Understanding how and why anesthesia providers select analgesic agents.
Takeaways:
- Many options exist for managing intraoperative and postoperative surgical pain.
- Medications used for pain management can be broken down into opioids and non-opioids.
Each medication has specific indications and considerations a postanesthesia care unit nurse must be mindful of when monitoring patients in a postoperative setting.
By Huy Vo, MSN, CRNA; Erica Clayton, MSN, CRNA; and Jessica Stolyarskaya, BA

Ms. Rodriguez is awake but lethargic, and her vital signs are within normal limits. While performing your pain assessment, Ms. Rodriguez reports dull back pain along her incision site and tells you that her pain level is 8 out of 10. As the nurse receiving this patient, what are your postoperative concerns for managing Ms. Rodriguez’s pain?
Developing an understanding of the various pain medications administered by anesthesia providers (as well as the basic principles of opioid pharmacology) will help you anticipate your patient’s needs so you can develop a care plan that safely and effectively treats her pain.
Opioid pharmacology overview
Patients undergoing surgery experience noxious stimuli that result in the release of various inflammatory mediators (such as substance P, glutamate, and prostaglandins). These mediators activate the body’s pain receptors, causing transmission of a stimulus from the peripheral nerve receptors to the spinal cord and brain, which ultimately leads to the sensation of pain. Opioids work at each step along this path to reduce pain. (See Understanding pain physiology.)
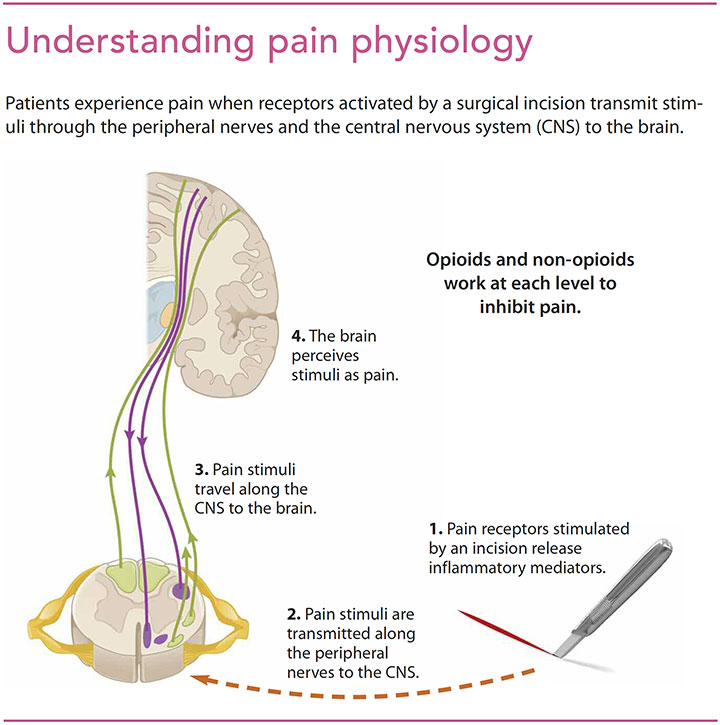

