On April 17, the U.S. Food and Drug Administration (FDA) approved Crysvita (burosumab), the first drug approved to treat adults and children ages 1 year and older with x-linked hypophosphatemia, a rare, inherited form of rickets.
Read more via FDA.
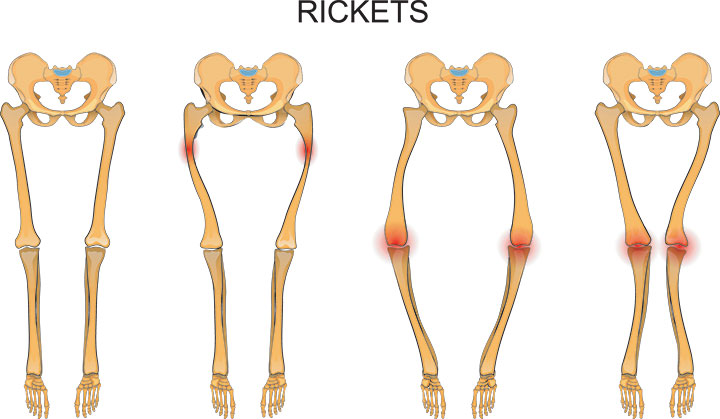

On April 17, the U.S. Food and Drug Administration (FDA) approved Crysvita (burosumab), the first drug approved to treat adults and children ages 1 year and older with x-linked hypophosphatemia, a rare, inherited form of rickets.
Read more via FDA.