Nurses can help reduce potential loss of quality of life.
Takeaways:
- Approximately 16% of adults age 18 years and older report trouble hearing
- Symptoms of hearing loss can range from temporary and inconvenient to permanent and disabling
- Reviewing hearing loss in relation to the type (conductive, sensorineural, or mixed) and ear anatomy helps nurses identify common and concerning causes and presentations.
- Conductive hearing loss frequently is temporary and treatable, whereas sensorineural hearing loss tends to be more serious, progressive, and permanent.
Approximately 16% of U.S. adults age 18 years and older report hearing loss, according to the Centers for Disease Control and Prevention. Symptoms range from temporary and inconvenient, to permanent and disabling. Patients wait an average of 10 years before actively seeking treatment for hearing loss, according to a report from the National Institute on Deafness and Other Communication Disorders (NIDCD). Lack of treatment can lead to social isolation, depression, impaired communication, neurological decline, and decreased quality of life. Nurses may be the first to recognize hearing loss and have an opportunity to start a conversation with the patient.
To understand the causes of hearing loss, nurses must have knowledge of hearing physiology. Sound is a vibration that funnels through the outer ear to the middle ear via the eardrum (tympanic membrane) and the three bones, or ossicles (malleus, incus, and stapes), in the middle ear. Vibration of the ossicles transmits sound waves to the cochlear sensory hair cells in the inner ear. These cells convert the waves into an electrical signal that travels via the auditory nerve to the brain where it’s perceived as sound. (See Anatomy of the ear.)
Anatomy of the ear
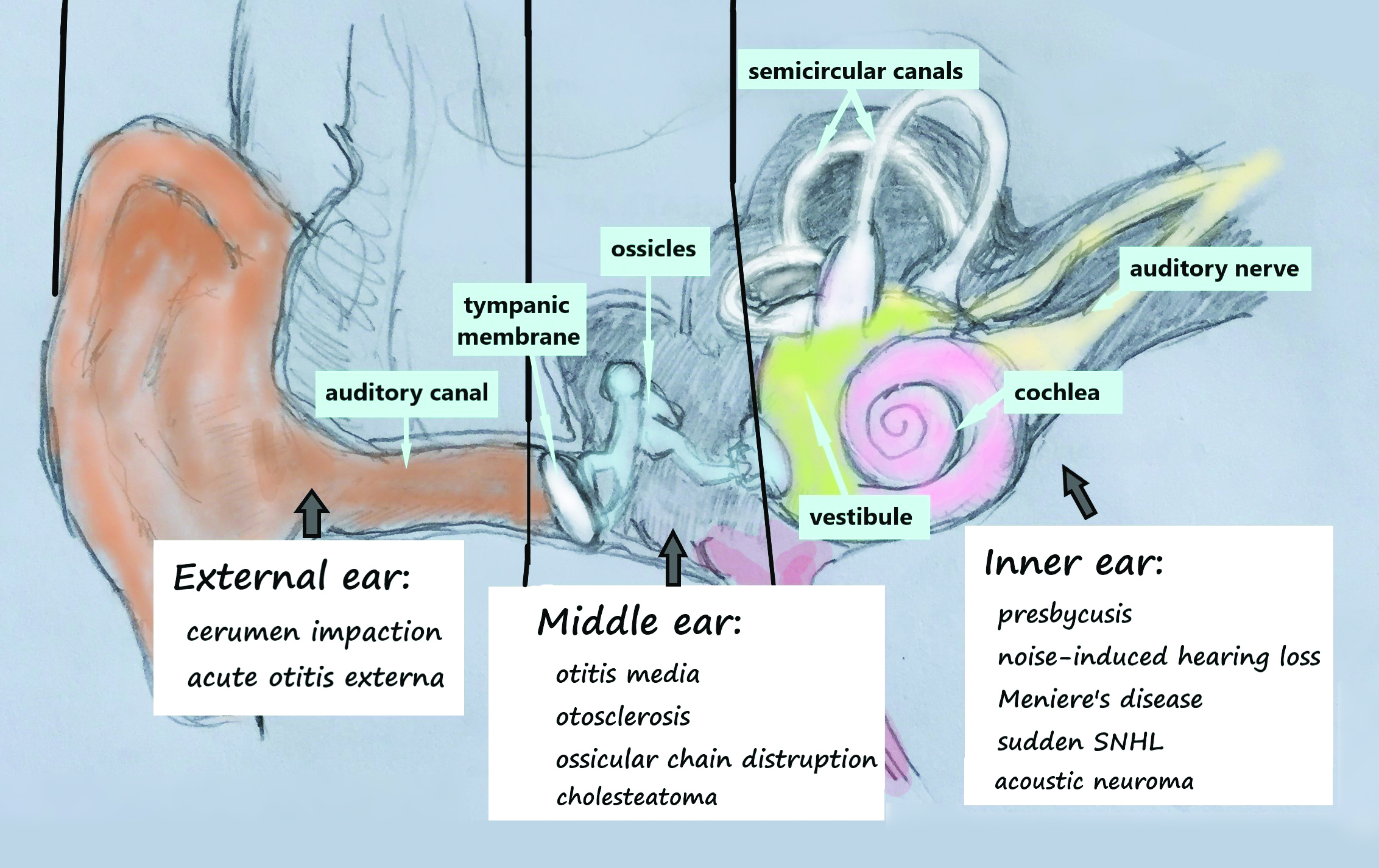

Hearing loss, which can originate anywhere along this pathway, is classified by type (conductive, sensorineural, and mixed [any combination of conductive and sensorineural]) in relation to ear anatomy and physiology. This review will familiarize nurses with the causes of conductive and sensorineural hearing loss in adults and the implications for nursing practice. Autoimmune, vascular, neurogenic, oncological, and congenital hearing loss are beyond the scope of this review. (See Causes of hearing loss.)
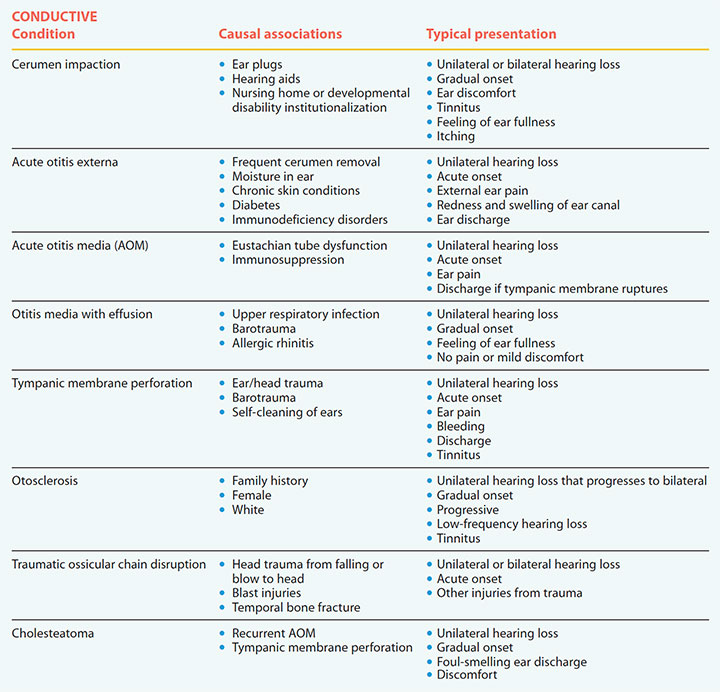
Causes of hearing loss – Conductive
Hearing loss is classified as conductive, sensorineural, or mixed (a combination of any of the conductive and sensorineural conditions). These tables describe causes and typical presentations.

