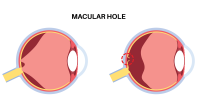
On July 15, the Food and Drug Administration (FDA) approved Tecnis Symfony intraocular lens (IOL), the first IOL that provides cataract patients with an extended depth-of-focus, which helps improve their visual acuity at near, intermediate, and far distances. Read more