When you participate in the usability process, you provide vital safety information.
Key Takeaways
– As part of the science of human factors, usability engineering studies the interactions between people and the systems in which they function.
– Nurses can contribute to the usability process and patient safety through involvement in device development, procurement, and hazard analysis.
– Applying human factors heuristics (known standards) to medical device design can help identify potential usability problems that may contribute to adverse events or safety hazards.
In 2008, the Institute for Healthcare Improvement developed the Triple Aim as a framework to guide healthcare delivery in the United States. (See Triple Aim goals.) To meet these goals, healthcare organizations must function at high levels of efficiency, efficacy, and safety, which requires an engaged workforce who understands the organization’s mission and purpose.
Medical devices undergo a lengthy process before arriving at the bedside for use in patient care. Although many factors affect device safety, a robust usability engineering process is key to success. As part of the science of human factors, usability engineering studies the interactions between people and the systems in which they function, including ease of use, the conditions under which a device is used, the tasks completed by the user and the expected outcomes of use, and device users.


You can contribute to the usability process in three areas: device development, procurement, and hazard analysis.
1 Device development
Your input can begin at the initiation of product development. From simply providing feedback on current practices to participating in formal usability testing, you can help shape the final device design. Your insight about current devices, workflow, and potential risks can help generate vital input that might not be discovered without you. (See Nursing involvement in product development.)
When products are ready for user testing, you can continue to have impact by participating in simulated-use studies where you perform tasks that are part of your daily work.
Once a device is near market-ready, a human factors validation study is conducted to meet the expectations of the U.S. Food & Drug Administration (FDA). Validation is the final verification of new device safety, ensuring that the design has been optimized to reduce risk. By participating during this phase, you help provide assurance that devices will be operated safely after reaching the market.
Nursing involvement in product development
Developed by the MedStar Health National Center for Human Factors in Healthcare, this process for usability depicts typical phases of product development and how nurses can contribute at each step along the way.
| Product development phase | Nursing activities |
|---|---|
| – Allow observations in the clinical environment. – Provide feedback on currently used systems. – Participate in interviews and focus groups. – Discuss workflow constraints. – Help generate assessment of device risk. – Identify heuristic violations in new designs. – Conduct iterative prototype evaluations. – Participate in formative user testing. |
– Participate in simulated use testing. – Identify potential use problems and hazards. | |
2 Device procurement
After a device has been approved by the FDA, you can have a role in the procurement process, helping to determine which devices should be used in your healthcare environment. Procurement decisions typically rely on information from device fairs and manufacturer demonstrations. However, this information often focuses only on positive features, failing to provide insights into how a device might realistically function in practice. Ideally, nurses should test these products before a purchasing decision is made.
A recent case study of a hospital system’s new infusion pump purchase illustrates the importance of nursing involvement during procurement. After the decision had been narrowed to two infusion pumps, human factors engineers conducted comparison usability testing with the goal of gathering data to help determine which pump better promoted safe and effective use. This included collection of crucial data obtained through nurse performance of potential high-risk tasks in a simulated setting. The data from the testing demonstrated that one pump had three potentially serious safety flaws, leading to the purchase of the safer device. This result wouldn’t have been possible without the data collected from nurse participation.
3 Device hazard analysis
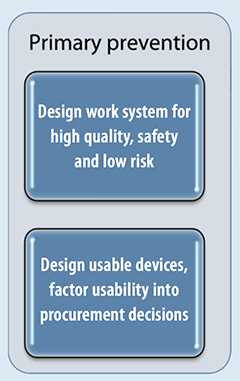
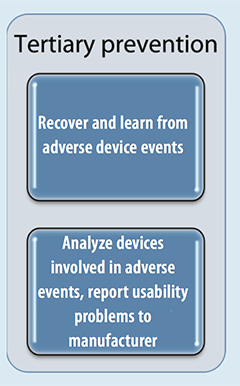
Unfortunately, we can’t always prevent risky or unusable devices from getting into healthcare environments, so sometimes we have to rely on secondary and tertiary methods to improve safety. (See Quality and safety transformational model.)
You can use several techniques to identify usability issues that may contribute to adverse events or safety hazards. By assessing a device based on human factors heuristics (known standards), you can generate solutions to keep an unusable device from causing harm. For example, if a device interface generates an error message that doesn’t identify the problem, it’s violating the “Good Error Message” standard, and an update to the message wording should be considered. (See Medical device heuristics.)
Quality and safety transformational model
This model for risk reduction developed by MedStar Health National Center for Human Factors in Healthcare illustrates how usability should be considered during each prevention phase. Primary prevention focuses on designing usable devices and incorporating well-designed devices into the work system, while secondary and tertiary prevention techniques are used when poorly designed devices already exist.