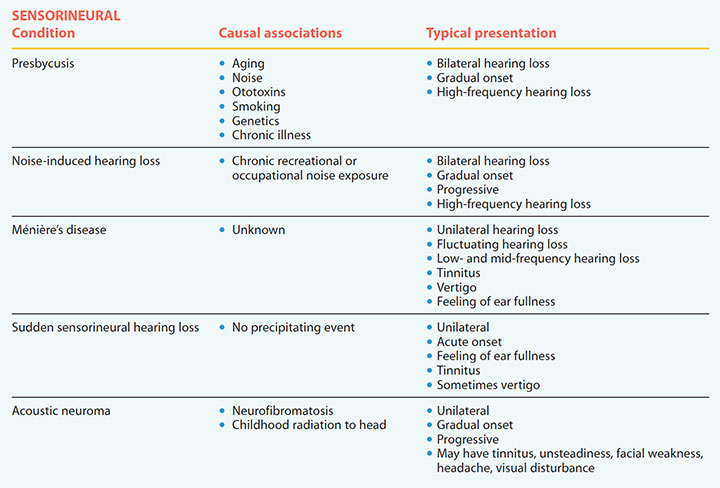
Causes of hearing loss – Sensorineural
Hearing loss is classified as conductive, sensorineural, or mixed (a combination of any of the conductive and sensorineural conditions). These tables describe causes and typical presentations.

Conductive hearing loss
Conductive hearing loss occurs when sound can’t be transmitted from the environment through the external or middle ear to the cochlea. It can result from a blockage of the external or middle ear from substances such as earwax (cerumen), swelling, fluid, pus, a tumor, or a foreign body. Abnormalities of the ossicles also can cause conductive hearing loss. Most causes can be medically or surgically managed.
Cerumen impaction
Cerumen is a naturally occurring, protective substance in the ear canal. Typically, it’s self-clearing, but occlusion can occur if cerumen doesn’t naturally clear or if the patient pushes it deeper into the canal. With marked impaction, sound can’t conduct to the middle ear. According to Yang and colleagues, cerumen impaction is common, affecting 1 in 20 adults and up to 57% of nursing home residents. Patients at particular risk include those who use ear plugs or hearing aids, which may stimulate cerumen production and block outflow. For reasons that aren’t completely clear, others at risk include those who live in nursing homes or are institutionalized for developmental disabilities, possibly due to cognitive deficits or, in the case of some forms of intellectual disability, anatomical differences within the ear canal.
Patients with cerumen impaction frequently report hearing loss, ear discomfort, tinnitus, and a feeling of fullness in the ear. Blockage can cause difficulty hearing normal conversation. Cerumen impaction should be treated only by experienced clinicians via manual removal with specialized instruments, irrigation, or cerumenolytics.
Acute otitis externa
Acute otitis externa (AOE)—sometimes called “swimmer’s ear”—is a painful infection of the external ear canal caused predominantly by bacterial pathogens. Symptoms present acutely and include external ear pain, redness and swelling of the ear canal, ear discharge, itching, fullness, and possibly hearing loss.
Common risk factors for AOE include frequent self-removal of cerumen, warm and moist climates, hearing aid use, chronic skin conditions, diabetes, immunodeficiency disorders, and swimming. Severe AOE may cause marked inflammation and discharge, which prevent sound conduction through the ear canal. Uncomplicated AOE is treated with analgesics and topical agents such as antibiotics, steroids, and acetic acid drops.
Acute otitis media
Acute otitis media (AOM) is a bacterial infection of the middle ear characterized by an acute onset of ear pain with middle ear inflammation and fluid, which prevent adequate sound transmission to the inner ear. According to Rijk and colleagues, the incidence of AOM in adults is only 5/1,000 person-years.
The most common symptoms are unilateral earache, discharge, and hearing loss. Spontaneous rupture of the tympanic membrane (TM) may occur more frequently in adults than in children, further complicating the condition. Common AOM treatment for adults is an oral antibiotic; watchful waiting usually is indicated for children. Barring any complications, hearing should return to normal after the infection is cleared.
Otitis media with effusion
Similar to AOM, otitis media with effusion (OME) can cause conductive hearing loss as a result of fluid and inflammation in the middle ear. The middle ear mucosa becomes inflamed, causing negative air pressure in the middle ear. This negative pressure, coupled with the inability of the eustachian tube to open properly, causes fluid to flow (effuse) from the mucosa and fill the middle ear cavity. Unlike AOM, OME isn’t an infectious process and usually is painless. However, it may result from fluid accumulation caused by AOM or predispose the patient to infection because of trapped fluid and bacteria. OME is associated with eustachian tube dysfunction.
