signs.) Clinical signs vary for each infant and are based on several factors, including the type and amount of drug exposure, the time of last maternal drug use, and the drug’s half-life.
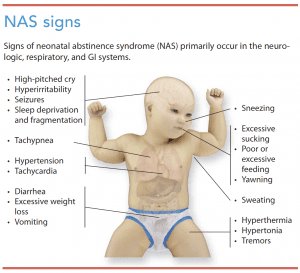

For example, signs of NAS from in utero heroin exposure typically begin within 24 hours of birth, while signs of withdrawal from methadone usually begin between 24 to 72 hours after birth and in some cases may not manifest until 5 to 7 days after. The length and extent of withdrawal signs vary. An infant exposed to a drug with a shorter half-life (for example, hydrocodone, which has an average half-life of 4 hours) may be safely discharged if no withdrawal signs occur by 3 days after birth. However, an infant exposed to a drug with a prolonged half-life (for example, methadone, which has a half-life of 19 to 41 hours) should be observed for at least 5 to 7 days. The lasting effect of drug exposure on signs of withdrawal is unknown. A study by Patrick and colleagues indicates that infants exposed to drugs with prolonged half-lives may have NAS signs for 4 to 6 months.
In addition to in utero drug exposure, genetics, sex, birth weight, gestational age, infant metabolism, maternal tobacco use, and exposure to other prenatal medications play a role in the development and severity of withdrawal.
Diagnosis
Diagnosing an infant with NAS requires an accurate history of the mother’s drug use (including the last drug used and the time of consumption) and evidence of withdrawal. A scoring system can be used to aid diagnosis. Points are assigned based on the severity of each sign, and the total score helps determine the treatment plan. No strong evidence exists that one scoring tool is superior to another; however, research shows that a standard approach to diagnosis and treatment using a scoring tool improves outcomes and is recommended by the American Academy of Pediatrics.
Finnegan NAS scoring system
The Finnegan NAS scoring system, developed in 1975, is the seminal and most common tool used to guide pharmacologic NAS treatment. Nurses evaluate infants every 1 to 4 hours, based on their age, and score them on the presence and severity of common withdrawal signs, including central nervous system, metabolic vasomotor, respiratory, and GI disturbances. Pharmacologic treatment is recommended for any infant who receives a score ≥ 8 on three consecutive evaluations.
The Finnegan tool is long and complex, but a simplified short form recently was developed as a more efficient option. The short form allows for rapid assessment with limited items for scoring; however, although simpler to use, limited evidence exists to validate its use.
Other assessment tools to guide pharmacologic NAS treatment include the 11-item Lipsitz Neonatal Drug-Withdrawal Scoring System, the seven-item Neonatal Narcotic Withdrawal Index, the seven-item Neonatal Withdrawal Inventory, and the 19-item MOTHER NAS Scale. These tools haven’t been as widely adopted as the Finnegan tool.
Eat, Sleep, Console scoring tool
The new Eat, Sleep, Console (ESC) NAS assessment scoring method uses regular assessments of the infant’s ability to eat, sleep, and be consoled to determine the need for pharmacologic treatment. (See Eat, Sleep, Console.) Several studies of the ESC approach report decreased lengths of stay, less unnecessary exposure to pharmacologic treatments, and lowered care costs.
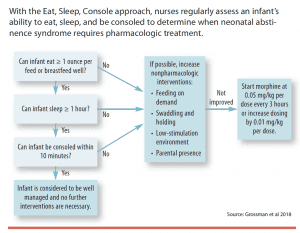

Treatment
The standard of care for infants with NAS, first described in the 1970s, includes holding, nonnutritive sucking, swaddling, pressure, rubbing, swaying, rocking, and reducing external stimulation. Infants with NAS who require hospitalization traditionally have been admitted to special care nurseries or the neonatal intensive care unit (NICU).
For babies who have persistent scores indicating withdrawal and who aren’t responsive to initial interventions, a typical pharmacologic treatment plan includes opioid administration (usually oral morphine or methadone). Approximately 50% to 80% of infants exposed to opiates in utero receive pharmacologic management.
