On Aug. 17, the U.S. Food and Drug Administration (FDA) approved Besponsa (inotuzumab ozogamicin) for the treatment of adults with relapsed or refractory B-cell precursor acute lymphoblastic leukemia (ALL). Besponsa is a targeted therapy that is thought to work by binding to B-cell ALL cancer cells that express the CD22 antigen, blocking the growth of cancerous cells.
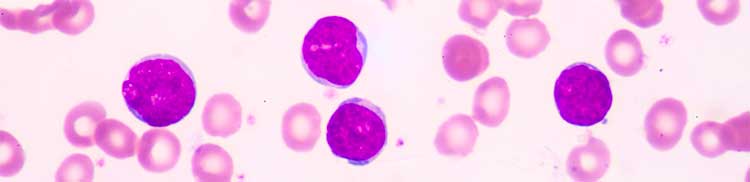

B-cell precursor ALL is a rapidly progressing type of cancer in which the bone marrow makes too many B-cell lymphocytes, an immature type of white blood cell. The National Cancer Institute estimates that approximately 5,970 people in the United States will be diagnosed with ALL this year and approximately 1,440 will die from the disease.
-> read more via the FDA Press Release
The FDA granted this application Priority Review and Breakthrough Therapy designations.
-> read more via the FDA Press Release
The FDA, an agency within the U.S. Department of Health and Human Services, protects the public health by assuring the safety, effectiveness, and security of human and veterinary drugs, vaccines and other biological products for human use, and medical devices. The agency also is responsible for the safety and security of our nation’s food supply, cosmetics, dietary supplements, products that give off electronic radiation, and for regulating tobacco products.

















