New lung cancer treatment modalities and their adverse effects
- Targeted and immunotherapy differ from chemotherapy in mechanisms of action and side-effect profile.
- Nurses must understand the various mechanisms of action and unique side-effects to effectively assess and manage patient responses.
- Immunotherapy-related adverse events require early detection and aggressive management to optimize patient outcomes.
Learning Objectives
- Describe new treatment modalities for lung cancer, including targeted therapy and immunotherapy.
- Discuss adverse effects of new lung cancer treatment modalities.
- Describe how to help patients manage common side effects of new treatment modalities.
The author, planners, and peer reviewers of this CNE activity have disclosed no relevant financial relationships with any commercial companies pertaining to this activity. See the last page of the article to learn how to earn CNE credit.
Expiration: 2/1/26
New treatment modalities for lung cancer, including targeted therapy and immunotherapy, have contributed to improved patient survival rates. However, these treatments come with unique adverse effects. The nurse’s role in managing symptoms of these adverse effects requires an understanding of treatment mechanisms of action and common side effects.
Most oncogenic drivers (gene mutations responsible for the initiation and maintenance of cancer) with corresponding Food and Drug Administration (FDA)–approved molecular targeted therapies are found in non-small cell lung cancer (NSCLC), adenocarcinoma subtype. Significantly fewer FDA-approved targeted therapies exist to treat squamous cell subtype. This article focuses on treatment of metastatic NSCLC.
The FDA has approved several types of targeted therapies for treating lung cancer, including angiogenesis inhibitors, signal transduction inhibitors, and immunotherapies. Each type of targeted therapy has unique and distinct adverse effect profiles. (See Targeted therapy defined.)
Angiogenesis inhibitors
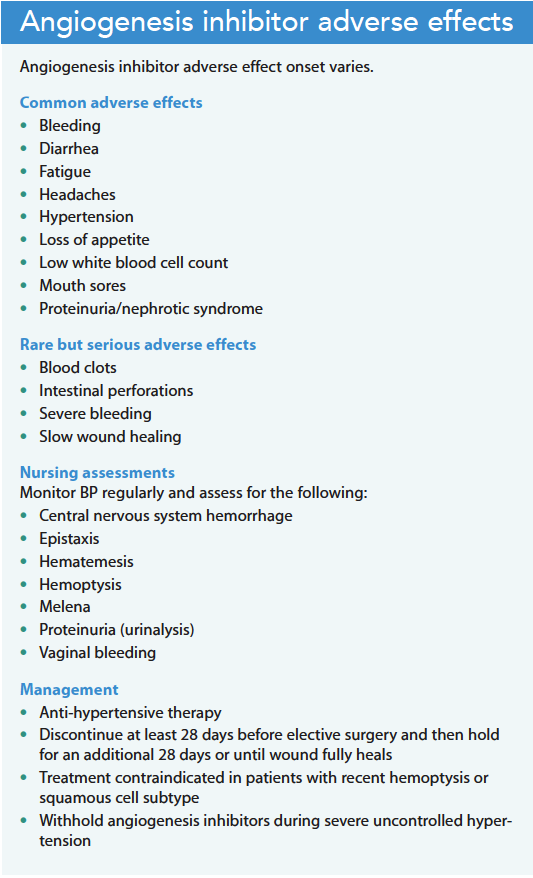
Tumors require a blood supply for continued growth. Tumor angiogenesis (the formation and continued growth of blood vessels into tumors) plays a crucial role in tumor growth, progression, and metastasis. Vascular endothelial growth factor (VEGF), a signaling protein, stimulates the formation of new blood vessels and is overexpressed in tumors. Angiogenesis inhibitors (including bevacizumab and ramucirumab) block VEGF action. Providers may use these anti-VEGF therapies as first-line treatment or along with other targeted therapies. The distinctive adverse effects of angiogenesis inhibitors stem from targeting blood vessel formation and growth rather than the tumor itself. (See Angiogenesis inhibitor adverse effects.)
Nursing considerations
Adverse effects of angiogenesis inhibitors include hypertension, slow-healing wounds, proteinuria, increased risk of internal bleeding. Rarely, these treatments result in intestinal perforation. Education before and during therapy includes encouraging patients to keep a blood pressure log and report abnormal readings. In addition, they should be instructed to notify their nurse or provider if they experience any bleeding or bruising issues or nonhealing wounds. Patients should consult with their oncology team before any surgery while receiving angiogenesis inhibitor therapy.