In response to the rise in NAS incidence, recognition of its complexity, and the impact of social factors, such as education, socioeconomic status, and living situation (social determinants of health), evaluation and treatment are being revisited. Newer evidence-based approaches strongly recommend a comprehensive and holistic approach that includes addressing the mother’s psychosocial needs, evaluating the impact of social determinants of health, and taking advantage of community resources.
Challenges
Nurses encounter many challenges when considering appropriate evaluation, diagnosis, and treatment for infants with NAS.
Challenge 1: Rising incidence
Increased incidence of NAS has improved awareness and highlighted the number of healthcare resources required to care for these patients. Infants with NAS have complex physiologic and behavioral states that demand increased nursing care and unit resources. Nurses’ primary responsibility is to provide care and support for the infant, but they also must support the mother, who in addition to a history of substance use disorder also may have complex psychosocial situations that complicate her care of a newborn. To meet the needs of each infant with NAS and support his or her mother, appropriate nurse staffing and adequate support and resources for families are essential.
Evidence shows that standardized assessment and treatment of infants with neonatal abstinence syndrome (NAS) results in better patient outcomes, although no consensus exists as to the best protocol to use. Regardless of which protocol is chosen, nurses should establish and adhere to a consistent approach to evaluating and assessing these infants. All nurses who perform newborn assessments should complete education related to the protocol.
Vermont Oxford Network
The Vermont Oxford Network has established a multicenter quality collaborative focused on standardizing the care of infants with NAS. A toolkit available from the network focuses on developing and implementing:
- a standardized process for identifying, evaluating, treating, and discharging infants with NAS
- a standardized process for measuring and reporting NAS and drug exposure
- a culture of healing for the mother-infant dyad.
Challenge 2: Care variation
Care of infants with NAS varies widely because no consensus exists on the best diagnostic or treatment strategy. Variations in care and using a subjective, rather than objective, scoring tool increase the potential for unnecessary exposure to pharmacologic treatment. Lack of standardization, inconsistent care plans, and pharmacologic treatment are associated with longer lengths of stay, unnecessary separation of the mother and infant, and poor long-term developmental outcomes. Evidence of improved outcomes supports local improvement efforts that focus on standardizing care. (See Follow a standard.)
Challenge 3: Care complexity
Many women with a substance use disorder also experience mental illness, trauma, economic hardship, intimate partner violence, unintended pregnancy, and housing instability. Each of these circumstances complicate efforts to decrease and eliminate substance use during pregnancy and after birth and may negatively impact the NAS treatment plan. Caring for an infant is challenging even for healthy mothers with stable support systems, but those with a substance use disorder may have strained relationships with family and friends, leading to a lack of support as well as no access to transportation to receive care. These women also face significant social stigma within the community and healthcare system.
Training and education for healthcare providers focused on the care of these women is limited, and perceptions are often negative. Such attitudes and unconscious (or conscious) bias reinforce stigma, contribute to inequitable treatment, disempower these mothers, and decrease trust in healthcare providers, all of which are associated with poor mother and infant outcomes.
An infant’s recovery from NAS depends on positive attachment to a mother who can respond effectively to her infant’s needs. A mother’s ability to bond and respond appropriately is determined in large part by the type and quality of support she has. Healthcare teams must acknowledge existing biases and provide equitable and quality care that addresses and supports mothers’ complex social needs.
Challenge 4: NICU environment
Most infants who require hospitalization for NAS are admitted to the NICU, which frequently is loud and chaotic and may exacerbate signs of the condition. The physical space also usually separates the mother-infant dyad. A calm, nurturing environment conducive to mother-child bonding is best for infants with NAS.
Evidence-based care models facilitate concurrent care for mothers and infants and support rooming-in and breastfeeding. In NICUs that can’t support this model, other hospital units, including pediatric units, are used. Nurses on these units must be adequately educated to understand the needs of infants with NAS and mothers with a substance use disorder. Comprehensive, multidisciplinary supportive care, regardless of the hospital unit, requires resources such as breastfeeding support, social work, volunteer cuddler programs, and pharmacists.
