Distinguishing between the two conditions can help ensure prompt and appropriate treatment.
Learning Objectives
- Describe how to differentiate delirium and dementia and assess for delirium superimposed on dementia (DSD).
- Discuss risk factors for DSD and the three subtypes.
- Describe how to prevent DSD and manage individuals with the condition.
The authors and planners of this CNE activity have disclosed no relevant financial relationships with any commercial companies pertaining to this activity. See the last page of the article to learn how to earn CNE credit.
Expiration: 11/1/25
Takeaways:
- Delirium superimposed on dementia (DSD) occurs in 89% of older hospitalized adults.
- Avoid medications with high anticholinergic burden and antipsychotics.
- Reduced DSD-related mortality requires early recognition and treatment of the underlying cause.
The Diagnostic and Statistical Manual of Mental Disorders (DSM-5) describes delirium occurring in patients with Alzheimer’s disease and Alzheimer’s disease–related dementias (AD/
ADRDs) as delirium superimposed on dementia (DSD) and delirium superimposed on Alzheimer’s disease. Although delirium and dementia have distinctly different causes and presentations, clinicians (including bedside and advanced practice nurses) may have difficulty distinguishing between the two when they occur simultaneously, especially when the clinicians don’t know the patient’s baseline cognitive status.
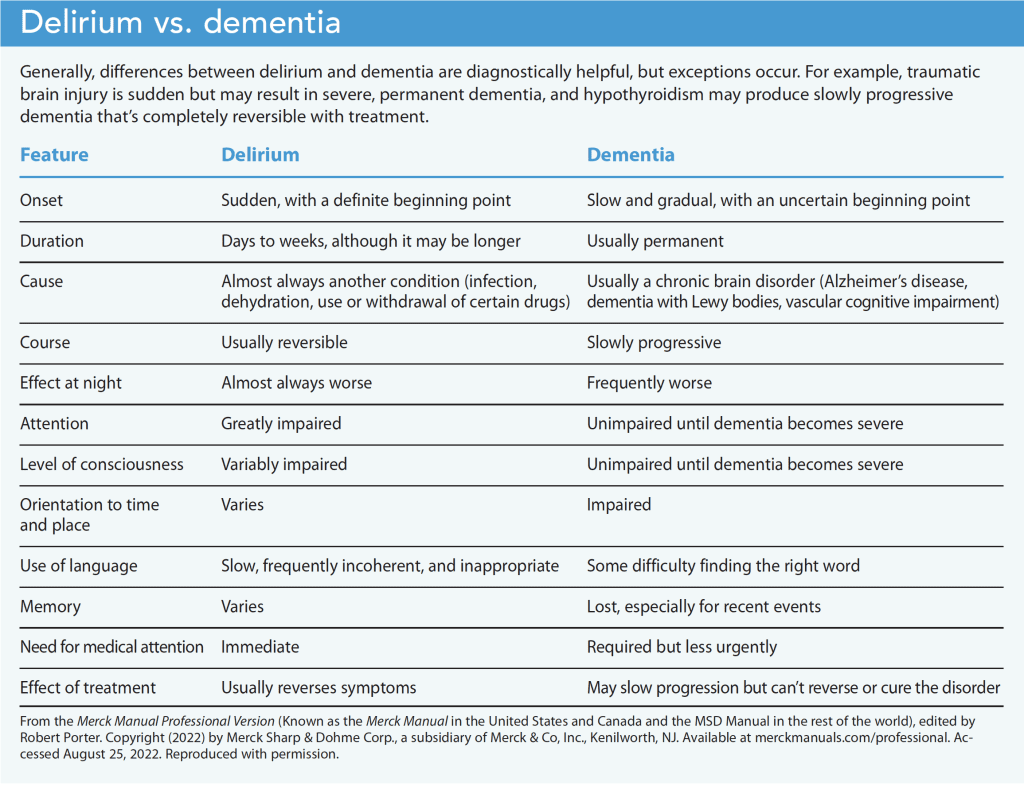
Differentiating delirium and dementia
Up to 89% of older adults experience delirium, an acute neuropsychiatric syndrome and clinical emergency that requires acute care. The condition frequently goes unrecognized in individuals with dementia. Delirium, a potentially reversible acute state, is characterized by abrupt changes from baseline. Patients may experience acute mental status changes, neuropsychiatric symptoms, and psychomotor dysfunction with onset within hours or days.