Signal transducing inhibitors
Signal transduction transmits cellular signals from outside the cell to inside to effect an appropriate response. Advances in the knowledge of signal transduction pathways have allowed for the development of a new class of drugs, signal transduction inhibitors, which block the activities of these pathways. Researchers have identified many targetable pathways in the treatment of lung cancer, and several drugs targeting these pathways have been developed but are beyond the scope of this article. The most common signal transducing inhibitors used to treat lung cancer belong to a class of drugs that target the epidermal growth factor (EGF)/epidermal growth factor receptor (EGFR) signaling pathway cascade.
Signaling pathway cascade
As the EGF protein circulates in the blood, it attaches to the EGFR (or ligand), which activates the EGFR cascade. EGFR is a type of tyrosine kinase (TK) receptor. TKs play a critical role in cell signaling. Once bound to its ligand, EGFR-TK can phosphorylate (activate) other proteins, allowing the signal to go into the cell nucleus and aid gene transcription. Mutations of any of the proteins in this tightly controlled system may affect the regulation of cell growth processes, resulting in uncontrolled cell growth or cancer.
TK inhibitors (TKIs), a group of pharmacologic agents, disrupt the signal transduction pathways of protein kinases via several modes. The FDA has approved more than 50 TKIs, but this article focuses on the most common EGFR-TKIs effective for treating metastatic NSCLC (erlotinib, afatinib, gefitinib, osimertinib, and dacomitinib). EGFR-TKIs are the primary treatment option for NSCLCs that harbor an EGFR mutation.
Nursing considerations
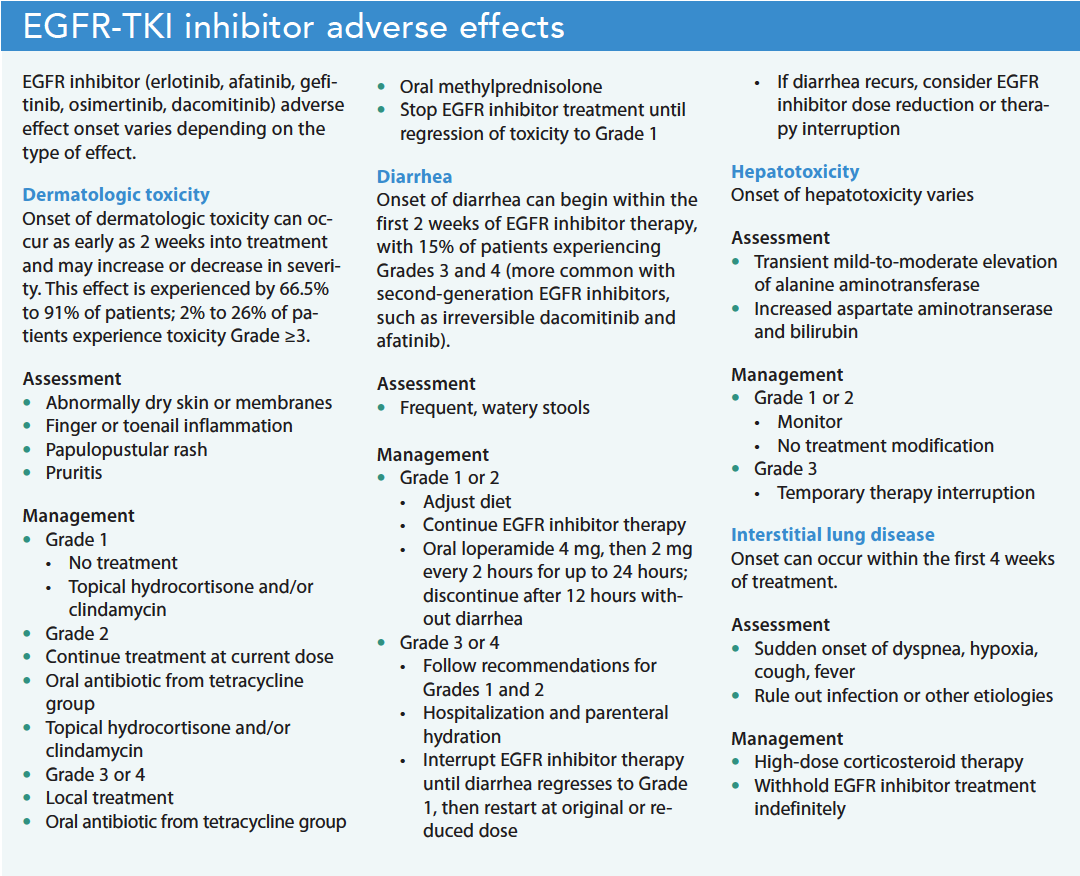
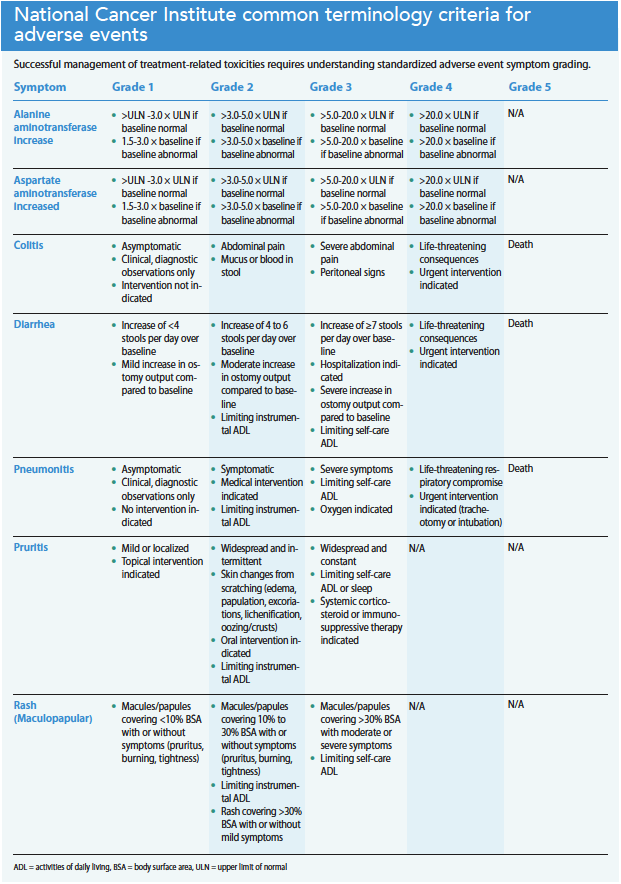
Successful management of treatment-related adverse events requires knowledge of the standardized toxicity grading criteria. Although EGFR-TKIs may cause anorexia, alopecia, and stomatitis, the most common adverse effects include dermatologic toxicity and diarrhea. Less common but serious adverse effects include hepatotoxicity and interstitial lung disease. (See EGFR-TKI adverse effects.)
Dermatologic toxicity.
Potentially painful acneiform rash, the most common dermatologic toxicity (90% of patients), typically appears on the face, scalp, upper back, and chest. Other symptoms include xerosis (abnormally dry skin or membranes [mouth, eyes]) and pruritus (itching). Paronychia (finger or toenail inflammation) can develop when bacteria enter broken skin near the cuticle and nail fold, resulting in infection.
