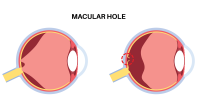
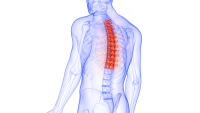
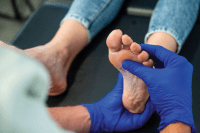
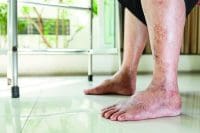
The U.S. Food and Drug Administration (FDA) recently issued warning letters to 15 companies, notifying them the sale of their illegally marketed diabetes products violates federal law. The action is intended to o remove from the market illegal products, including some labeled as dietary supplements, that claim to mitigate, treat, cure, or prevent diabetes and related complications. Read more.