Early identification and management are key to good outcomes.
Learning Objectives
- Compare onset, diagnosis, and treatment of diabetic ketoacidosis (DKA) and hyperglycemic hyperosmolar state.
- Identify special considerations for preventing DKA recurrence.
The author and planners of this CNE activity have disclosed no relevant financial relationships with any commercial companies pertaining to this activity. See the last page of the article to learn how to earn CNE credit.
Expiration: 9/1/24
Diabetic ketoacidosis (DKA) and hyperglycemic hyperosmolar state (HHS) are endocrine emergencies. (See DKA and HHS: Head-to-head comparison.) Hospitalizations for diabetes and DKA are rising, possibly because of increased diabetes prevalence and higher insulin costs. HHS is associated with high mortality risks, which is particularly worrisome during the COVID-19 pandemic, when healthcare resources have been stretched thin. To help ensure early identification and treatment of these conditions, this article provides an overview of both. (See Diabetes physiology review.)
DKA and HHS: Head-to-head comparison
This side-by-side comparison highlights the differences and similarities between diabetic ketoacidosis (DKA) and hyperglycemic hyperosmolar state (HHS).
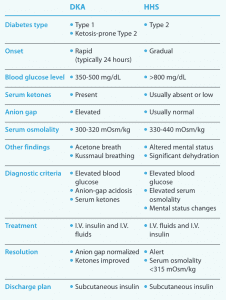

DKA
DKA typically occurs in patients with Type 1 diabetes, although prevalence in those with Type 2 diabetes is rising (accounting for an estimated 1 in 5 cases). To diagnose DKA, these three elements must be present: diabetes (hyperglycemia), ketones (serum), and anion-gap acidosis. In addition, DKA must be distinguished from alcoholic ketoacidosis (which typically occurs without hyperglycemia), starvation ketosis (which occurs with prolonged starvation or removal of carbohydrates from the diet), and other causes of anion-gap metabolic acidosis such as lactic acidosis, advanced kidney disease, or excessive ingestion of aspirin, acetaminophen, or methanol.
DKA is the result of a lack of insulin (or relative lack of insulin, when the normal insulin dose isn’t enough to provide adequate glycemic control, such as when the individual is ill or taking a prescribed steroid) in conjunction with increased counter-regulatory hormones, such as cortisol, and peripheral insulin resistance. When muscle and fat cells can’t use carbohydrates for energy, they produce amino acids and free fatty acids, leading to gluconeogenesis in the liver and ketone production, resulting in hyperglycemia, ketosis, dehydration, and electrolyte imbalances.
In some patients, DKA may be the initial indication of diabetes. In those with longstanding diabetes, DKA may be the result of missed insulin doses or a relative lack of insulin (for example, taking usual insulin doses during times of stress such as illness, infection, steroid use, or new myocardial infarction or stroke). DKA also can occur because of an insulin pump malfunction (such as a tubing kink) or inserting into a site with poor insulin absorption. Patients with eating disorders may withhold insulin doses to create a catabolic state that leads to weight loss (taking insulin typically causes weight gain), leading to DKA. In addition, the oral diabetes medications sodium glucose-cotransporter 2 inhibitors (SGLT-2 inhibitors) have been associated with increased risk for DKA, especially in patients who also are taking insulin. The cause may be a combination of an increased loss of glucose through the kidneys, decreased insulin needs, and increased insulin resistance from underlying stress or illness.
Signs and symptoms
DKA typically evolves rapidly with initial signs and symptoms of polyuria, polydipsia, fatigue, abdominal pain, nausea, and vomiting. Over time, mental status changes may arise, leading to coma. During a physical exam, a person with DKA typically shows signs of dehydration, including tachycardia and hypotension, with deep, rapid Kussmaul breathing and a fruity smelling acetone odor on the breath.
Diabetes physiology review
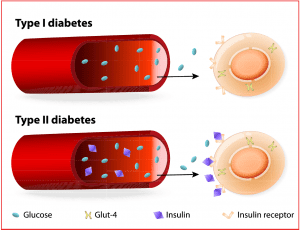

The ß cells of the pancreas produce insulin, which works in the liver, muscle, and adipose tissue to maintain glucose homeostasis, promoting peripheral glucose uptake. Insulin also prevents fatty acids and amino acids from converting into keto acids and glucose (gluconeogenesis). When insulin production is inhibited, Type 1 or Type 2 diabetes may result.