OME can develop after an upper respiratory tract infection, barotrauma from pressure changes associated with flying or scuba diving, and allergic rhinitis. It affects children more frequently than adults. The incidence of OME in adults hasn’t been established, but it may be a symptom of an underlying nasopharyngeal carcinoma that damages the eustachian tube.
Uncomplicated OME typically resolves spontaneously, but recurring or persistent OME requires otolaryngology consultation. Treatment in adults hasn’t been well studied but may include inserting tympanostomy tubes to drain the fluid and equalize pressure.
TM perforation
Holes in the TM reduce the surface area of the eardrum, decreasing sound transmission to the ossicles. Perforation can occur as a result of AOM, head or ear trauma, barotrauma, and self-inflicted punctures during ear cleaning. Symptoms depend on the cause but may include ear pain, bleeding, discharge, tinnitus, and hearing loss. Significant hearing loss is associated with larger holes.
Patients with large perforations, perforations that don’t heal within 2 months, or vertigo are at greater risk for damage to the ossicles and require otolaryngology consultation. Treatment depends on the cause and severity of the defect and may include surgical reconstruction or grafting.
Otosclerosis
Otosclerosis occurs as a result of abnormal ossicles remodeling, which prevents sound waves from vibrating the bones normally. According to the American Hearing Research Foundation, the condition is most common in Whites. About 1 in 10 White adults develop otosclerosis, and White women are affected twice as frequently as men. Onset is between ages 10 and 45 years, but most common in the twenties. Although the etiology is unclear, otosclerosis has a hereditary component. Someone who has a parent with the condition has a 25% chance of developing otosclerosis.
Symptoms start with unilateral, low-frequency hearing loss, tinnitus, and possibly vertigo. Hearing loss gradually progresses in severity and eventually affects both ears. Hearing aids can help, but surgical treatment that replaces the bone with a prosthesis is the most effective intervention for improving hearing and reducing tinnitus.
Traumatic ossicular chain disruption
The ossicular chain (malleus, incus, and stapes) is subject to fracture or dislocation by direct head trauma from falls, blows to the head, or blast injuries. Damage to these bones—which can prevent adequate sound conduction to the inner ear—may be overlooked because of other more demanding injuries associated with the trauma. Temporal bone fractures have a high association with ossicular chain disruption. Treatment includes reconstruction of the ossicular chain.
Cholesteatoma
A cholesteatoma is a noncancerous tumor made up of keratinizing squamous epithelium in the middle ear. According to Jackson and colleagues, the condition in children commonly originates at birth (congenital cholesteatoma); however, acquired cholesteatoma affects 9 to 12 adults annually. Risk factors include recurrent AOM and TM perforation. Cholesteatoma pathogenesis isn’t well defined, but it is locally invasive and can erode the TM and ossicles.
Typically, cholesteatoma grows slowly over years and presents as gradual hearing loss, ear discharge, and eventual discomfort. Untreated cholesteatoma may advance into the inner ear, causing permanent sensorineural hearing loss. Early detection and surgical excision are key to restoring normal hearing.
Nursing implications
To decrease the risk of infection, TM perforation, and cerumen impaction, discourage patients from cleaning their ears with cotton tipped swabs, candling, or daily olive oil drops. Pain, cerumen impaction, and persistent ear discharge should be evaluated by a provider.
Persistent hearing loss that lasts more than 1 to 2 weeks after treatment of the underlying condition, including hearing loss after head trauma, should be re-evaluated for complications. Assess patients for a family history of hearing loss and refer those with gradual, progressive conductive hearing loss to an otolaryngologist to rule out serious causes such as otosclerosis and cholesteatoma.
