Today implantable cardioverter-defibrillators (ICDs) are in much more common use than they were just 10 years ago. Initially, they were used only in patients who’d survived sudden cardiac death—and, largely because of the low survival rate for this condition, the devices were relatively uncommon. But with clinical trials showing ICDs can help avert death, some patients without a history of arrhythmia but with risk factors for sudden cardiac death are receiving them as primary prevention therapy.
ICDs detect ventricular tachycardia (VT) or ventricular fibrillation (VF), and deliver a shock to restore sinus rhythm. The concept is fairly simple, but over the years, ICD implementation has evolved into a complex algorithm-driven system. The ICD is no longer just another device. It has become a complicated therapy, and clinicians must understand its technical aspects and ramifications for patient care.
How ICDs work
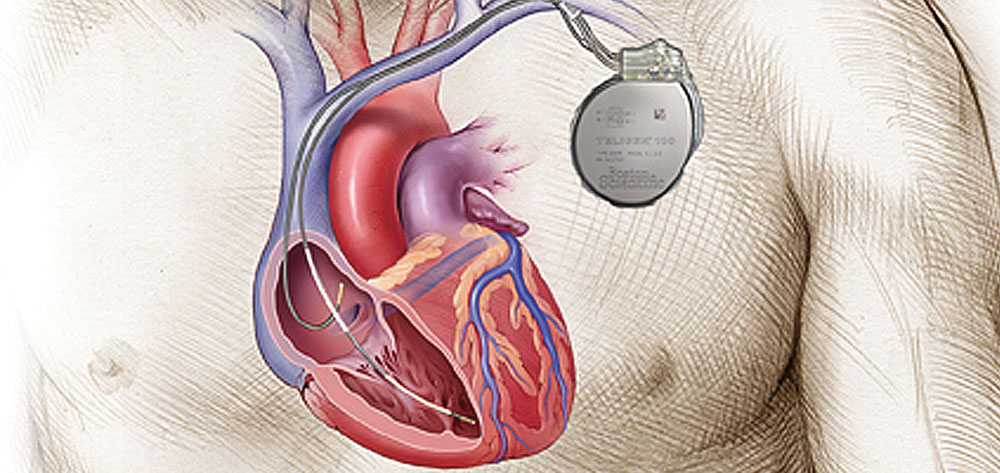
An ICD records the patient’s intrinsic heart rhythm and then paces, cardioverts, or defibrillates as appropriate. It has two major components.
- The pulse generator resembles an implanted pacemaker but is bigger because of its large battery and capacitor system. The system must be charged in excess of 800V to deliver a single shock.
- The leads or conducting wires serve as sensing and pacing leads and have large coils to efficiently deliver the charge. In some cases, a single lead goes to the ventricle; in others, an additional atrial lead allows atrioventricular (AV) synchronous pacing if clinically warranted. (See An ICD from the inside by clicking the PDF icon above.)
ICD algorithms
ICDs use complex numerical algorithms to detect VT or VF onset based primarily on the heart rate. For example, the device might be programmed to deliver a shock when the heart rate exceeds 200 beats/minute for 30 beats. Selection of programmable parameters hinges on such factors as the indication for the ICD or rates of previously documented VT.
So-called SVT (supraventricular tachycardia) discriminators are additional algorithms used to withhold shocks for atrial fibrillation (AF) or other SVTs. These algorithms may include onset criteria, stability criteria, and/or AV dissociation.
- Onset criteria pertain to how abruptly the arrhythmia arises. For example, sinus tachycardia has a more gradual onset than VT or AF.
- Stability criteria refer to how regular the heart rhythm is. AF has more irregular heartbeat intervals than sinus tachycardia or VT.
- An example of AV dissociation is an episode of VT. In this arrhythmia, atrial activity has no association with ventricular activity. Typically, ventricular beats are more numerous than atrial beats.
Here’s an example of how SVT discriminators work: An episode of AF is conducted so rapidly that the ventricular rate meets VT detection criteria, suggesting a need for ICD therapy. But the ICD takes no action because its SVT discrimination criteria determine that the patient doesn’t have VT but an atrial-driven rhythm, based on the association between atrial and ventricular beats.
Once a ventricular arrhythmia is detected, the ICD delivers therapy—either anti-tachycardia pacing or a shock. Using anti-tachycardia pacing as the first line of therapy can reduce the need for a shock by successfully converting the rhythm. (See How ICD use has evolved by clicking on the PDF icon above.)
Benefits and drawbacks
Considered effective 90% of the time, the ICD is by far the best therapy for preventing sudden cardiac death from ventricular arrhythmia. But it’s no panacea. Complications may occur, including infection, hemorrhage, pneumothorax, cardiac tamponade, or device malfunction. Also, despite SVT discriminators, an ICD can be fooled into delivering an unnecessary shock for a rapid but nonlethal rhythm, such as sinus tachycardia or AF, which may make the patient markedly anxious.
One researcher developed a clinical perspective addressing the practical aspects of the decision to use an ICD: For every 100 patients who have ICDs and are followed for 5 years:
- seven or eight lives will be saved
- five to fifteen complications will occur
- 10 to 20 unnecessary shocks will be delivered
- 30 patients will die from other aspects of their cardiac disease.
Patient follow-up
Patients must be monitored regularly to ensure their ICDs are functioning appropriately and programmed to meet their clinical needs. As part of routine follow-up every 3 to 6 months, the ICD is “interrogated.” In this evaluation, a manufacturer-specific computer programmer is used to connect with the ICD and download stored information, including battery life, lead integrity, and stored arrhythmic events with intracardiac electrogram tracings. Such interrogations may occur in the clinic, where wireless technology or a telemetry wand placed over the patient’s chest allows automatic communication with the device. In clinic evaluations, stored information is downloaded and analyzed; the clinician uses the same technology to change ICD settings.