In contrast, the umbrella term dementia refers to several chronic neurocognitive syndromes that cause progressive neurodegenerative changes in one or more neurocognitive domains, including language, learning and memory, social cognition, complex attention, executive function, and perceptual–motor function. As dementia progresses, some patients develop neuropsychiatric symptoms that mimic delirium, but are substantially worsened by acute medical or physiologic changes. Delirium should be ruled out in the event of acute onset of neuropsychiatric symptoms, such as hallucinations, delusions, aggression, and sleep disturbances.
These behavioral and psychological symptoms of dementia also mimic the hallucinations and delusions common with delirium and might intensify in DSD. Notably, those with Lewy body dementia may experience the same symptoms as DSD (fluctuating course, inattention, sleep disturbances) and other neuropsychiatric symptoms (vivid visual hallucinations). These symptoms may worsen over weeks or months, which differentiates DSD from Lewy body dementia.
Most skilled professionals find DSD difficult to recognize and clinically challenging because of its presentation complexity and the lack of standardized diagnostic criteria that separate dementia characteristics from the delirium assessment. (See Delirium vs. dementia.)
DSD risk factors
Risk factors for DSD include a medical history of or acute onset of some medical conditions (for example, systemic or local infection [urinary tract or pressure injury infection], electrolyte disturbances, heart failure, coronary artery disease, stroke, or an acute exacerbation of pulmonary disease). Geriatric syndromes that may precipitate DSD include polypharmacy, depression, fecal impaction, pain, and sleep deprivation. Prescription and over-the-counter medications with high anticholinergic properties, sedatives, some calcium channel and beta antagonists, and psychotropics may cause or worsen delirium. Stressors associated with medical procedures, surgeries, or any related complications also increase the risk for DSD. Missed opportunities for prevention and early recognition compound the risk for poor outcomes, potentially life-threatening complications, and delirium-associated high mortality rates.
DSD subtypes
Knowing delirium subtypes (hypoactive, hyperactive, and mixed) can aid the initial diagnosis and help document progress in symptom resolution. Subtypes can fluctuate significantly, so perform assessments more than once daily and compare them across the previous 24 hours. Consider using the Richmond Agitation and Sedation Scale (RASS) to help identify subtypes.
Hypoactive DSD
Hypoactive DSD is characterized by one or more of the following characteristics: change within the past 24 hours with slowing or lack of movement, paucity of speech with or without prompting, difficulty arousing without auditory or tactile stimuli, or decreased responsiveness.
Case study: Charles, a 70-year-old man with a recent diagnosis of early-stage dementia, is admitted to the hospital after a fall at home. While in the emergency department (ED), he interacts appropriately with healthcare providers and nursing staff. Charles is oriented and cooperates with his care plan. Twelve hours after admission, he demonstrates inattention during conversations, frequently withdrawing and responding only partially to questions.
The nurse notices from the hand-off report that Charles is experiencing an acute change in cognition from his baseline. He can’t recite the days of the week backwards and is unable to recall life events from long-term memory. Charles’ RASS assessment scale is -2. The nurse contacts the healthcare provider to report an acute change in awareness, attention, and arousal indicative of DSD. The provider completes a delirium workup and identifies a metabolic disturbance. The hypoactive DSD resolves within 24 hours after correcting the disturbance.
Hyperactive DSD
Features of hyperactive DSD range from simple restlessness to constant movement, agitation, or combativeness. Visual and perceptual disturbances (hallucinations and delusions) may become more frequent and severe. Nurses are more likely to identify hyperactive DSD as a result of the interference it creates when providing care.
Case study: Mary, an 85-year-old woman with a medical history of moderate AD/ADRD, is admitted to the hospital with dehydration and an unstageable pressure injury of her coccyx. Three days after admission, Mary shows clinical improvement with a positive response to her plan of care, but the nurse notices an acute change in her behavior. In the past 24 hours, Mary is combative during care, hyperalert, experiencing new visual hallucinations, and inattentive. Her RASS score is +4 (combative).
The chart review shows that Mary had several new incontinent voiding episodes and poorly controlled pain. The nurses’ assessment indicates new suprapubic tenderness, and lab findings reveal a urinary tract infection. With antibiotic treatment and pain medication adjustments, Mary’s hallucinations, inattention, and arousal improve over the next 48 hours.
Mixed DSD
Mixed DSD manifests as flucuating delirium with RASS scores ranging from positive values (agitation, combative) to negative values (lethargic, stuporous) over 24 to 48 hours. The patient may be difficult to arouse in the morning but develops agitation and combativeness that night.