- Type 1 diabetes is characterized by T lymphocytes infiltrating the pancreatic islet cells, resulting in ß cell destruction and inhibited insulin production. The liver, muscle, and adipose tissue can’t absorb nutrients while continuing to release stored glucose, amino acids, and fatty acids into the bloodstream, altering fat metabolism and producing ketones. The continued absence (or relative absence) of insulin leads to hyperglycemia and ketoacidosis, which characterize diabetic ketoacidosis (DKA).
- Type 2 diabetes is a state of insulin resistance with ß cells initially increasing insulin production to meet demand. However, prolonged insulin resistance makes this unsustainable, and ß cell function begins to decline, worsening hyperglycemia. Type 2 diabetes also is associated with abnormal fat metabolism and increased glucose production from the liver. Ketosis is rare in Type 2 diabetes, but the prevalence of ketosis-prone Type 2 diabetes is rising. It’s characterized by relapsing episodes of ß cell dysfunction and absolute insulin deficiency, especially in times of extreme stress. In those instances, some people with Type 2 diabetes may experience DKA. They also may experience a hyperglycemic hyperosmolar state.
Lab findings
Laboratory findings include hyperglycemia (blood glucose typically >250 mg/dL and frequently 350 to 500 mg/dL) with low bicarbonate (<15 mEq/L) and an elevated anion gap (>12). Both urine and serum ketones are elevated, as is ß-hydroxybutyrate. If the patient is dehydrated, blood urea nitrogen and creatinine will be elevated. Serum potassium typically is elevated, with hyperkalemia occurring as potassium ions shift outside the cells as a result of acidosis. A total potassium deficit usually occurs in DKA because of acidosis, GI losses, and osmotic diuresis. When potassium is low, the patient should be monitored closely, as hypokalemia can lead to life-threatening arrhythmias. Sodium also may be low, resulting from losses caused by increased urine output and vomiting, although hyperglycemia itself can lead to pseudohyponatremia. However, because high blood glucose levels may cause a falsely low sodium level (many electronic health records systems have a calculator to determine the actual sodium level based on blood glucose level), the sodium level may need to be corrected. Hyperventilation may cause low pH (6.9 to 7.2) and low PCO2 (15 to 20 mm Hg).
Management
DKA treatment goals include restoring volume status and peripheral perfusion, improving glycemic control, and correcting electrolyte imbalances and ketosis. This is done by administering I.V. regular insulin and I.V. fluids. In addition, underlying causes must be identified and treated.
I.V. regular insulin begins with a bolus of 0.1 units/kg followed by an infusion at 0.1 units/kg/hr. Hypokalemia (<3.5 mEq/L) is an exception. Because insulin will worsen hypokalemia, potassium should be replaced to >3.5 mEq/L before starting I.V. insulin. Check blood glucose hourly and expect it to decrease by 50 to 75 mg/dL/hr. As acidosis resolves, blood glucose levels will decline more quickly. Standardized I.V. regular insulin infusion protocols, including computerized protocols, have proven safe and effective. However, protocols specific to DKA are recommended because those used to correct hyperglycemia in Type 2 diabetes may be too aggressive.
Because DKA is a state of dehydration, with estimated deficits of 4 to 6 liters, I.V. fluid replacement is key. Normal saline is the starting fluid of choice, typically at a rate of 1 L/hr for the first 2 to 3 hours. After the first 2 liters, decrease the rate to 300 to 400 mL/hr. When the patient’s blood glucose is <250 mg/dL, change to I.V. fluids containing 5% dextrose. The addition of dextrose counterbalances the insulin infusion, which needs to continue until acidosis is corrected, as indicated by normalization of bicarbonate and anion gap. In addition, the dextrose infusion reduces the risk of cerebral edema, which can occur when blood glucose is corrected too rapidly.
