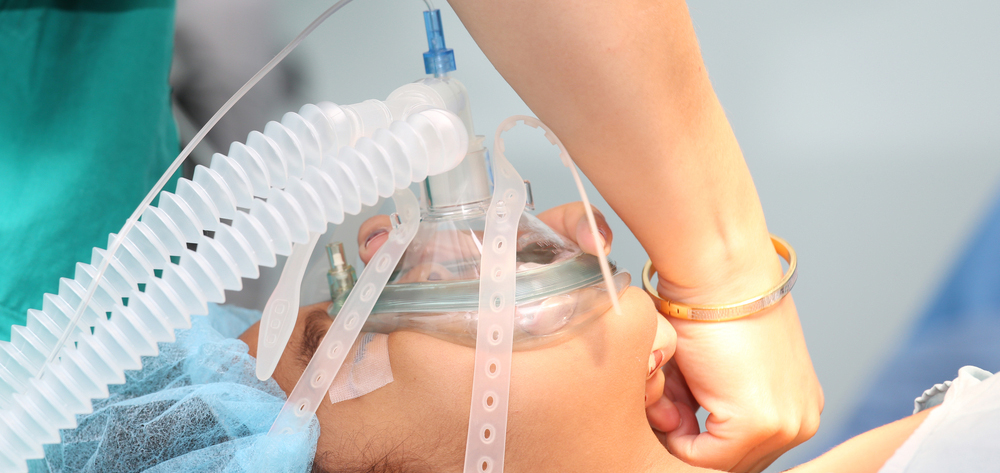
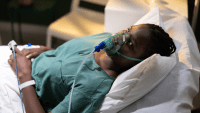
Over the last decade, noninvasive ventilation—now used almost exclusively with positive pressure—has seen a revival. It’s safer, more comfortable, and more convenient than ventilation involving an artificial airway. But it’s not for everyone.
During the early 20th century, ventilatory assistance generally was noninvasive and involved the delivery of negative pressure. The prime example is the iron lung used to treat patients with postpolio syndrome. By the 1960s, with advances in artificial airways and positive-pressure application, mechanical ventilation using artificial airways became the standard of care for patients in acute respiratory failure.
Respiratory failure may result from central nervous system failure, primary lung disease (such as chronic obstructive pulmonary disease [COPD]), infection, neuromuscular disease, or traumatic injury (such as pulmonary contusion, hemothorax, or pneumothorax). Depending on the abruptness and severity of the precipitating event, these conditions may necessitate immediate artificial airway insertion and mechanical ventilation. However, if respiratory failure progresses over time and the patient can sustain some respiratory reserve, a trial of noninvasive positive-pressure ventilation (NPPV) may be a good alternative to invasive ventilation.
Positive perks
In its latest incarnation, noninvasive ventilation uses positive rather than negative pressure. NPPV may decrease intubation complications—particularly hospital-acquired pneumonia and airway injury—while preserving the patient’s airway defense mechanisms, speech, and swallowing. What’s more, it can be initiated, discontinued, and reinitiated easily in patients who need only intermittent ventilatory support.
NPPV may be especially useful for “Do not intubate” patients, such as those with end-stage COPD, who may develop an infection and require ventilatory support but don’t want or need intubation. NPPV also allows the use of ventilatory support in such patient care areas as emergency departments and rehabilitation units.
###Break###
Benefits of NPPV
Studies show NPPV is safe and causes few major complications when used with well-selected patients. In carefully chosen candidates with respiratory failure, it provides ventilatory assistance without the complications that have been linked to artificial airways, such as ventilator-associated pneumonia (VAP), airway injury, sinusitis, increased need for sedatives or paralytics (or both), altered communication, and decreased ability to perform activities of daily living.
Modes and settings
With any mode, the goal of NPPV is to unload the breathing muscles and relieve respiratory distress while avoiding excessive discomfort caused by air pressure and flow. The most effective settings can be determined only by trial and error while clinicians assess the patient’s response.
NPPV may be used with:
- continuous positive-airway pressure (CPAP)
- bilevel positive-airway pressure (BiPAP)
- controlled ventilation modes.
It may be delivered by a dedicated NPPV machine or by a conventional intensive care unit (ICU) ventilator equipped with a noninvasive option. The choice hinges on patient condition, equipment availability, and clinician comfort and experience.
For the spontaneously breathing patient, CPAP helps counterbalance intrinsic positive end-expiratory pressure (PEEP) and promotes exhalation by stenting the airways open. The patient spontaneously provides the entire minute ventilation (respiratory rate x tidal volume).
Also called pressure support ventilation, BiPAP senses patient effort and releases air flow to target the set inspiratory pressure; once achieved, this pressure returns to the lower level or PEEP. At either level, breathing is unrestricted. Cycling between two pressure levels creates a pressure differential that promotes carbon dioxide (CO2) removal; it may be a better choice than CPAP if the main goal is to decrease the partial pressure of CO2 and unload the respiratory muscles. As long as lung compliance remains constant, BiPAP helps ensure reliable ventilation.