Challenge 5: Social implications
To adequately address the social needs of infants with NAS and their mothers, nurses must collaborate with social workers, case managers, and community-based organizations. Syvertsen and colleagues propose a framework that considers NAS management a “cascade of care” that begins with prevention, prenatal care, and drug treatment for the mother, extends throughout hospitalization from labor and delivery to the NICU, and continues after discharge as the mother and infant transition to their home and community. This approach requires a multidisciplinary team that includes physicians, nurses, social workers, occupational therapists, and case managers who provide care to the mother and infant during the hospital stay and connect them with resources after discharge.
Intervention from state child protective services may be appropriate for some infants with NAS to ensure they’re safe and that their families have the necessary support to care for their new babies. Although some states still criminalize pregnant women with a substance use disorder, the American Academy of Nursing (and many other professional organizations) calls for an end to this practice and supports a public health response that provides care and support.
Nursing implications
Studies have identified the benefit of prenatal care screening and early intervention, but barriers to care may mean that NICU and other nurses are the first sustained contact that mothers of infants with NAS have with the healthcare system. Nurses have an opportunity to encourage and facilitate integrated, comprehensive, multidisciplinary care; establish trust; empathize with mothers to increase maternal-infant bonding; understand the context of substance use and effective treatment strategies; and encourage harm reduction. (See 3 key takeaways.)
Achieving these outcomes requires education for nurses and self-reflection about substance use assumptions and biases.
Caring for infants with neonatal abstinence syndrome (NAS) and their mothers with a substance use disorder includes promoting mother-infant bonding, encouraging breastfeeding, and administering medication-assisted treatment as prescribed.
Bonding
Maternal-infant bonding has the greatest long-term effect on developmental and behavioral outcomes for infants with NAS. Nurses will have the biggest impact when care is thoughtful and intentionally focused on bonding and supporting maternal caregiving skills. The World Health Organization identified five principles for caring for mothers with a substance use disorder:
- prioritizing prevention
- ensuring access to prevention and treatment services
- respecting patient autonomy
- providing comprehensive care
- safeguarding against discrimination and stigmatization.
Breastfeeding
Breastfeeding has positive effects for both the mother (sobriety promotion, infant bonding, and enhanced self-esteem) and infant (reduced severity of NAS and decreased need for pharmacologic treatment). Several professional organizations (including the American College of Obstetricians and Gynecologists, American Academy of Pediatrics, and the Academy of Breastfeeding Medicine) recommend breastfeeding for mothers on opioid agonist therapy and have published guidelines supporting this practice. Nurses should be aware of their organization’s policy regarding breastfeeding and maternal substance use. Evidence shows that support from healthcare providers is a strong predictor of whether mothers breastfeed.
Medication-assisted treatment
Although medication-assisted treatment (MAT) during pregnancy carries risks for the fetus, MAT with methadone or buprenorphine is the standard of care for mothers with a substance use disorder and is associated with better maternal and fetal outcomes.
Education
Nurse distress and frustration when caring for infants with NAS frequently is linked to lack of knowledge and understanding about mental illness and substance use, as well as the accompanying social complexities (poverty, homelessness, and trauma). Organizations should offer education that helps nurses build trusting partnerships with mothers with mothers affected by substance use disorder.
Self-assessment
Nurses can prepare to care for infants with NAS and their mothers by examining any implicit biases they may have about substance use that might affect their ability to develop rapport and establish trust. Substance use disorders frequently are linked to trauma, and nurses must provide care that acknowledges the trauma and safeguards against it.