In most cases, the initial electrolyte imbalances should resolve on their own with I.V. regular insulin and I.V. fluids. Potassium may need to be replaced if it’s low or drops too quickly while insulin infuses. Treatment with sodium bicarbonate is controversial. It may lead to hypokalemia and impaired tissue oxygenation if it corrects acidosis too rapidly or to cerebral acidosis when cerebrospinal fluid pH is lowered. Avoid sodium bicarbonate except in cases of severe acidosis (pH <7.0) or significantly elevated potassium. Additionally, although phosphate typically is low in DKA, replacement is rarely needed except in severe cases (<1 mg/dL). (See Treating DKA outside of the ICU.)
Treating DKA outside of the ICU
Managing diabetic ketoacidosis (DKA) in the ICU with I.V. regular insulin remains the standard of care, but it has been shown (including in a systematic review by Andrade-Castellanos and colleagues) that uncomplicated, mild-to-moderate DKA (pH ≥7.0, serum bicarbonate 10-18 mEq/L, anion gap >12, and alert or drowsy mental status) can be safely and effectively managed outside of the ICU using subcutaneous rapid-acting insulin, typically administered every 2 hours. No significant difference was shown to exist between the I.V. regular insulin group and the subcutaneous rapid-acting insulin group in time to DKA resolution or in rates of hypoglycemia.
These studies typically excluded pregnant patients and those with hypotension or underlying cardiac, hepatic, or renal disease. Those patients would be more safely treated in an ICU setting with I.V. regular insulin.
Nurse staffing, education, and resources should be allocated to help ensure a smooth transition to treating DKA outside of the ICU when appropriate. This treatment option may help reduce healthcare costs and save ICU resources.
Resolution and transition to subcutaneous insulin
DKA is considered resolved with ketoacidosis correction, not improved glycemic control alone. The American Diabetes Association considers DKA resolved when two of the following occur: anion gap normalized (<12 mEq/L), bicarbonate ≥15 mEq/L, and pH >7.3. In addition, the patient’s mental status should be improved and the patient should be ready to eat. Most importantly, to prevent DKA recurrence, the healthcare team should identify and treat any precipitating causes.
After DKA resolves, the patient can be transitioned back to subcutaneous insulin, either in the form of multiple daily injections or a home insulin pump. Most subcutaneous insulins have an onset of action from several minutes to several hours, but I.V. regular insulin has an estimated half-life of <10 minutes, so subcutaneous insulin must overlap with I.V. regular insulin infusion by 1 to 2 hours. If this isn’t done, DKA may recur.
Patients whose diabetes is normally well-controlled but have experienced DKA because of an accidentally missed insulin dose can resume home insulin doses. However, for patients with an underlying illness or who must take steroids, home insulin doses may be inadequate to cover increasing demands. These patients may require higher insulin doses, which the provider will calculate by extrapolating from the previous 6 hours of stable I.V. regular insulin infusion rates.
Generally, the total daily insulin requirement for someone with Type 1 diabetes is 0.5 units/kg with half as long-acting basal insulin and half divided into three rapid-acting bolus insulin doses to cover meals. Therefore, caution should be used when re-starting what is documented as a home insulin dose. Re-starting a dose that seems inordinately high relative to weight and I.V. regular insulin needs may result in hypoglycemia.
A normally functioning pancreas secretes a basal dose of insulin, even in resting and fasting states, and releases insulin boluses within 8 to 10 minutes of ingesting food. When patients are transitioning from I.V. regular insulin, in addition to basal insulin, they need rapid-acting insulin boluses to cover meals. These generally are administered in the form of an insulin-to-carbohydrate ratio (for example, 1 unit of rapid-acting insulin for every 15 grams of carbohydrates consumed).
Rapid-acting insulin to cover meals is separate from correction-dose or sliding-scale insulin, which also should be in place and used to correct for hyperglycemia and to bring an elevated blood glucose level back within target range. Basal insulin and sliding-scale rapid-acting insulin without meal-dependent insulin won’t provide adequate glycemic control in someone with Type 1 diabetes who’s eating. More importantly, sliding-scale insulin alone without basal insulin will result in a return to DKA. Basal insulin should never be withheld in patients with Type 1 diabetes because they have an absolute lack of endogenous insulin.
Recurrent DKA and prevention
DKA prevalence is increasing worldwide, but a significant percentage of hospital admissions represents recurrent DKA with multiple hospital admissions in a calendar year.