Patient education includes discussion of good skincare, daily use of a light lotion and antiseptic-containing soap substitute, and minimal sun exposure. Toxicity prevention, early recognition, and intervention help minimize subsequent bacterial infections and mitigate debilitating effects. Depending on the severity or grading of the toxicity, treatment recommendations include good skincare with or without the use of prophylactic antibiotics, steroids with or without antibiotics, and EGFR-TKI interruptions.
Diarrhea.
Diarrhea, the second most common (20% to 90% of patients) adverse effect of EGFR-TKIs, typically occurs within the first 2 weeks of treatment. The etiology is unclear but electrolyte imbalances may occur without appropriate management. Most patients experience lower grades of toxicity but long durations of Grades 1 and 2 may cause significant problems.
Diarrhea management begins with pre-treatment patient education. Dietary recommendations include increasing daily fluid intake, eating frequent small meals, and avoiding fried, high-fiber, greasy, and spicy foods. Depending on the toxicity grade, treatment includes dietary management, loperamide, hospitalization, parenteral hydration, and EGFR-TKI interruption. No evidence exists for the efficacy of octreotide in EGFR-TKI–induced diarrhea.
Hepatotoxicity.
Hepatotoxicity, a less-frequent adverse effect, occurs in about 10% of patients. Most have no symptoms and don’t require management. Clinical manifestations include transitional mild-to-moderate increases in alanine transaminase, aspartate aminotransferase, and bilirubin. Most cases don’t require treatment interruption. In the presence of Grade 3 toxicity, temporary EGFR-TKI interruption can be effective. Treatment restart can occur when the toxicity reaches ≤Grade 1. However, if ≥Grade 3 recurs, the treatment dose may require modification.
Interstitial lung disease.
Interstitial lung disease, a rare (0–5.7%) adverse effect, is fatal in approximately one-third of patients. Symptoms include acute onset of dyspnea, hypoxia, cough, and possibly fever within the first 4 weeks of treatment. Prompt recognition ensures proper treatment, so patients should immediately contact their provider with any of these symptoms. The provider will discontinue EGFR-TKI therapy until ruling out infection or another etiology. If ruled out, the provider may order high-dose steroid therapy and withhold EGFR-TKI treatment indefinitely.