Similar to the universal precautions approach, nurses should assume that all parents of NICU patients have a history of trauma and then seek to understand and address their psychosocial needs. This approach decreases the potential for bias and ensures more consistent care. It also allows for understanding when mothers of infants with NAS show signs of defensiveness, anger, and frustration, and why a mother might be absent, distant, or depressed. Viewing each mother’s behavior through this lens creates an opportunity to gain understanding of her broader life circumstances and to address the barriers and challenges she faces to provide a stable and secure home environment. This understanding allows for the provision of equitable care and the ability to identify appropriate support and resources.
Foster respect and compassion
Providing standardized care for infants with NAS must be accompanied by equal care and support of the mother. Fostering a respectful and compassionate environment and establishing a nonjudgmental, trusting relationship with mothers will help them learn to interpret and manage signs of NAS in their infants.
Nursing assessment and critical thinking skills are instrumental to recognizing signs of infant withdrawal and facilitating diagnosis and treatment. Nurses should collaborate with hospital leadership, community partners, and elected representatives to advocate for policy change that supports and protects infants with NAS and their mothers and facilitates better access to care and resources related to substance use disorders. (See Nursing best practices.)
When caring for infants with neonatal abstinence syndrome (NAS) and mothers with a substance use disorder:
- acknowledge that implicit bias is present within the healthcare system and aim to provide nonbiased care by examining preconceptions that may affect your ability to develop rapport and establish trust
- develop a respectful and empathetic nurse-patient relationship
- promote a calm, nurturing environment
- promote colleagues’ understanding of substance use disorders and provide education on the importance of harm reduction (as opposed to insisting on abstinence) to facilitate treatment adherence and engagement
- provide maternal support by modeling appropriate infant care, encouraging maternal-infant bonding, and striving to avoid unnecessary mother-child separation
- offer assistance, guidance, and emotional and technical support to promote breastfeeding
- reduce barriers to care by advocating for multidisciplinary, collaborative care with social workers, mental health care providers, lactation experts, case managers, and community organizations
- collaborate at the organizational, community, and state levels to effect policy change and increase public awareness of NAS
- help establish and update standardized unit guidelines for NAS management.
Interventions for pregnant women and women of childbearing age with a substance use disorder
- Screen for substance use disorders when possible.
- Promote access to prevention, prenatal care, and treatment services.
- Educate patients about medication-assisted treatment as prescribed by the provider.
Interventions for infants with NAS
- Perform patient assessment and scoring.
- Implement nonpharmacologic comfort measures, including:
- restful sleep
- decreased external stimulation (provide dark, quiet room)
- infant holding
- nonnutritive sucking
- swaddling, swaying, and rocking.
- Administer pharmacologic treatment as ordered by the provider.
- Provide treatment education to the infant’s family.
Elizabeth Schierholz is a neonatal nurse practitioner at Children’s Hospital of Colorado in Aurora, and a research fellow in data science to patient values at the University of Colorado School of Medicine in Aurora. Rachel French is a predoctoral research fellow at the University of Pennsylvania School of Nursing in Philadelphia. Anne-Marie Boucher is a neonatal nurse practitioner at C.S. Mott Children’s Hospital in Ann Arbor, Michigan.
Access these resources for more information about neonatal abstinence syndrome.
- Helping children born dependent on opioids. NOVA. November 5, 2018. to.pbs.org/2Wl7oYr
- Public health approaches to addressing neonatal abstinence syndrome. Children’s Safety Network. March 20, 2018. childrenssafetynetwork.org/webinar/public-health-approaches-addressing-neonatal-abstinence-syndrome
- Weiner J. To treat babies for drug withdrawal, help their mothers, too. Undark. December 12, 2018. bit.ly/2JBnBak
- Cohen J. A nurse’s lesson: Babies in opioid withdrawal still need mom. Kaiser Health News. March 29, 2016. bit.ly/2uqIMRV
- Neonatal abstinence syndrome—PediaCast CME 042. Nationwide Children’s Hospital. February 27, 2019. bit.ly/2FyLo6J
- Egan J. Children of the opioid epidemic. The New York Times Magazine. May 9, 2018. nyti.ms/2jI1G1n
References:
Alexander K. Social determinants of methadone in pregnancy: Violence, social capital, and mental health. Issues Ment Health Nurs. 2013;34(10):747-51.