Most of these cases are caused by missed insulin doses rather than underlying illnesses. A number of factors are associated with recurrent DKA, including fragmented healthcare (often involving admission to multiple hospitals), lower education levels, lower socioeconomic status, lack of health insurance, younger age, substance use, and other psychosocial issues. In many cases, those with recurrent DKA have poor access to insulin and transportation. Studies by Desai and colleagues, Ehrmann and colleagues, and Gaffney and colleagues also point to recurrent DKA occurring more often in females than males, which may be related to eating disorders, missing insulin doses in an effort to lose weight, and other mental health concerns such as depression.
To reduce hospital admissions, DKA prevention strategies, especially for patients with multiple admissions, should be a component of care. Improved patient and family education to help reduce hospital admissions and care coordination across the healthcare spectrum are key to prevention. Nurses can help patients obtain access to insulin (particularly longer-acting basal insulins that allow for more dosing flexibility) and diabetes technology (such as insulin pumps and continuous glucose monitoring). In addition, individual and family behavioral health coaching can help meet specialized needs such as addressing specific cultural or family concerns related to healthcare beliefs, foods, and periods of fasting. As virtual appointments and telemedicine play an increasing role in healthcare, regular communication and follow-up between patients with recurrent DKA and healthcare providers can reduce admissions and improve healthcare engagement. Virtual visits also may be helpful for those with transportation concerns or who live in areas with limited access to healthcare.
In younger adults, the transition from pediatric to adult care can be challenging and frequently is associated with a period of declining glycemic control. For older pediatric patients, smooth care transitions can be achieved by increasingly involving them in decision-making, including scheduling appointments and providing referrals to new providers and other individualized resources.
Special considerations
Several considerations, including insulin pumps, euglycemic DKA, and pregnancy, require special attention to prevent DKA recurrence.
Insulin pumps. DKA in someone who wears an insulin pump commonly occurs because of a disturbance (kinked or dislodged tubing or air bubbles) in insulin delivery. In addition, patients with scar tissue or lipohypertrophy may place their insulin pump site in an area of poor insulin absorption.
After DKA resolves, the patient should resume an insulin pump with a new infusion set, new insertion site, and new reservoir of insulin. Nurses should teach the patient to change injection sites and supplies according to manufacturer recommendations, which typically is every 2 to 3 days. They also should remind patients to make changes only when they can closely monitor their glycemic control. These patients should have a supply of basal insulin and rapid acting insulin to use via injection if they encounter problems with the pump or supplies.
Patients should be told that if they have any concern about the pump, they shouldn’t use it until they’ve confirmed that it’s working safely and effectively. In the meantime, they should use multiple daily insulin injections until the pump can be resumed.
Euglycemic DKA. DKA typically is defined by hyperglycemia, but euglycemic DKA may occur with normal or near-normal blood glucose levels. This is particularly true in cases of pregnant patients; starvation; insulin administration soon before hospital arrival; or those taking SGLT-2 inhibitors, which lower the renal threshold for glucose excretion through the urine.
With normal or near-normal blood glucose levels, euglycemic DKA may be easy to miss but should be considered in patients whose other lab results, particularly ß-hydroxybutyrate and arterial pH, point to DKA or if the patient has anion-gap acidosis that doesn’t resolve as expected.
Treatment for euglycemic DKA is the same as for DKA with hyperglycemia: I.V. regular insulin and I.V. fluids. If the patient is taking SGLT-2 inhibitors, the medication should be stopped. To prevent euglycemic DKA, patients taking SGLT-2 inhibitors shouldn’t stop or significantly decrease insulin doses and they should avoid excessive alcohol intake or ketogenic (low-carbohydrate) diets. Before any scheduled surgery or stressful physical activities, patients should stop taking SGLT-2 inhibitors.
Pregnancy. Pregnancy can result in increased insulin resistance, so women with pre-existing diabetes may require more insulin, particularly as their pregnancy progresses. Missing insulin doses or failing to adequately adjust insulin in response to increasing insulin resistance can result in significant hyperglycemia and DKA.
In addition, ketogenesis increases during pregnancy, so DKA can occur at a lower glucose level than in nonpregnant individuals. Pregnant women with underlying diabetes should have access to ketone strips for self-monitoring.