Devices are used within complex work systems composed of multiple components that alone or collectively affect device usage and safety. The Systems Engineering Initiative for Patient Safety model breaks the work system into five interacting components:
- person—for example, device users (including their physical and psychological characteristics, skills, and education), biomedical staff, and families
- tools and technology—for example, electronic health records, medical devices and accessories, and interoperability
- tasks—for example, step-by-step procedures, common and unique usage scenarios, workflow, and workload
- environment—for example, space layout, location of equipment, noise, and temperature
- organizational factors—for example, policies and procedures, protocols, training, work culture, and teamwork.
Device usability issues affect not only the device, but also other areas of the work system. In turn, work system elements can have a detrimental effect on successful device use. For example, a poorly lit device interface might function well in a daytime environment but can become a safety hazard when used in low lighting. Just as considering these factors during device design can result in products that fit better into the expected work systems, you can think about the system components when analyzing your work environment for potential usability hazards.
How to get involved
You can take several routes to get involved in helping develop and evaluate the devices that may be introduced into your work environment. Some healthcare systems employ usability engineers who work with device manufacturers during development. MedStar Health National Center for Human Factors in Healthcare, for example, conducts usability activities during all development phases and places a high value on nursing involvement. Nurses at MedStar can reach out directly to the human factors team to participate. If your healthcare system doesn’t have these capabilities, you will find that many usability firms welcome your participation.
In your organization, ask your managers how you can have input on new purchases. If formal usability studies aren’t being conducted, use some of the approaches mentioned in this article to assess devices through a human factors lens and consider how the new device might affect the work system. For instance, walk through tasks that might be risky or present problems when performed in your daily work and apply heuristics to detect potential use problems.
If a poorly designed device already exists within your system, examine current hazards and near misses from a systems perspective to generate potential solutions. Use heuristics as a guide or a checklist to evaluate current devices and determine how they might be modified to better fit your needs. This same approach can be used after serious safety events to determine if poor usability was a factor. If you find critical usability problems, talk to management about communicating with the device manufacturer so they can incorporate that information into their next design and create workarounds as appropriate.
Your input can help not only to mitigate hazards in your current environment, but also to develop the next generation of safer, more usable products.
Medical device heuristics
Applying human factors heauristics to medical device design can help identify potential usability problems. These 14 heauristics were created specifically to assess medical device interfaces.
![]()
![]()
Consistency and standards – Devices should maintain consistency with use of words, situations, and actions, and follow conventions of product design.
![]()
![]()
Visibility of system state – Appropriate feedback and display of information ensures users stay informed about what’s happening with the system.
![]()
![]()
Match between system and world – The image of the system perceived by users should match the model the users have.
![]()
![]()
Minimalism – Extraneous information is a distraction and can cause slowdowns.
![]()
![]()
Minimize memory load – Task completion shouldn’t require excessive memorization.
![]()
![]()
Informative feedback – User actions should trigger prompt, informative feedback.
![]()
![]()
Flexibility and efficiciency – Flexibility allows users to create customization and shortcuts to accelerate performance.
![]()
![]()
Good error messages – Informative error messages help users understand the nature of errors, learn from them, and recover quickly.
![]()
![]()
Prevent errors – Appropriately designed user interfaces prevent errors from happening in the first place.
![]()
![]()
Clear closure – Task completion should be clearly communicated to users.
![]()
![]()
Reversible actions – Reversible actions allow users to recover from errors and also encourage exploratory learning.
![]()
![]()
Use user’s language – Device language should be understood by the intended users.
![]()
![]()
Users control – Users should not feel that they are controlled by the systems.
![]()
![]()
Help and documentation – Always provide help when needed.
Adapted from Zhang J, Johnson TR, Patel VL, Paige DL, Kubose T. Using usability heuristics to evaluate patient safety of medical devices. J Biomed Inform. 2003;36(1-2):23-30.
Natalie A. Abts is a program manager in usability services at the MedStar National Center for Human Factors in Healthcare in Washington, DC. Patricia R. McCartney is director of nursing research at MedStar Washington Hospital Center in Washington, DC.
References
Carayon P, Schoofs Hundt A, Karsh BT, et al. Work system design for patient safety: The SEIPS model. Qual Saf Health Care. 2006; 15(Suppl 1):i50-8.
International Organization for Standardization. ISO 9241-11:1998 Ergonomic requirements for office work with visual display terminals (VDTs). Part 11: Guidance on usability. March 1998. https://www.iso.org/standard/16883.html. Updated January 25, 2013.
U.S. Department of Health and Human Services. Food & Drug Administration Center for Devices and Radiological Health. Applying human factors and usability engineering to medical devices: Guidance for industry and Food and Drug Administration Staff. Updated February 3, 2016. www.fda.gov/downloads/MedicalDevices/DeviceRegulationandGuidance/GuidanceDocuments/UCM259760.pdf.
Zhang J, Johnson TR, Patel VL, Paige DL, Kubose T. Using usability heuristics to evaluate patient safety of medical devices. J Biomed Inform. 2003;36(1-2):23-30.





