Sensorineural hearing loss
Sensorineural hearing loss (SNHL) occurs when the mechanical sound that passes through the middle ear can’t be processed in the inner ear. It’s more complex than conductive hearing loss, and normally involves damage to the sensory nerve cells in the cochlea, the auditory nerve, or a combination of both.
Presbycusis
Age is the most significant factor associated with hearing loss in adults. According to the NIDCD, one in three adults age 65 to 75 years have age-related hearing loss; nearly half of those older than 75 report difficulty hearing. Approximately 15% of adults age 18 and older report some hearing loss, with the greatest amount of loss in those between 60 to 69. Presbycusis (age-related hearing loss) is a form of sensorineural hearing loss that can be caused by noise exposure, ototoxins (such as aminoglycoside antibiotics and the chemotherapeutic agent cisplatin), smoking, family history, and chronic illness. Presbycusis is thought to result from degenerative changes to the cochlear hair cells and auditory neurons from cumulative environmental insults.
Because symptoms start gradually and progress slowly, they may be overlooked. The hearing loss is symmetrical, bilateral, and most pronounced at high frequencies. As it progresses, patients have more difficulty understanding conversations, especially with background noise. Patients also may have more trouble understanding women and children than men as a result of high frequency loss. Presbycusis is linked to cognitive and physical decline, social isolation, depression, dementia, and decreased quality of life.
Adverse effects of presbycusis can be mitigated with hearing aids; however, more than 70% of patients who can benefit don’t use them, according to NIDCD data. Barriers to hearing aid use include cost, social stigma, lack of insurance coverage, and difficulty manipulating and adjusting to the device. For more profound hearing loss, surgically inserted cochlear implants, which bypass severely damaged hair cells to directly stimulate the auditory nerve, are an option. Hearing aids and cochlear implants can improve hearing, speech perception, social function, and overall well-being.
Noise-induced hearing loss
Noise-induced hearing loss, which affects one in four adults, is the second most common form of SNHL. Although presbycusis involves noise exposure, noise-induced hearing loss is considered a separate entity and may be the result of recreational exposure (loud music, firearms, motorcycles) or occupational exposure (aircraft, machinery, power tools). Both the intensity of the noise and duration of exposure create the risk for damage to the sensory hair cells in the cochlea. Louder sounds may be harmful with less duration, and even sounds that are “comfortably” loud may cause damage with longer durations.
Symptoms, which can be temporary or permanent, affect the higher frequencies and often include tinnitus. Noise-induced hearing loss can be treated with hearing aids.
Ménière’s disease
Ménière’s disease results from increased fluid in the vestibular system of the inner ear, which is responsible for balance. It affects 50 to 200 per 100,000 adults annually and is most prevalent in adults between age 40 and 60 years, according to a clinical practice guideline from the American Academy of Otolaryngology-Head and Neck Surgery (AAO-HNSF). Hearing loss, tinnitus, and recurring “attacks” of vertigo are the most common symptoms, but patients also may experience aural fullness, similar to a plugged ear. Hearing loss fluctuates from low to mid frequency and usually is unilateral. Vertigo may stabilize over time, but hearing loss tends to worsen; 82% of those with Ménière’s disease experience persistent moderate to severe hearing loss.
Management to reduce acute and long-term symptoms include lifestyle modifications (reduced salt, alcohol, stress) and medication (such as antihistamines, oral or intratympanic steroids, and diuretics). Hearing aids may improve hearing and quality of life.
Sudden sensorineural hearing loss
Sudden sensorineural hearing loss (SSNHL) may affect only 5 to 20 per 100,000 adults annually, according to a clinical practice guideline from AAO-HNSF, but it’s an otologic emergency that requires an urgent referral to an otolaryngologist. SSNHL etiology is unknown, but viral, autoimmune, and vascular causes have been suggested. The condition presents as acute, unilateral hearing loss with symptom onset in less than 72 hours with no precipitating event. Patients frequently report ear fullness and tinnitus. Vertigo also may occur, leading to misdiagnosis of Ménière’s disease. In addition, aural fullness may be erroneously attributed to fluid in the middle ear.