Immune checkpoint inhibitors
Immunotherapies trigger a patient’s immune system to destroy cancer cells. They’re primarily used to strengthen and regulate the immune system and microenvironment so immune cells can attack and clear tumor cells. Many immune checkpoint inhibitors (ICIs), the most common type of immunotherapy used to treat lung cancer, involve inhibiting the interaction of the PD-L1 protein and the PD-1 receptors on the T-cell. PD-L1 and PD-1 interaction prevents the immune system from recognizing and attacking cancer cells. Current FDA-approved lung cancer immunotherapy drugs disrupt that interaction, so T-cells can recognize and respond to cancer cells.
Durvalumab and atezolizumab target the PD-L1 protein, and nivolumab and pembrolizumab target the PD-1 receptor. Ipilimumab, another immunotherapy, can block the CTLA4 pathway so the body can ramp up the number of immune cells available to respond to cancer cells.
Nursing considerations
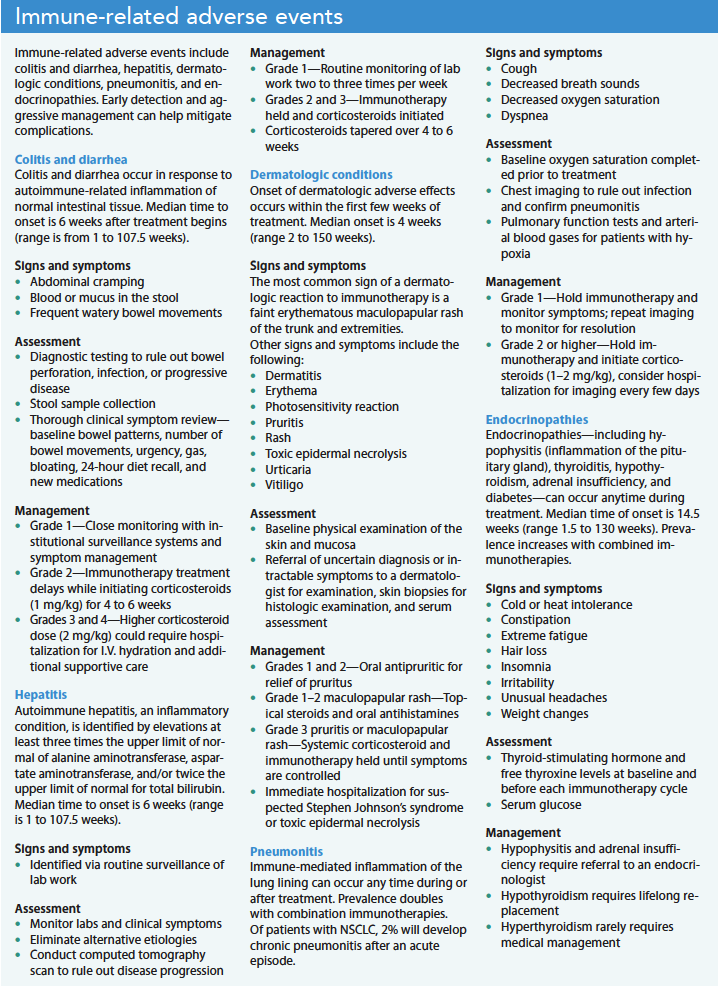
ICIs rely on increased use of the body’s immune system to identify and treat cancer. They upregulate the body’s immune system so much that it begins attacking healthy cells, resulting in immune-related adverse events (irAEs). irAEs can occur in any organ system at any point after treatment begins, even after discontinuation. (See Immune-related adverse events.)
The most commonly affected organs are the lungs, GI tract, thyroid, liver, and skin. Most irAEs are mild to moderate, but severe irAEs can be life-threatening. Relative to chemotherapy-induced toxicities, irAEs have a delayed onset and a prolonged duration. Unlike chemotherapy-induced toxicities, irAEs require early and prompt intervention for patient safety. Treatment may include dose reduction or interruption, permanent ICI cessation, and systemic corticosteroid therapy. With prompt detection and intervention, most patients can remain on ICI therapy. Although severe irAEs are rare, they can result in harm if left untreated.
Adverse events differ among specific immunotherapy agents. Many common ICIs (nivolumab, pembrolizumab, atezolizumab, and ipilimumab) may cause colitis/diarrhea, dermatitis, endocrinopathies (hyper- or hypothyroidism and hypophysitis), hepatitis, and pneumonitis. These irAEs are managed differently and more promptly relative to other targeted therapies and chemotherapy.