Case study: Jason, a 75-year-old man with a history of mid-stage AD/ADRD, arrives in the ED with an acute alteration in mental status and abdominal pain. His wife reports changes in mental status over the past 24 hours, which varied from confusion, disorientation, persistent sleepiness, restlessness, and agitation at night (RASS +4) to lethargy in the morning (RASS -2) with inattention not consistent with his baseline cognition.
A thorough assessment indicates a fecal impaction and fluid volume depletion. Over the next 48 hours, after the fecal impaction is removed and Jason receives I.V. fluids for 24 hours, his fluctuating mental status improves. Looking back at the electronic health record (EHR) for the past 24 hours can help prevent falsely attributing this subtype to a patient’s dementia.
Diagnosis
The DSM-5, the gold standard reference for delirium diagnostic criteria, doesn’t include robust guidance for DSD. In the delirium section, the DSM-5 notes that the acute onset and temporal course of delirium can be difficult to ascertain in older adults with prior neurocognitive disorders or AD/ADRD. In the neurocognitive disorders section, the DSM-5 addresses the variation in the course of AD/ADRD subtypes where the possibility of DSD should be considered. If healthcare professionals follow the DSM-5 criteria to consider delirium only when an acute onset or fluctuation from a pre-existing neurocognitive disorder occurs, they may not attribute changes in level of arousal, new onset of neuropsychiatric symptoms, or inattention to possible DSD.
To prevent misinterpretation, the DSM-5 delirium diagnostic criteria include a provision that the patient must have an additional acute (days to weeks) disturbance in cognition different from their baseline. Clinicians can determine a patient’s baseline cognition using collateral sources, ideally someone with intact cognition who knows the individual well. When in doubt, nurses and providers in acute care settings may consider consulting with a geriatric psychiatrist or a geriatrician. (See Delirium: Diagnostic criteria.)
Delirium: Diagnostic criteria
Diagnostic criteria for delirium include acute onset and disturbance in attention and arousal with an additional disturbance in one or more areas of cognition—memory, orientation, language, visual–spatial ability, and perception. An impaired state of arousal is a cardinal indicator for delirium, as is an acute change or reduction in awareness of one’s orientation to environment and attention. Changes in arousal include all states of altered arousal (except coma) in the continuum of delirium, from hyperarousal to stupor.
Psychological hallmarks of delirium include acute changes in perception, including hallucinations and delusions. Inattention criteria are met for individuals with AD/ADRD who are too drowsy to demonstrate inattention through testing and a change exists from baseline over a 24-hour period. Global attention is best assessed when the clinician interviewing the patient adds an element for vigilant attention, easily accomplished with a brief conversation to trigger long-term memory that should remain intact in those with AD/ADRD.
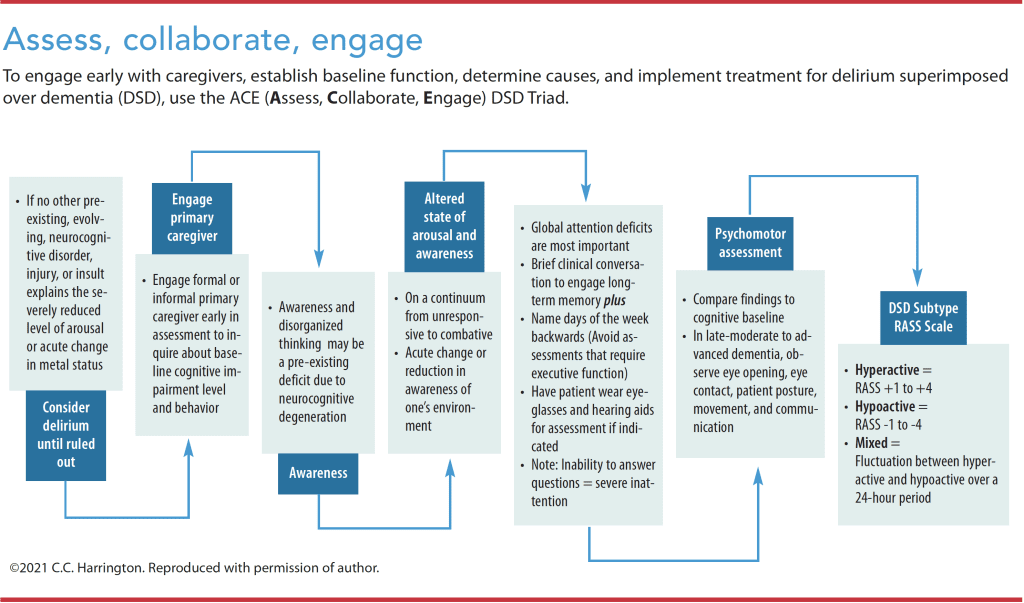
DSD triad assessment
DSD characteristics differ from delirium without dementia. The distinguishing features between behavior and psychological symptoms of dementia vs. DSD may be the acute onset and identification of causative triggers for cognition and function changes. Early engagement of formal or informal primary caregivers can help establish an accurate baseline cognitive assessment and prompt DSD diagnosis. Reliable DSD assessment reduces the need for medications and restraints that may result in injury to self or others and worsen DSD severity and duration.