Bagley SM, Wachman EM, Holland E, Brogly SB. Review of the assessment and management of neonatal abstinence syndrome. Addict Sci Clin Pract. 2014;9(1):19.
Child Welfare Information Gateway. What is child welfare? A guide for educators. 2018. childwelfare.gov/pubs/cw-educators
Coyle MG, Brogly SB, Ahmed MS, Patrick SW, Jones HE. Neonatal abstinence syndrome. Nat Rev Dis Primers. 2018;4(1):47.
D’Apolito KC. Assessing neonates for neonatal abstinence: Are you reliable? J Perinat Neonatal Nurs. 2014;28(3):220-31.
Dobbing J, Sands J. Head circumference, biparietal diameter and brain growth in fetal and postnatal life. Early Hum Dev. 1978;2(1):81-7.
Finnegan LP, Kron RE, Connaughton JF, Emich JP. Assessment and treatment of abstinence in the infant of the drug-dependent mother. Int J Clin Pharmacol Biopharm. 1975;12(1-2):19-32.
Gomez Pomar, Finnegan LP, Devlin L, et al. Simplification of the Finnegan neonatal abstinence scoring system: Retrospective study of two institutions in the USA. BMJ Open. 2017;7(9):e016176.
Grisham LM, Stephen MM, Coykendall MR, Kane MF, Maurer JA, Bader MY. Eat, sleep, console approach: A family-centered model for the treatment of neonatal abstinence syndrome. Adv Neonatal Care. 2019;19(2):138-44.
Grossman MR, Berkwitt AK, Osborn RR, et al. An initiative to improve the quality of care of infants with neonatal abstinence syndrome. Pediatrics. 2017;139(6):e20163360.
Grossman MR, Lipshaw MJ, Osborn RR, Berkwitt AK. A novel approach to assessing infants with neonatal abstinence syndrome. Hosp Pediatr. 2018;8(1):1-6.
Holmes AV, Atwood EC, Whalen B, et al. Rooming-in to treat neonatal abstinence syndrome: Improved family-centered care at lower cost. Pediatrics. 2016;137(6):e20152929.
Hudak ML, Tan RC, American Academy of Pediatrics Committee on Drugs, Committee on Fetus and Newborn. Neonatal drug withdrawal. Pediatrics. 2012;129(2):e540-60.
Jessup MA, Oerther SE, Gance-Cleveland B, et al. Pregnant and parenting women with a substance use disorder: Actions and policy for enduring therapeutic practice. Nurs Outlook. 2019;67(2):199-204.
Keough L, Fantasia HC. Pharmacologic treatment of opioid addiction during pregnancy. Nurs Womens Health. 2017;21(1):34-44.
Kramlich D, Kronk R. Relational care for perinatal substance use: A systematic review. MCN Am J Matern Child Nurs. 2015;40(5):320-6.
Maguire D. Care of the infant with neonatal abstinence syndrome: Strength of the evidence. J Perinat Neonatal Nurs. 2014;28(3):204-11.
Maguire D, Cline GJ, Parnell L, Tai CY. Validation of the Finnegan neonatal abstinence syndrome tool—short form. Adv Neonatal Care. 2013;13(6):430-7.
Marcellus L. Supporting women with substance use issues: Trauma-informed care as a foundation for practice in the NICU. Neonatal Netw. 2014;33(6):307-14.
McQueen KM, Murphy-Oikonen J. Neonatal abstinence syndrome. N Engl J Med. 2016;375(25):2468-79.
Newman A, Davies GA, Dow K, et al. Rooming-in care for infants of opioid-dependent mothers: Implementation and evaluation at a tertiary care hospital. Can Fam Physician. 2015;61(12):e555-61.
Patrick SW, Burke JF, Biel TJ, Auger KA, Goyal NK, Cooper WO. Risk of hospital readmission among infants with neonatal abstinence syndrome. Hosp Pediatr. 2015;5(10):513-9.