Nurses should contact the oncology team if the patient exhibits a Grade 2 or higher adverse event while receiving immunotherapy. Nurses also should provide patient education to ensure prompt recognition of irAE signs and symptoms and immediate communication with providers.
Understand the basics
Understanding the basics of new lung cancer treatment modalities helps nurses perform proper assessments and recognize adverse effects that may impact patient quality of life and ongoing therapy. Providing patient education and communicating promptly with a patient’s oncology team aid care coordination in all settings and improve patient outcomes.
Monica L. Beck is an assistant professor at the University of Alabama Huntsville College of Nursing in Huntsville. Haley Hoy is a professor at the University of Alabama Huntsville College of Nursing. Rachel McMahill is a clinical nurse navigator at Clearview Cancer Center in Huntsville, Alabama.
American Nurse Journal. 2023; 18(2). Doi: 10.51256/ANJ022306
Key words: lung cancer, metastatic non-small cell lung cancer (NSCLC), targeted therapy, immunotherapy, treatment adverse effects
References
American Cancer Society. Targeted drug therapy for non-small cell lung cancer. December 13, 2022. cancer.org/cancer/lung-cancer/treating-non-small-cell/targeted-therapies.html
American Lung Association. Lung cancer immunotherapy. November 17, 2022. lung.org/lung-health-diseases/lung-disease-lookup/lung-cancer/treatment/types-of-treatment/immunotherapy
Ettinger DS, Wood DE, Aisner DL, et al. Non-small cell lung cancer, Version 3.2022, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw. 2022;20(5). doi:10.6004/jnccn.2022.0025
Gordon R, Kasler MK, Stasi K, et al. Checkpoint inhibitors: Common immune-related adverse events and their management. Clin J Oncol Nurs. 2017;21(2 suppl):45-52. doi:10.1188/17.CJON.S2.45-52
Hirsch FR, Scagliotti GV, Mulshine JL, et al. Lung cancer: Current therapies and new targeted treatments. Lancet. 2017;389:299-311. doi:10.1016/S0140-6736(16)30958-8
Kooshkaki O, Derakhshani A, Hosseinkhani N, et al. Combination of ipilimumab and nivolumab in cancers: From clinical practice to ongoing clinical trials. Int J Mol Sci. 2020;21(12):4427. doi:10.3390/ijms21124427
National Cancer Institute. Targeted therapy to treat cancer. May 31, 2022. cancer.gov/about-cancer/treatment/types/targeted-therapies/targeted-therapies-fact-sheet
National Consensus Project for Quality Palliative Care. Clinical Practice Guidelines for Quality Palliative Care, 4th ed. National Coalition for Hospice and Palliative Care; 2018. nationalcoalitionhpc.org/ncp.
Schneider BJ, Naidoo J, Santomasso BD, et al. Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: ASCO guideline update. J Clin Oncol. 2021;39(36):4073-4126. doi:10.1200/JCO.21.01440
Tan S, Li D, Zhu X. Cancer immunotherapy: Pros, cons and beyond. Biomed Pharmacother. 2020;124:109821. doi:10.1016/j.biopha.2020.109821
Teleanu RI, Chircov C, Grumezescu AM, Teleanu DM. Tumor angiogenesis and anti-angiogenic strategies for cancer treatment. J Clin Med. 2020;9(1):84. doi:10.3390/jcm9010084
Thompson JA, Schneider BJ, Brahmer J, et al. Management of immunotherapy-related toxicities, Version 1.2022, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2022;20(4):387-405. doi:10.6004/jnccn.2022.0020
U.S. Department of Health and Human Services. Common terminology criteria for adverse events (CTCAE) Version 5.0. November 27, 2017. ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/ctcae_v5_quick_reference_5x7.pdf
Williams LA, Ginex PK, Ebanks GL, et al. ONS guidelines for cancer treatment-related skin toxicity. Oncol Nurs Forum. 2020;47(5):539-56. doi:10.1188/20.ONF.539-556
Wood LS, Moldawer NP, Lewis C. Immune checkpoint inhibitor therapy: Key principles when educating patients. Clin J Oncol Nurs. 2019;23(3):271-80. doi:10.1188/19.CJON.271-280
Yuan M, Huang LL, Chen JH, Wu J, Xu Q. The emerging treatment landscape of targeted therapy in non-small-cell lung cancer. Sig Transduct Target Ther. 2019;4:61. doi:
10.1038/s41392-019-0099-9
















1 Comment. Leave new
Completed this informative CE in American Nurse magazine. Sharing with Hematology/Oncology team nursing. Found the cross platform approach to therapy and what to look for in side effects were appreciated.