An appropriate assessment of the DSD triad (Awareness, Arousal, and Attention) is key to early recognition and identification of the underlying causes. Long-term memory is preserved in AD/ADRD, so a brief conversation about social history (or other relevant topic) adds critical subjective data to the diagnostic criteria. However, levels of disturbance in arousal, awareness, and impairments in multiple cognitive domains are significantly more severe in patients with DSD. Disorganized thinking and lack of awareness may be a preexisting loss resulting from AD/ADRD neurocognitive deficits, so early conversations with primary caregivers can help identify cognitive changes from baseline.
Psychomotor retardation and inability to make or maintain eye contact, sustain posture, or communicate may be relevant awareness assessment findings in those with moderate-to-severe dementia. Use attention tests (such as naming the days of the week backwards) that don’t require executive function and accommodate those with hearing impairment. In those with late-moderate to advanced dementia, observe eye opening, eye contact, patient posture, movement, and communication using the Observational Scale of Level of Arousal. (See Assess, collaborate, engage.)
DSD causes
DSD can result from systemic illness, dehydration, medications (such as antipsychotics), pain, and sensory deprivation.
Systemic illness
Older adults experience immunosenescence, a gradual age-related deterioration of the immune system, which makes fever and elevated white blood cell counts poor indicators of acute illness. Although not causative, a correlation exists between the presence of urinary catheters and delirium prevalence. Fecal impaction and urinary retention also may cause delirium symptoms.
A thorough nursing assessment can help the provider narrow their differential diagnosis for potential causes of DSD, which might include a typical infection, medication changes, or dehydration. Atypical causes include myocardial infarction (particularly in those with diabetes who may not experience typical symptoms), acute pulmonary conditions, or poor glycemic control (individuals with DSD are at higher risk of hypoglycemia and hyperosmolality).
Dehydration
Older adults have a decreased thirst drive and are sensitive to fluid imbalances. Monitor intake and output, encourage oral fluids, and check I.V. rehydration to maintain fluid balance and avoid fluid overload. Identifying and encouraging oral fluids of preference to maintain hydration frequently is sufficient.
Medications
Medications, both those started and stopped, have the potential to precipitate DSD. Inadvertently discontinued medications, including significant opioid dosage reductions, may exacerbate pain or lead to withdrawal symptoms. High-risk medications known to increase DSD risk include opioids, psychoactive agents, antipsychotic medications in hypoactive DSD, and medications known to have a high anticholinergic burden such as diphenhydramine and amitriptyline.
Choose non-opioid medications when possible for patients with AD/ADRD; opioids may cause delirium, especially in opioid-naïve patients. Use calcium channel blockers and beta antagonists with caution as they’ve been linked to increased delirium risk. Avoid initiating cholinesterase inhibitors to prevent or treat DSD postoperatively, and don’t use benzodiazepines for agitation associated with AD/ADRD or hyperactive DSD. Melatonin, a neurotransmitter that improves circadian rhythms, has shown promise in reducing delirium incidence, but it’s not currently recommended as an evidence-based treatment.
Current evidence doesn’t support routine use of typical (first generation) or atypical antipsychotics to prevent or treat DSD. First-generation antipsychotics, as well as risperidone, have higher anticholinergic burdens that may worsen DSD. All antipsychotics have black box warnings for use in AD/ADRD because of increased risk of stroke, myocardial infarction, and death. If used, providers should seek and document risk–benefit conversations, including verbal consent for use from the patient’s responsible party. Consider monitoring for corrected QT (QTc) intervals.