Treating DKA in pregnancy includes I.V. regular insulin and I.V. fluids, but higher insulin doses and fluids with higher dextrose content may be needed, particularly later in pregnancy. DKA is associated with a high rate of stillbirth, so fetal monitoring is recommended.
HHS
HHS occurs in the presence of hyperglycemia, hyperosmolality, and dehydration but without ketosis. It typically occurs in older individuals with Type 2 diabetes and in those with underlying stressors such as stroke, cardiac disease, or pneumonia. High carbohydrate intake, such as through tube feeding or I.V. nutrition, can lead to HHS, as can some medications, such as steroids and thiazide diuretics.
HHS is caused by a relative lack of insulin paired with inadequate fluid intake, leading to hyperglycemia, which then causes osmotic diuresis and further volume depletion. Lack of ketosis likely is due to only a relative, rather than an absolute, lack of insulin, which reduces the development of ketones. Because HHS typically occurs in older individuals with underlying comorbidities, it has a significantly higher mortality rate than DKA.
Signs and symptoms
HHS usually develops more slowly than DKA does. Patients may experience several weeks of rising glucose levels, polyuria, polydipsia, and weakness along with a declining mental status. Signs of dehydration (including dry mucous membranes), tachycardia, and orthostasis are evident on physical exam.
Lab findings
Lab findings include significant hyperglycemia (often >800 mg/dL). Initially, sodium may appear low, but as dehydration worsens, sodium rises, leading to elevated serum osmolality (from 330 to 440 mOsm/kg; normal is 280 to 295 mOsm/kg). Ketones usually are absent in HHS, although they may be present in a fasting state. Acidosis typically doesn’t occur unless a patient has an underlying condition such as sepsis, myocardial infarction, or renal failure, all of which may present as lactic acidosis rather than DKA.
Management
I.V. fluids and I.V. insulin are key components in HHS management. However, because of significant dehydration with a free water deficit of several liters, I.V. fluids play a more significant role and typically less insulin is required than when treating DKA. In some cases, fluids alone can significantly reduce blood glucose levels. Unless serum ketosis is present, I.V. regular insulin can be delayed. In most cases, 0.9% normal saline is initiated, transitioning to 5% dextrose with 0.45% normal saline after blood glucose reaches <250 mg/dL.
As in DKA, standardized insulin protocols for I.V. regular insulin infusion are recommended, and patients should be transitioned to subcutaneous insulin after blood glucose levels, electrolytes, fluid volume status, and mental status have improved. In addition, subcutaneous insulin should be continued on initial discharge home before later transitioning back to oral diabetes agents. As in DKA, close, early outpatient follow-up is needed.
Prevent future admissions
Both DKA and HHS are emergencies characterized by hyperglycemia, dehydration, and electrolyte abnormalities. Fluids and insulin are the mainstays of treatment. As hospital admissions for both conditions continue to increase, early identification and treatment can reduce length of stay and healthcare costs. In addition, individualized education and improved care coordination can help prevent future admissions.
References
American Diabetes Association. Children and adolescents: Standards of medical care in diabetes—2021. Diabetes Care. 2021;44(suppl 1):S180-99. doi:10.2337/dc21-S013
American Diabetes Association. Diabetes care in the hospital: Standards of medical care in diabetes—2021. Diabetes Care. 2021;44(suppl 1):S211-20. doi:10.2337/dc21-S015
American Diabetes Association. Management of diabetes in pregnancy: Standards of medical care in diabetes—2021. Diabetes Care. 2021;44(suppl 1):S200-10. doi:10.2337/dc21-S014