In the past few years, home interrogations (with downloading of information only) have become increasingly common. They allow the patient to be evaluated without leaving home, while giving the clinician the chance to view comprehensive data from a repository on the Web.
Nursing considerations
Although anti-tachycardia pacing may be painless, patients almost always feel a shock from the ICD, which many liken to a kick in the chest. Typically, the energy delivered is 30 joules or more, equivalent to dropping a 25-lb weight onto the chest from 1 foot away. However, patients don’t have long-lasting discomfort and are almost uniformly grateful for the lifesaving therapy.
In my clinic, we instruct patients to call us if they experience a single shock and feel well afterwards, so we can arrange for an evaluation. For multiple shocks delivered on the same day or for other cardiac symptoms, such as palpitations, chest pain, or shortness of breath, we advise patients to come to the emergency department. Larger hospitals commonly have device services available to interrogate ICDs; however, manufacturers’ representatives are always on call and will visit any facility to interrogate devices. Patients must be evaluated after therapy to ensure that the device recognized and treated the rhythm appropriately and to evaluate the need for a change in medical therapy (such as an increased beta-blocker dosage or initiation of an antiarrhythmic drug).
Also be aware of the following:
- Hospital patients with ICDs may receive therapy to abort sudden cardiac death. If a patient’s ICD delivers a shock, the cardiology team should be consulted for evaluation.
- When a patient has surgery involving electrocautery/an electosurgical unit, consideration should be given to disabling the anti-tachycardia therapies to prevent inappropriate shocks due to interference. All patients whose ICDs have been disabled should have continuous cardiac monitoring, and external defibrillation equipment available.
- Appropriate device therapy can be exhausted when a patient is in recalcitrant VF. Therefore, external defibrillation can and should be administered.
Caution patients that the ICD can interact with environmental equipment. Instruct them to maintain a 12-inch distance between the ICD and a running engine or handheld tool, as the electromagnetic field produced could lead to inappropriate shock or pacing in-hibition. For the same reason, instruct them not to undergo magnetic resonance imaging and not to linger or lean against the sensor when passing through store security systems.
Advise patients who have ICDs for secondary prevention not to drive for 6 months after an arrhythmic event. Those with primary prevention indications only and no symptoms can resume driving 1 week after ICD implantation. However, if they receive a shock for VT/VF, driving recommendations revert to those for patients using ICDs for secondary prevention.
ICDs are here to stay, and an increasingly large number of patients entering the healthcare system at all points have them. Knowing how an ICD works and understanding that it’s not just another device but a complex therapy enables clinicians to manage these patients appropriately.
Selected references
Bardy GH, Lee KL, Mark DB, et al; for the Sudden Death in Heart Failure Trial (SCD-HeFT) Investigators. Amiodarone or an implantable cardioverter-defibrillator for congestive heart failure. N Engl J Med. 2005;352:225-237.
Buxton AE, Lee KL, Fisher JD, Josephson ME, Prystowsky EN, Hafley G; for the Multicenter Unsustained Tachycardia Trial Investigators. A randomized study of the prevention of sudden death in patients with coronary artery disease. N Engl J Med.1999;341(25):1882-1890.
Epstein AE, Baessler CA, Curtis AB, et al. Addendum to “Personal and public safety issues related to arrhythmias that may affect consciousness: implications for regulation and physician recommendations: a medical/scientific statement from the American Heart Association and the North American Society of Pacing and Electrophysiology.” Public safety issues in patients with implantable defibrillators: a scientific statement from the American Heart Association and the Heart Rhythm Society. Circulation. 2007;115:1170-1176.
Epstein AE, DiMarco JP, Ellenbogen KA, et al. ACC/AHA/HRS 2008 guidelines for device-based therapy of cardiac rhythm abnormalities: executive summary. Circulation. 2008;117:2820-2840.
Moss AJ, Hall WJ, Cannom DS, et al. Improved survival with an implanted defibrillator in patients with coronary disease at high risk for ventricular arrhythmia. Multicenter Automatic Defibrillator Implantation Trial Investigators. N Engl J Med. 1996;335:1933-1940. http://nejm.highwire.org/cgi/content/full/335/26/1933. Accessed November 23, 2009.
Moss AJ, Zareba W, Hall WJ, et al; for the Multicenter Automatic Defibrillator Implantation Trial II Investigators, Prophylactic implantation of a defibrillator in patients with myocardial infarction and reduced ejection fraction. N Engl J Med. 2002;346:877-883.
Stevenson LW, Desai AS. Selecting patients for discussion of the ICD as primary prevention for sudden death in heart failure. J Card Fail. 2006;12(6):407-412.
Wathen MS, De Groot PJ, Sweeney MO, et al. Prospective randomized multicenter trial of empirical antitachycardia pacing versus shocks for spontaneous rapid ventricular tachycardia in patients with implantable cardioverter-defibrillators: Pacing Fast Ventricular Tachycardia Reduces Shock Therapies (PainFREE Rx II) trial results. Circulation. 2004;110:2591-2596.
Wilcoff BL, Auricchio A, Brugada J, et al. HRS/EHRA expert consensus on the monitoring of cardiovascular electronic devices (CIEDs): description of techniques, indications, personnel, frequency and ethical consideration. Heart Rhythm. 2008;5(6):907-925.
Liza Prudente Moorman is a nurse practitioner in the Electrophysiology/Atrial Fibrillation Center at the University of Virginia Health System in Charlottesville.