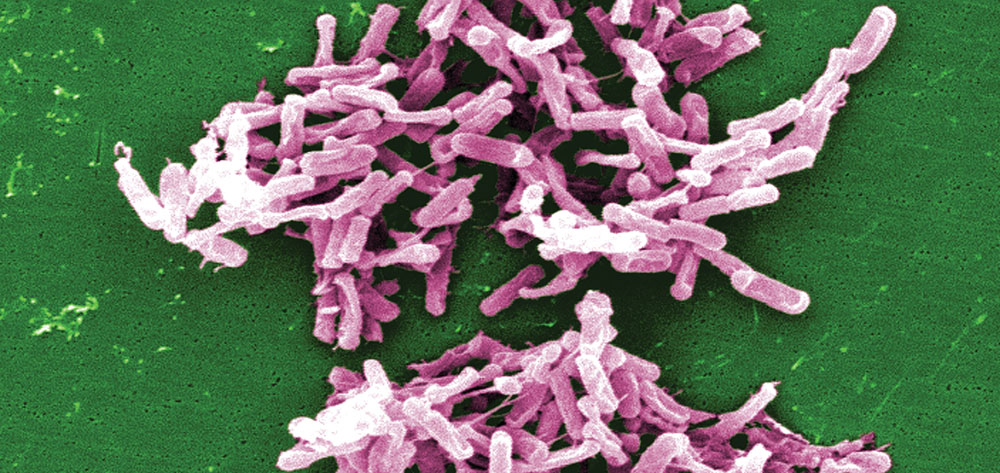
Move over, MRSA. Clostridium difficile is fast becoming a rampant hospital infection. A gram-positive, spore-forming, anaerobic bacillus, C. difficile is now the most common cause of antibiotic-associated diarrhea, infecting or colonizing more than 13 of every 1,000 in-patients. A 2008 study conducted by the Association for Professionals in Infection Control and Epidemiology found infection rates 6.5 to 20 times greater than previous estimates. This means that on any given day, at least 7,178 inpatients are infected with C. difficile.
Like many microbes, C. difficile has changed over the years. The diarrhea it causes (termed C. difficile–associated diarrhea, or CDAD) has gotten more severe and more prevalent—and a more virulent and resistant strain, called the North American Pulsed Field Type 1, has emerged.
Most victims contract CDAD in hospitals or nursing homes after receiving antibiotics. Of these, 0.6% to 1.5% die, with CDAD the direct or indirect cause of death. Stopping the spread of this potentially lethal infection demands early recognition and treatment.
Scope of the problem
According to the Centers for Disease Control and Prevention, C. difficile outbreaks have occurred in almost every state. Canada has also been affected; in 2004, the disease caused more than 100 deaths over an 18-month period at a Quebec hospital.
Scarier still, C. difficile is showing up more often in healthy people who’ve neither been in the hospital nor taken recent antibiotics. In 2003, of the 33 cases reported in the Philadelphia area, 23 were in otherwise healthy patients who hadn’t been admitted to a hospital within the previous 3 months; 10 were in healthy pregnant women or women who’d given birth recently and been hospitalized briefly; one patient died despite treatment.
Terrible toxins
A common bacterium of the GI tract, C. difficile normally aids digestion and absorption of food and other nutrients. It’s part of the normal flora in about 3% of healthy adults and about 10% to 30% of hospitalized or chronically ill patients.
But when C. difficile grows out of control, it produces two toxins that result in CDAD. Called toxins A and B, they cause inflammation, fluid and mucus secretion, and mucosal damage leading to diarrhea or colitis. The resulting infection can cause problems ranging from mild to severe diarrhea to pseudomembranous colitis, toxic megacolon, intestinal perforation, sepsis, and death. These toxins are found in the stool of 15% to 25% of patients with antibiotic-associated diarrhea (AAD) and in 95% of patients with pseudomembranous colitis. (See Pseudomembranous colitis and toxic megacolon by clicking on the PDF icon above.)