To conserve hearing, specialist treatment should be initiated urgently and may include oral or intratympanic steroids and hyperbaric oxygen therapy. Time from symptom onset to initial treatment should be no more than 2 weeks, although the exact time frame is debatable. Spontaneous recovery can occur; however, even with treatment, hearing loss and tinnitus may be permanent.
Acoustic neuroma
An acoustic neuroma (vestibular schwannoma) is a slow-growing benign tumor of the myelin-producing Schwann cells of the eighth cranial nerve. According to Goshtasbi and colleagues, this rare but serious tumor affects 1.1 to 1.7 per 100,000 people, mostly adults between 40 and 63 years. Patients typically present with a progressive, unilateral SNHL, but the hearing loss also may be sudden. Other symptoms may include tinnitus, unsteadiness, facial weakness, headache, and visual disturbance. Large tumors may compress the brain stem, causing gait abnormalities. Childhood cancer treatment radiation of the head is a risk factor.
Refer patients with suspected acoustic neuroma to an otolaryngologist. Treatment options include radiotherapy, surgery, and watchful waiting. The best treatment depends on multiple factors, such as tumor size, symptoms, patient age, and underlying health conditions.
Nursing implications
Encourage patients to use appropriate hearing protection to minimize the risk of SNHL. Assess patients for potentially hazardous noise exposure with the “shout test,” where a patient states they must shout to a person who is only an arm’s length away to be heard over background noise. The most common types of SNHL are gradual, bilateral, progressive, and high frequency, which means that symptoms initially may go unnoticed by the patient. Although presbycusis and noise-induced hearing loss are the most common types of SNHL, be aware of more serious causes and their associated symptoms.
To help distinguish SNHL from more innocuous conductive causes (such as fluid or wax), perform a quick screening with the “hum test.” Instruct the patient to hum and ask if they hear the hum louder in the affected ear (such as you would if you plugged your ear). If so, the hearing loss is most likely conductive rather than sensorineural. All unilateral hearing loss thought to be sensorineural requires audiology and specialty referral to rule out serious causes. Sudden SNHL is an otologic emergency. (See Screening for hearing loss.)
Take the opportunity
Hearing loss, whether it’s conductive or sensorineural, can affect a patient’s quality of life. Nurses have an opportunity to identify and screen patients, educate them about hearing conservation, offer treatment as ordered by a provider, coordinate referrals, and support long-term management, including hearing aid use when appropriate. AN
Tamara Link is board-certified family nurse practitioner and assistant professor at the University of North Carolina Wilmington.
References
Alzhrani F, Mokhatrish MM, Al-Momani MO, Alshehri H, Hagr A, Garadat SN. Effectiveness of stapedotomy in improving hearing sensitivity for 53 otosclerotic patients: Retrospective review. Ann Saudi Med. 2017;37(1):49-55. doi:10.5144/0256-4947.2017.49
American Hearing Research Foundation. Otosclerosis. americanhearing.org/disease/otosclerosis/
Basura GJ, Adams ME, Monfared A, et al. Clinical practice guideline: Ménière’s disease executive summary. Otolaryngol Neck Surg. 2020;162(4):415-34. doi:10.1177/0194599820909439
Bennett RJ, Fletcher S, Conway N, Barr C. The role of the general practitioner in managing age-related hearing loss: Perspectives of general practitioners, patients and practice staff. BMC Fam Pract. 2020;21(1):87. doi:10.1186/s12875-020-01157-2