Andrade-Castellanos CA, Colunga-Lozano LE, Delgado-Figueroa N, Gonzalez-Padilla DA. Subcutaneous rapid-acting insulin analogues for diabetic ketoacidosis. Cochrane Database Syst Rev. 2016;(1):CD011281. doi:10.1002/14651858.CD011281.pub2
Benoit SR, Hora I, Pasquel FJ, Gregg EW, Albright AL, Imperatore G. Trends in emergency department visits and inpatient admissions for hyperglycemic crises in adults in the U.S., 2006-2015. Diabetes Care. 2020;43(5):1057-64. doi:10.2337/dc19-2449
Desai D, Mehta D, Mathias P, Menon G, Schubart UK. Health care utilization and burden of diabetic ketoacidosis in the U.S. over the past decade: A nationwide analysis. Diabetes Care. 2018;41(8):1631-8. doi:10.2337/dc17-1379
Ehrmann D, Kulzer B, Roos T, Haak T, Al-Khatib M, Hermanns N. Risk factors and prevention strategies for diabetic ketoacidosis in people with established type 1 diabetes. Lancet Diabetes Endocrinol. 2020;8(5):436-46. doi:10.1016/S2213-8587(20)30042-5
Evans K. Diabetic ketoacidosis: Update on management. Clin Med (Lond). 2019;19(5):396-8. doi:10.7861/clinmed.2019-0284
Everett E, Mathioudakis NN. Association of socioeconomic status and DKA readmission in adults with type 1 diabetes: Analysis of the US National Readmission Database. BMJ Open Diabetes Res Care. 2019;7(1):e000621. doi:10.1136/bmjdrc-2018-000621
Fayfman M, Pasquel FJ, Umpierrez GE. Management of hyperglycemic crises: Diabetic ketoacidosis and hyperglycemic hyperosmolar state. Med Clin North Am. 2017;101(3):587-606. doi:10.1016/j.mcna.2016.12.011
Flores M, Amir M, Ahmed R, et al. Causes of diabetic ketoacidosis among adults with type 1 diabetes mellitus: Insulin pump users and non-users. BMJ Open Diabetes Res Care. 2020;8(2):e001329. doi:10.1136/bmjdrc-2020-001329
Gaffney A, Christopher A, Katz A, et al. The incidence of diabetic ketoacidosis during “emerging adulthood” in the USA and Canada: A population-based study. J Gen Intern Med. 2019;34(7):1244-50. doi:10.1007/s11606-019-05006-6
Gardner DG. Endocrine emergencies. In: Gardner DG, Shoback D, eds. Greenspan’s Basic and Clinical Endocrinology. 10th ed. New York, NY: McGraw Hill Education; 2017; 783-807.
Handelsman Y, Henry RR, Bloomgarden ZT, et al. American Association of Clinical Endocrinologists and American College of Endocrinology position statement on the association of SGLT-2 inhibitors and diabetic ketoacidosis. Endocr Pract. 2016;22(6):753-62. doi:10.4158/EP161292.PS
Herrick CJ, McGill JB. Type 2 diabetes mellitus. In: Baranski TJ, McGill JB, Silverstein JM, De Fer TM, Ciesielski TM, eds. The Washington Manual: Endocrinology Subspecialty Consult. 4th ed. Philadelphia, PA: Wolters Kluwer; 2020; 278-94.
Hughes JW, McGill JB. Type 1 diabetes mellitus. In: Baranski TJ, McGill JB, Silverstein JM, De Fer TM, Ciesielski TM, eds. The Washington Manual: Endocrinology Subspecialty Consult. 4th ed. Philadelphia, PA: Wolters Kluwer; 2020; 263-77.
Masharani U, German MS. Pancreatic hormones and diabetes mellitus. In: Gardner DG, Shoback D, ed. Greenspan’s Basic and Clinical Endocrinology. 10th ed. New York, NY: McGraw Hill Education; 2018; 595-682.
Mays JA, Jackson KL, Derby TA, et al. An evaluation of recurrent diabetic ketoacidosis, fragmentation of care, and mortality across Chicago, Illinois. Diabetes Care. 2016;39(10):1671-6. doi:10.2337/dc16-0668
Tran TTT, Pease A, Wood AJ, et al. Review of evidence for adult diabetic ketoacidosis management protocols. Front Endocrinol (Lausanne). 2017;8:106. doi:10.3389/fendo.2017.00106
Vellanki P, Umpierrez GE. Increasing hospitalizations for DKA: A need for prevention programs. Diabetes Care. 2018;41(9):1839-41. doi:10.2337/dci18-0004
Lindsay K. Buchert is an adult nurse practitioner with the inpatient endocrine service in the department of internal medicine at Atrium Health Carolinas Medical Center in Charlotte, North Carolina.




















3 Comments.
I real helped so much.
Great Job!!!!
Very educative topic
Very thorough discussion of this vital topic!