How C. difficile causes infection
For a person to become infected with C. difficile, the normal GI flora must be disrupted (most commonly, by antibiotic use). Also, C. difficile bacteria must be ingested and the bacteria must overproduce. Once these events occur, the patient may become colonized with C. difficile, may develop CDAD, or both. Patients with colonization lack clinical manifestations but test positive for the bacteria, its toxins, or both.
Colonization is much more common than CDAD itself; some research suggests it may protect against symptomatic disease by conferring immunity.
Why some patients develop CDAD while others don’t remains unknown. However, production of toxin A or B is essential to its development.
Risk factors
Antibiotic therapy causes about 90% of C. difficile infections. Although fluoroquinolones are the most commonly implicated antibiotic class, most antimicrobials except aminoglycosides have been linked to CDAD.
Other risk factors for CDAD include age greater than 65, severe underlying illness, nasogastric intubation, use of antiulcer medications (proton pump inhibitors and histamine-receptor antagonists), and longer hospital stays. Most common in healthcare settings, CDAD has a prevalence of 4% to 20% in long-term care facilities.
Assessment
CDAD signs and symptoms include watery, foul-smelling diarrhea, fever, nausea, abdominal pain and tenderness, and appetite loss. In some patients, diarrhea may be the sole sign or symptom.
CDAD can be mild, moderate, or severe.
- Mild CDAD causes mild to moderate nonbloody diarrhea, possibly accompanied by low abdominal cramping. Physical examination is unremarkable.
- Moderate CDAD causes copious amounts of watery diarrhea, along with abdominal pain and distention and possible fever, nausea, dehydration, and occult blood in the stool. Sigmoidoscopy shows the characteristic membrane with yellow plaques in the distal colon. Moderate CDAD increases the risk of paralytic ileus and toxic megacolon.
- Severe CDAD is diagnosed when the patient has complications, such as colitis, sepsis, volume depletion, electrolyte imbalance, hypotension, peritonitis, paralytic ileus, or toxic megacolon.
No standard incubation period has been established. Signs and symptoms may arise during antibiotic therapy, immediately afterward, or up to 8 weeks afterward. One study found an incubation ranging from 2 to 60 days.
Relapse and recurrence
Relapse of C. difficile infection is highly characteristic and poses a major complication of antibiotic treatment. Although it usually occurs within 1 week after antibiotic withdrawal, relapse can arise as late as 8 weeks after withdrawal. Typically, it causes signs and symptoms identical to those of the initial illness. Although relapse may result from the initial strain of C. difficile, 48% to 56% of relapsed patients are suffering from a new strain.
Recurrence is frustrating, too. What’s more, determining if a second episode is a reinfection or a relapse is nearly impossible. Within 2 months of the initial diagnosis, 12% to 24% of patients develop a second CDAD episode. In patients who’ve had two or more episodes, the risk of additional recurrences increases to 50% or even 65%.
Diagnosis
Stool specimen analysis is the most common test for C. difficile. Other tests vary in turnaround time, sensitivity (reaction), specificity (toxin type A or type B), cost, and availability.
- Enzyme immunoassay is available in most clinical laboratories. This easy-to-use method has a sensitivity of 80% to 95% and a 2-hour turnaround, but it doesn’t distinguish between toxins A and B.
- Immunochromatographic toxin A testing has a sensitivity of 60% to 85% and a 1-hour turnaround, but it can’t differentiate strains A and B.
- Anaerobic cultures take 72 hours, are expensive, and have variable accuracy due to nonstandardized methods and culture media.
- Tissue cytotoxic assays may yield false-positive results.
- Endoscopy is invasive and has only a 51% sensitivity.
Management and nursing care
Antibiotic withdrawal is crucial in treating CDAD. In up to 25% of cases, the disease resolves once antibiotic therapy is discontinued.