Centers for Disease Control and Prevention. QuickStats: Percentage of adults Aged ≥18 years with any hearing loss, by state — National Health Interview Survey, 2014–2016. MMWR Morb Mortal Wkly Rep. 2017;66(50):1389. doi:10.15585/mmwr.mm6650a7
Chandrasekhar SS, Tsai Do BS, Schwartz SR, et al. Clinical practice guideline: Sudden hearing loss (update). Otolaryngol Head Neck Surg. 2019;161(suppl 1):S1-45. doi:10.1177/0194599819859885
Cunningham LL, Tucci DL. Hearing loss in adults. N Engl J Med. 2017;377(25):2465-73. doi:10.1056/NEJMra1616601
Foley RW, Shirazi S, Maweni RM, et al. Signs and symptoms of acoustic neuroma at initial presentation: An exploratory analysis. Cureus. 2017;9(11):e1846. doi:10.7759/cureus.1846
Fortunato S, Forli F, Guglielmi V, et al. A review of new insights on the association between hearing loss and cognitive decline in ageing. Acta Otorhinolaryngol Ital. 2016;36(3):155-66. doi:10.14639/0392-100X-993
Goshtasbi K, Abouzari M, Moshtaghi O, et al. The changing landscape of vestibular schwannoma diagnosis and management: A cross‐sectional study. Laryngoscope. 2020;130(2):482-6. doi:10.1002/lary.27950
Jackson R, Addison AB, Prinsley PR. Cholesteatoma in children and adults: Are there really any differences? J Laryngol Otol. 2018;132(7):575-8. doi:10.1017/S0022215118000890
Kühn AL, Chang Y-M. Traumatic incudomalleolar disarticulation. J Clin Neurosci. 2019;62:242-3. doi:10.1016/j.jocn.2018.12.017
Michaudet C, Malaty J. Cerumen impaction: Diagnosis and management. Am Fam Physician. 2018;98(8):525-9.
Michels TC, Duffy MT, Rogers DJ. Hearing loss in adults: Differential diagnosis and treatment. Am Fam Physician. 2019;100(2):98-108.
Mills R, Hathorn I. Aetiology and pathology of otitis media with effusion in adult life. J Laryngol Otol. 2016;130(5):418-24. doi:10.1017/S0022215116000943
National Institute on Deafness and Other Communication Disorders. Age-related hearing loss. July 17, 2018. www.nidcd.nih.gov/health/age-related-hearing-loss
National Institute on Deafness and Other Communication Disorders. NIDCD working group on accessible and affordable hearing health care for adults with mild to moderate hearing loss. June 23, 2016. www.nidcd.nih.gov/research/workshops/accessible-and-affordable-hearing-health-care/2009
Nguyen T, Pulickal G, Singh A, Lingam R. Conductive hearing loss with a “dry middle ear cleft”—A comprehensive pictorial review with CT. Eur J Radiol. 2019;
110:74-80. doi:10.1016/j.ejrad.2018.11.024
Nooni K, Sreekanth G. Otitis media with effusion: Comparative effectiveness of treatments. IOSR J Dent Med Sci. 2016;15(7):29-34. doi:10.9790/0853-150722934
Pagrani M, Srivastava A, Mohan C. Adult onset acute otitis media—A preliminary report. J Evol Med Dent Sci. 2014;3(19):5094-9. doi:10.14260/jemds/2014/2559
Rijk MH, Hullegie S, Schilder AGM, et al. Incidence and management of acute otitis media in adults: A primary care-based cohort study. Fam Pract. Published online January 28, 2021:cmaa150. doi:10.1093/fampra/cmaa150
Rosenfeld RM, Schwartz SR, Cannon CR, et al. Clinical practice guideline: Acute otitis externa. Otolaryngol Head Neck Surg. 2014;150(suppl 1):S1-24. doi:10.1177/0194599813517083
Schwartz SR, Magit AE, Rosenfeld RM, et al. Clinical practice guideline (update): Earwax (cerumen impaction). Otolaryngol Head Neck Surg. 2017;156(suppl 1):S1-29. doi:10.1177/0194599816671491
Wu H, Zhang L, Han D, et al. Summary and consensus in 7th International Conference on Acoustic Neuroma: An update for the management of sporadic acoustic neuromas. World J Otorhinolaryngol Head Neck Surg. 2016;2(4):234-9. doi:10.1016/j.wjorl.2016.10.002
Yang EL, Macy TM, Wang KH, Durr ML. Economic and demographic characteristics of cerumen extraction claims to Medicare. JAMA Otolaryngol Head Neck Surg. 2016;142(2):157-61. doi:10.1001/jamaoto.2015.3129