Pain
Older adults with more advanced dementia may be unable to articulate pain, which predisposes them to DSD. Use self-report in combination with observation to provide the most reliable pain assessment. Assess for six behavioral domains—facial expression, vocalization, and body movement, as well as changes in interpersonal interactions, activity routine, and mental status. Preferred observational pain assessment tools for all stages of dementia include the Pain Assessment in Advanced Dementia (PAINAD) Scale and the Pain Assessment Checklist for Seniors with Limited Ability to Communicate (PACSLAC).
The five-item PAINAD scale measures breathing, negative vocalizations, facial expression, body language, and consolability in individuals with advanced dementia. Rate each behavior on a scale of 0 to 2, where 0 represents normal functioning and 2 represents behaviors suspected of indicating pain (noisy labored breathing, loud moaning or groaning, facial grimacing, rigidity, or inconsolability).
Complete the PACSLAC based on observations of the patient during activity or movement (such as transferring out of bed or walking). This screening tool assesses individualized responses over time, so the current score is compared to the previous score. An increased score suggests a likely increase in pain; a lower score indicates decreased pain.
Sensory deprivation
Patients with vision or hearing impairments should wear their glasses and hearing aids as much as possible. If patients with DSD have hearing impairment but don’t have a hearing aid, use a pocket amplifier to improve communication. Identify and allow one designated family member at the bedside for social engagement and to reduce sensory deprivation. Lack of sleep also may trigger DSD. Ask the patient or a family member about personal preferences for sensory engagement activities and sleep routines.
Nonpharmacologic nursing interventions
Maintain a high index of suspicion for DSD in patients with a history of dementia and implement nonpharmacologic nursing interventions to help reduce its incidence and prevalence. Effective nonpharmacologic interventions include establishing and maintaining the patient’s sleep regimen and routine, ensuring they wear eyeglasses and hearing aids as needed, assisting with feeding and hydration if indicated, promoting early mobilization with reduced time in bed, providing patient-centered engagement, and educating family members about delirium.
Your primary goals include identifying and managing causative factors as soon as possible and maintaining safety precautions. In acute care and long-term facilities, that includes hourly rounding, frequent toileting, assisting with ambulation, encouraging family presence, and avoiding restraints.
Safeguard vulnerable patients
Early recognition and communication are key to DSD identification and treatment. Communicating with the healthcare team will prompt further assessments for treatable causes, and conversations with formal or informal primary caregivers can help determine the patient’s baseline cognitive status. These critical source reports can come from a family caregiver, primary care provider, a nurse, or the EHR.
Avoiding increased morbidity, discharge to higher levels of care, and death in hospitalized patients with dementia requires that nurses have a high index of suspicion for DSD. Nurses serve as the first line of defense against DSD and have the best opportunity to safeguard these vulnerable patients using evidence-based interventions.
Candace C. Harrington is an assistant professor and gerontology NP professor at the University of Louisville School of Nursing in Louisville, Kentucky. Ardis M. Roederer is an acute care NP at Central Baptist Hospital in Lexington, Kentucky. Hope K. Eppley is a recent doctor of nursing practice graduate from the University of Louisville School of Nursing family NP track. Pamela Z. Cacchione is a professor of geropsychiatric nursing and Ralston Endowed Term Chair in Gerontological Nursing at the University of Pennsylvania School of Nursing in Philadelphia, a nurse Scientist at the Penn Presbyterian Medical Center, and a senior fellow at the Leonard Davis Institute of Health Care Economics University of Pennsylvania.
References
Adeola M, Azad R, Kassie GM, et al. Multicomponent interventions reduce high-risk medications for delirium in hospitalized older adults. J Am Geriatr Soc. 2018;66(8):1638-45. doi:10.1111/jgs.15438
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 5th ed. Washington D.C.: American Psychiatric Publishing; 2013.