Even with appropriate therapy, CDAD may progress, so be sure to monitor the patient closely to determine if signs and symptoms improve within 1 to 2 days after antibiotic therapy begins. Fever should subside within 24 to 48 hours; diarrhea, within 2 to 5 days.
Unless the patient’s condition deteriorates, treatment shouldn’t be considered a failure until the sixth or seventh day of therapy. If therapy doesn’t seem to be working and the disease progresses, a surgical consult may be needed.
Drug therapy
The first drug of choice is oral metronidazole 250 mg P.O. four times daily or 500 mg P.O. three times daily for 10 days. Metronidazole is relatively cheap, but some C. difficile strains have become resistant to it. Be sure to monitor the patient closely for response to therapy.
If metronidazole proves ineffective, vancomycin 125 to 500 mg P.O. four times daily for 10 days typically is tried next, although some forms of C. difficile resist it. Rifaximin 200 mg P.O. three times daily for 14 days is the next line of defense. Nitazoxanide 500 mg P.O. every 12 hours for 3 days is the drug of last resort.
Unfortunately, giving antimicrobials to CDAD patients is a double-edged sword, as these drugs can themselves both cure and cause AAD. (See Fortifying healthy GI flora.)
Patients with CDAD should not receive antiperistaltic drugs, such as loperamide (Imodium), diphenoxylate with atropine (Lomotil), or paregoric—either alone or in conjunction with any other therapy. These drugs prevent the body from excreting infected stool and may predispose the patient to toxic megacolon. For the same reason, opioids also should be avoided.
Other therapeutic options
Another treatment option is fecal bacteriotherapy. In this method, bacteria from a healthy person’s feces are transferred into the large bowel and rectum of a patient with CDAD.
Prevention
Preventing the spread of CDAD requires a cooperative effort among staff, patients, and visitors. Measures include contact precautions, stringent hand hygiene, environmental considerations, avoiding unnecessary antibiotics, and educating staff, patients, and visitors. The most crucial factor in controlling disease spread is strict adherence to infection control practices. (See Preventing the spread of C. difficile in health care facilities by clicking on the PDF icon above.)
As an environment becomes contaminated with C. difficile, appropriate disinfectant use and adherence to an appropriate cleaning regimen are crucial to decrease disease transmission. Dedicated or disposable equipment should be used whenever possible. Reusable equipment must be cleaned with a hospital-approved cleaning solution before it is placed in a patient room. Rooms must be cleaned with a hospital-approved cleaning solution; during routine cleaning, the commodes and floor must be cleaned thoroughly. Curtains need to be replaced with new ones if they become visibly soiled, or when the patient is discharged.
Ultimately, prevention efforts require clinicians to be more diligent about prescribing antibiotics only when appropriate, as broad-spectrum antibiotics alter the flora of the colon and set the stage for CDAD. Also, healthcare providers should instruct all patients to complete the full course of antibiotic therapy to prevent development of drug-resistant bacteria.
Selected references
Bartlett JG. Narrative review: the new epidemic of Clostridium difficile–associated enteric disease. Ann Intern Med. 2006;145(10):
758-764.
Centers for Disease Control and Prevention. Clostridium difficile information for healthcare providers. www.cdc.gov/ncidod/dhqp/
id_CdiffFAQ_hcp.html. Accessed June 16, 2010.
Cloud J, Kelly CP. Update on Clostridium difficile–associated disease. Curr Opin Gastroenterol. 2007;23:4-9.
Infection control: minimizing the growing threat of Clostridium difficile. Jt Comm Perspect Patient Safety. 2006;6(6):5-6.
Sunenshine RH, McDonald LC. Clostridium difficile–associated disease: new challenges from an established pathogen. Cleve Clin J Med. 2006;73(2):187-197.
<font=size=”-1″>Delilah Hall is a Clinical Nurse Specialist at Cleveland Clinic in Cleveland, Ohio.