Initially, inspiratory pressure may be set at a relatively low level (5 to 10 cm H2O) and then increased gradually as tolerated; or it may be started high (15 to 20 cm H2O) and lowered gradually if the patient shows intolerance. To minimize adverse effects, such as sinus pain and gastric insufflation, pressure should be no higher than 20 cm H2O. PEEP levels should be titrated to prevent derecruitment and counter intrinsic PEEP.
Controlled ventilation modes such as pressure-control ventilation may benefit patients who experience frequent apnea episodes or need a time-cycled inspiratory phase.
###Break###
Indications and contraindications
Patients who may be candidates for NPPV include those with severe COPD exacerbations or cardiogenic pulmonary edema.
Contraindications for NPPV include:
- medical instability
- respiratory arrest
- fluctuating level of consciousness
- inability to protect the airway
- profound hypoxemia, shock, or acidemia
- excessive secretions
- lack of cooperation
- agitation
- recent upper airway or GI surgery
- inability to fit the mask properly.
###Break###
Choosing the patient-ventilator interface
In mechanical ventilation, the patient–ventilator interface may be as simple as a mask or part of a bag-valve-mask unit, or it may be more invasive, such as an artificial airway. Arguably, the interface is the most important component of NPPV success. To promote effective NPPV, the interface should minimize leakage, enhance patient comfort, and allow synchronization with the ventilator.
The four primary noninvasive interfaces used are the nasal mask, facial mask, nasal prongs (or pillows), and helmet. A mouthpiece also may be used to administer NPPV, although this is rarely done in the ICU.
Keep in mind that although the NPPV interface is noninvasive, the patient is still receiving mechanical ventilation—and this isn’t a pleasant experience even in the best circumstances. Also, some patients may become claustrophobic, depending on the type of mask used. Switching to a nasal mask may lessen this sensation while also allowing the patient to speak, eat, and drink. On the other hand, a nasal mask may be less effective for patients who can’t adequately seal their lips. A helmet may decrease leaks but may worsen claustrophobia in some patients; also, it may be less effective for CO2 elimination due to increased dead space.
Obviously, there’s no single right choice of interface for all patients. Facial shape, anxiety level, illness acuity, and patient preference must all be considered. Nonetheless, all NPPV candidates should be capable of protecting their airways or removing the mask in the event of vomiting to minimize the aspiration risk. Feeding tubes aren’t contraindicated but may interfere with creating a face-to-mask seal. Achieving a seal also may be a challenge in patients without lower teeth, so dentures should be retained unless contraindicated. Finally, the mask may leak significantly or become too uncomfortable if too much pressure is applied.
Post-extubation NPPV
Respiratory failure after extubation carries a poor prognosis, including longer ventilator time, greater likelihood of discharge to a long-term care facility, and higher mortality. NPPV may help prevent the need for reintubation, although its routine use in all patients after extubation hasn’t been validated clinically. One study compared NPPV efficacy against that of standard medical therapy in patients at risk for extubation failure; the NPPV group had a reintubation rate about one-third lower than the standard medical therapy group.
NPPV also may be useful in patients with persistent weaning failure. In one prospective randomized trial, 43 patients who’d failed a weaning trial for 3 consecutive days were randomly assigned either for extubation to NPPV or to continue mechanical ventilation followed by a conventional weaning plan. Extubation to NPPV led to a shorter duration of mechanical ventilation, fewer complications, and improved survival. However, until more studies are done and evidence-based guidelines are developed, routine use of post-extubation NPPV should be evaluated individually and limited to appropriate patients.
###Break###
Nursing management
If your patient is on NPPV, monitor cough and secretions frequently. Inability to effectively mobilize secretions may warrant adding humidification to the NPPV system, along with administration of mucus thinning agents, expectorants, or both. If eye irritation occurs, use eyedrops or ophthalmic lubricant as ordered. If the patient develops ear pain and nasal and sinus congestion, provide good oral care, saline spray, or decongestants and reassess air flow and mask fit.
Gastric distention usually stems from air pressure. Normal gastro-esophageal sphincter pressure is about 20 cm H2O but may fall below this level during swallowing and in response to certain drugs. Although uncomfortable, gastric insufflation rarely warrants NPPV cessation. It may be eased by titrating inspiratory pressures to a lower level and maintaining the head of the bed at 30 degrees or higher.
Judicious pharmacologic therapy, including anxiolytics and appropriate analgesia, can improve tolerance of and synchrony with NPPV. Current sedation practices are diverse and guided mainly by clinical experience. As appropriate, follow standardized sedation protocols similar to those used for invasive ventilation. But be aware that even with appropriate sedation, patients may feel anxious and require frequent reassurance, coaching, and psychological support. Frequently assess mental status, vital signs, work of breathing, and ability to protect the airway to help prevent fatigue or intolerance. Stay alert for hemodynamic instability and other findings that warrant a switch to invasive ventilation. (See Reasons to switch to invasive ventilation.)