Apold S. Delirium superimposed on dementia. J Nurse Pract. 2018;14(3):183-9. doi:10.1016/j.nurpra.2017.12.010
Avelino-Silva TJ, Campora F, Curiati JAE, Jacob-Filho W. Association between delirium superimposed on dementia and mortality in hospitalized older adults: A prospective cohort study. PLOS Med. 2017;14(3):e1002264. doi:10.1371/journal.pmed.1002264
Fick DM, Agostini JV, Inouye SK. Delirium superimposed on dementia: A systematic review. J Am Geriatr Soc. 2002;50(10):1723-32. doi:10.1046/j.1532-5415.2002.50468.x
Fuchs-Lacelle S, Hadjistavropoulos T. Development and preliminary validation of the pain assessment checklist for seniors with limited ability to communicate (PACSLAC). Pain Manag Nurs. 2004;5(1):37-49. doi:10.1016/j.pmn.2003.10.001
Goebel JR, Ferolito M, Gorman N. Pain screening in the older adult with delirium. Pain Manag Nurs. 2019;20(6):519-25. doi:10.1016/j.pmn.2019.07.003
Huang J. Overview of delirium and dementia. Merck Manual: Professional Version. March 2021. merckmanuals.com/professional/neurologic-disorders/delirium-and-dementia/overview-of-delirium-and-dementia#:~:text=Overview
León-Salas B, Trujillo-Martín MM, Martínez Del Castillo LP, et al. Multicomponent interventions for the prevention of delirium in hospitalized older people: A meta-analysis. J Am Geriatr Soc. 2020;68(12):2947-54. doi:10.1111/jgs.16768
Morandi A, Grossi E, Lucchi E, et al. The 4-DSD: A new tool to assess delirium superimposed on moderate to severe dementia. J Am Med Dir Assoc. 2021;22(7):1535-42. doi:10.1016/j.jamda.2021.02.029
Morandi A, Zambon A, Di Santo SG, et al. Understanding factors associated with psychomotor subtypes of delirium in older inpatients with dementia. J Am Med Dir Assoc. 2020;21(4):486-92. doi.10.1016/j.jamda.2020.02.013
Nikooie R, Neufeld KJ, Oh ES, et al. Antipsychotics for treating delirium in hospitalized adults: A systematic review. Ann Intern Med. 2019;171(7):485-95. doi:10.7326/M19-1860
Nitchingham A, Caplan GA. Current challenges in the recognition and management of delirium superimposed on dementia. Neuropsychiatr Dis Treat. 2021;17:1341-52. doi:10.2147/NDT.S247957
Mulkey MA, Olson DM, Hardin SR. Top four evidence-based nursing interventions for delirium. Medsurg Nurs. 2019;28(6):357-62. doi:10.2147/NDT.S247957
Oudewortel L, Joling KJ, Hertogh CMPM, Wijnen VJM, van der Brug AAM, van Gool WA. Performance on bedside tests of attention and organized thinking in patients with dementia free from delirium. Inter Psychogeriatr. 2018;31(1):73-81. doi:10.1017/s1041610218000522
Parrish E. Delirium superimposed on dementia: Challenges and opportunities. Nurs Clin North Am. 2019;54(4):541-50. doi:10.1016/j.cnur.2019.07.004
Quispel-Aggenbach DWP, Holtman GA, Zwartjes HAHT, Zuidema SU, Luijendijk HJ. Attention, arousal and other rapid bedside screening instruments for delirium in older patients: A systematic review of test accuracy studies. Age Ageing. 2018;47(5):644-653. doi:10.1093/ageing/afy058
Saravana-Bawan B, Warkentin LM, Rucker D, Carr F, Churchill TA, Khadaroo RG. Incidence and predictors of postoperative delirium in the older acute care surgery population: A prospective study. Can J Surg. 2019;62(1):33-8. doi:10.1503/cjs.016817
Steensma E, Zhou W, Ngo L, et al. Ultra-brief screeners for detecting delirium superimposed on dementia. J Am Med Dir Assoc. 2019;20(11):1391-6. doi:10.1016/j.jamda.2019.05.011
Tieges Z, McGrath A, Hall RJ, Maclullich AMJ. Abnormal level of arousal as a predictor of delirium and inattention: An exploratory study. Am J Geriatr Psychiatry. 2013;21(12):1244-53. doi:10.1016/j.jagp.2013.05.003
van Velthuijsen EL, Zwakhalen SMG, Mulder WJ, Verhey FRJ, Kempen GIJM. Detection and management of hyperactive and hypoactive delirium in older patients during hospitalization: A retrospective cohort study evaluating daily practice. Int J Geriatr Psychiatry. 2018;33(11):1521-9. doi:10.1002/gps.4690
Yevchak A, Fick DM, Kolanowski AM, et al. Implementing nurse-facilitated person-centered care approaches for patients with delirium superimposed on dementia in the acute care setting. J Gerontol Nurs. 2017;43(12):21-8. doi:10.3928/00989134-20170623-01
American Nurse Journal 2022; 17 (11). Doi: 10.51256/ANJ112206