Patrick SW, Schumacher RE, Horbar JD, et al. Improving care for neonatal abstinence syndrome. Pediatrics. 2016;137(5):e20153835.
Penn Medicine News. Creating comfort through cuddling. March 29, 2018. pennmedicine.org/news/internal-newsletters/system-news/2018/april/creating-comfort-through-cuddling
Romisher R, Hill D, Cong X. Neonatal abstinence syndrome: Exploring nurses’ attitudes, knowledge, and practice. Adv Neonatal Care. 2018;18(2):E3-11.
Saia KA, Schiff D, Wachman EM, et al. Caring for pregnant women with opioid use disorder in the USA: Expanding and improving treatment. Curr Obstet Gynecol Rep. 2016;5(3):257-63.
Short VL, Cambareri K, Gannon M, Alexander K, Abatemarco DJ. A pilot study to assess breastfeeding knowledge, attitudes, and perceptions of individuals who work in perinatal opioid use disorder treatment settings. Breastfeed Med. 2019;14(5):307-12.
Smith JG, Rogowski JA, Schoenauer KM, Lake ET. Infants in drug withdrawal: A national description of nurse workload, infant acuity, and parental needs. J Perinat Neonatal Nurs. 2018;32(1):72-9.
Stanford Children’s Health. Neonatal abstinence syndrome. stanfordchildrens.org/en/topic/default?id=neonatal-abstinence-syndrome-90-P02387
Sutter MB, Gopman S, Leeman L. Patient-centered care to address barriers for pregnant women with opioid dependence. Obstet Gynecol Clin North Am. 2017;44(1):95-107.
Syvertsen JL, Toneff H, Madden DR, Clapp JD. Conceptualizing neonatal abstinence syndrome as a cascade of care: A qualitative study with healthcare providers in Ohio. Adv Neonatal Care. 2018;18(6):488-99.
Tobin KB. Changing neonatal nurses’ perceptions of caring for infants experiencing neonatal abstinence syndrome and their mothers: An evidenced [sic] -based practice opportunity. Adv Neonatal Care. 2018;18(2):128-35.
Tolia VN, Patrick SW, Bennett MM, et al. Increasing incidence of the neonatal abstinence syndrome in U.S. neonatal ICUs. N Engl J Med. 2015;372(22):2118-26.
Velez M, Jansson LM. The opioid dependent mother and newborn dyad: Non-pharmacologic care. J Addict Med. 2008;2(3):113-20.
Wachman EM, Grossman M, Schiff DM, et al. Quality improvement initiative to improve inpatient outcomes for neonatal abstinence syndrome. J Perinatol. 2018;38(8):1114-22.
Wachman EM, Schiff DM, Silverstein M. Neonatal abstinence syndrome: Advances in diagnosis and treatment. JAMA. 2018;319(13):1362-74.
Winkelman TNA, Villapiano N, Kozhimannil KB, Davis MM, Patrick SW. Incidence and costs of neonatal abstinence syndrome among infants with Medicaid: 2004-2014. Pediatrics. 2018;141(4):e20173520.
Wood T, Bordelon C, Fogger S. Shining a light: Integrating protocols into clinical practice for treatment of neonatal abstinence syndrome. J Addict Nurs. 2019;30(1):61-6.
World Health Organization. Guidelines for the identification and management of substance use and substance use disorders in pregnancy. 2014. apps.who.int/iris/bitstream/handle/10665/107130/9789241548731_eng.pdf;jsessionid=D540E1504888E85ABD91D1664BDE0C1A?sequence=1



















2 Comments.
That’s a judgement right there! Yes, it is true, doctors and nurses have to do so much to help the babies, but we must make room to address the root causes and root person. We do need to help the mother to help the baby, right? Never too late to start giving a helping hand!
The whole thing is very depressing. Doctors and nurses have to do so much to get these harmed babies healthy only to release them to a mother who would take opioids while pregnant.