Teach the patient and family about the:
- rationale for and methods of NPPV
- importance of keeping the mask or other interface on
- possibility that ventilatory treatment will need to be escalated if NPPV fails.
Monitoring for complications
Most NPPV complications stem from the patient-ventilator interface and air flow. Inspect the skin over the bridge of the patient’s nose, chin, and head; skin in these areas may break down from attempts to obtain an adequate seal with nasal or facial masks and straps. To reduce or prevent such breakdown, use protective hydrocolloid dressings. To detect intolerance to NPPV and signs and symptoms of discomfort, frequently assess the patient’s skin integrity and nonverbal behaviors.
To help prevent skin breakdown, ensure that the mask is of good quality and fits properly. If the patient will be on NPPV for more than several days or is expected to be discharged home on it, a high-quality moldable gel mask should be considered.
A key role in improving outcomes
In selected patients, NPPV can be an attractive alternative to invasive ventilation, reducing the risk of VAP and other complications, allowing earlier extubation, and in some cases even eliminating the need for intubation. Nonetheless, despite its relatively low risk, patients must be selected carefully based on the latest guidelines. As the use of mechanical ventilation continues to rise, NPPV will play an important role in optimizing healthcare resources, decreasing costs, and improving patient outcomes.
###Break###
Selected references
Antonelli M, Conti G, Esquinas A, et al. A multiple-center survey on the use in clinical practice of noninvasive ventilation as a first-line intervention for acute respiratory distress syndrome. Crit Care Med. 2007;35(1):18-25.
Antonelli M, Pennisi M, Montini L. Clinical review: noninvasive ventilation in the clinical setting—experience from the past 10 years. Crit Care. 2005;9:98-103.
Augustyn B. Ventilator-associated pneumonia: risk factors and prevention. Crit Care Nurse. 2007;27(4):32-39.
Caples S, Gay P. Noninvasive positive pressure ventilation in the intensive care unit: a concise review. Crit Care Med. 2005;33:2651-2658.
Dasta J, Mclaughlin T, Mody S, Piech C. Daily cost of an intensive care unit day: the contribution of mechanical ventilation. Crit Care Med. 2005;33:1266-1271.
Devlin J, Nav S, Fong J, et al. Survey of sedation practices during noninvasive positive pressure ventilation to treat acute respiratory failure. Crit Care Med. 2007;35(10):2298-2302.
Dos Santos C. Hyperoxic acute lung injury and ventilator-induced/associated lung injury: new insights into intracellular signaling pathways. Crit Care. 2007;11:126.
Ferrer M, Esquinas A, Arancibia F, et al. Noninvasive ventilation during persistent weaning failure. Am J Resp Crit Care Med. 2003;168:70-76.
Garpestad E, Brennan J, Hill N. Noninvasive ventilation for critical care. Chest. 2007;132:711-720.
Gavaghan S, Jeffries M. Your patient’s receiving noninvasive positive-pressure ventilation. Nursing. 2006;36:46-47.
Hess D. Noninvasive positive-pressure ventilator and ventilator associated pneumonia. Respir Care. 2005;50(7):924-929.
Hill N, Brennan J, Garpestad E, Nava S. Noninvasive ventilation in acute respiratory failure. Crit Care Med. 2007;35:2402-2407.
Ho KM, Wong K. A comparison of continuous and bi-level positive airway pressure non-invasive ventilation in patients with acute cardiogenic pulmonary oedema: a meta analysis. Crit Care. 2006;10:R4921.
Nava S, Gregoretti C, Fanfulla F, et al. Noninvasive ventilation to prevent respiratory failure after extubation in high-risk patients. Crit Care Med. 2005;33(11):2465-2470.
Nava S, Navalesi P, Carlucci A. Non-invasive ventilation. Minerva Anestesiol. 2008;74:1-6.
Preston R. Introducing non-invasive positive pressure ventilation. Nurs Stand. 2001;15:42-45.
Rana S, Hussam J, Gay P, et al. Failure of non-invasive ventilation in patients with acute lung injury; observational cohort study. Crit Care Med. 2006;10:R79.
Schumaker G, Hill N. Utilization of critical care resources is increasing—are we ready? J Intensive Care Med. 2006;21:191-193.
Tremblay L, Valenza F, Ribeiro SP, et al. Injurious ventilatory strategies increase cytokines and c-fos mRNA expression in an isolated rat lung model. J Clin Invest. 1997;99(5):944-952.
Trevisan CE, Vieira SR. Noninvasive mechanical ventilation may be useful in treating patients who fail weaning from invasive mechanical ventilation: a randomized trial. Crit Care. 2008;12:R51
Mark Bauman is a senior clinical nurse in the Select Trauma ICU at the R Adams Cowley Shock Trauma Center at the University of Maryland Medical Center in Baltimore. The planners and author of this CNE activity have disclosed no relevant financial relationships with any commercial companies pertaining to this activity.