Opioid receptors
Opioids provide analgesia by binding and activating specific receptors that decrease nociception (normal pain resulting from tissue injury). The four opioid receptor types—mu, kappa, delta, and epsilon—are distributed throughout the peripheral and central nervous system. All opioids are selective for the mu receptor, with the exception of meperidine, which is selective for both mu and kappa receptors.
Metabolism
With the exception of remifentanil, opioids undergo two processes for elimination from the body: biotransformation and excretion. Biotransformation (oxidative-reduction and conjugation) occurs within the liver, where the drug is broken down into a substrate that can be excreted through the kidneys. To a lesser extent, opioids also are excreted through the biliary system and GI tract.
Because opioids are excreted by the kidneys, you must be aware of the side effects they have on patients with renal failure (impaired renal clearance leads to impaired opioid clearance, which increases sedation and respiratory depression). Vigilantly monitor your patient’s response to the medication. Maintain oxygen saturation levels at or above 95%, and watch for respiratory depression (< 12 breaths/minute) and hypotension (10% to 15% below the patient’s baseline blood pressure as reported by the anesthesia provider).
Remifentanil is the only opioid metabolized in the blood and tissues of the body through a process called ester hydrolysis. This process allows for rapid metabolism within 9 to 18 minutes after the last medication administration with little to no side effect profile, regardless of the time or amount of medication infused.
Side effects
The most common opioid side effects are sedation and respiratory depression. The degree of sedation and respiratory depression is dose dependent.
Other common opioid side effects include itching, nausea, ileus, and urinary retention. Itching, which may occur as a result of histamine release, is seen primarily with morphine and meperidine. Nausea stems from a complex interaction between opioids and the nausea centers (chemotrigger zones) located in the medulla; it can be treated with antiemetics (such as ondansetron, metoclopramide, promethazine, diphenhydramine, and intramuscular ephedrine). Activation of mu receptors provides analgesia, but within the digestive tract activation can cause gut immobility, which places the patient at risk for postoperative ileus. And activation of mu receptors in the urinary tract may cause urinary retention.
Generally, low-dose opioids minimally affect a patient’s heart rate and blood pressure. However, when administered in high doses, bradycardia and hypotension may occur. Use care when administering opioids in patients at risk for hemodynamic instability, such as those who are elderly or have heart disease.
You may see miosis (pupillary constriction) in patients profoundly sedated with high-dose opioids that inhibit the pupilloconstrictor nucleus. These patients may require oxygen supplementation via facemask or an opioid antagonist (naloxone).
Opioid analgesic agents
During surgery, the patient’s anesthesia provider will use several analgesic agents, including opioids and non-opioids. Commonly used opioids include fentanyl, morphine, and hydromorphone; less commonly used are remifentanil, alfentanil, sufentanil, and meperidine.
Historically, surgical pain has been treated with opioids. However, both the American Association of Nurse Anesthetists and the American Society of Anesthesiologists recommend a multimodal approach to pain management. (See Multimodal analgesia explained.)


Commonly used opioids
The most commonly used opioids in anesthesia are fentanyl, morphine, and hydromorphone. (See Pharmacology of commonly used opioids.)
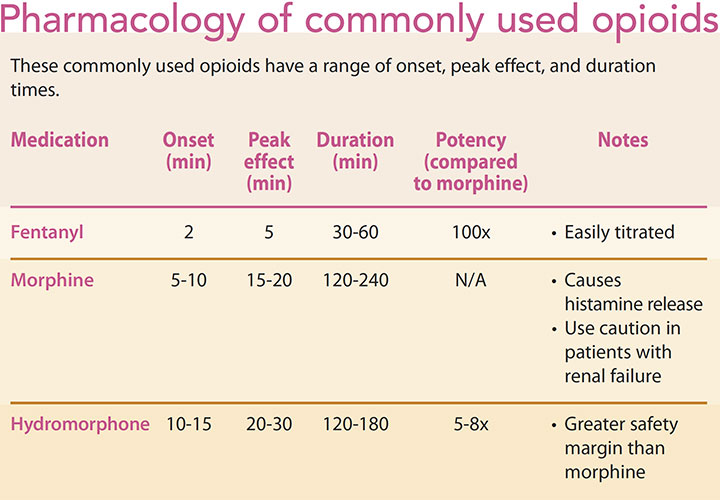

Fentanyl. Synthesized in the 1960s, fentanyl is 100 times more potent than morphine and easily titratable, making it the opioid of choice during surgery. With minimal effect on blood pressure and heart rate, fentanyl’s shorter duration of action gives it a relatively high safety margin. Fentanyl also is used for breakthrough pain after surgery.
Morphine. Morphine, which provides a high degree of analgesia, is commonly used as a first-line treatment for postoperative pain. However, like all opioids, it can cause adverse side effects. Morphine causes histamine release, which may result in respiratory distress or hypotension; patients with a history of asthma or cardiovascular disease are at particular risk. Because morphine is excreted through the kidneys, its active metabolite (morphine-3-glucuronide) may accumulate in patients with renal failure, placing them at risk for developing postoperative respiratory depression and seizures.
Hydromorphone. The advantages of hydromorphone over morphine are its faster onset and greater potency. Histamine release is rarely observed with hydromorphone, and it doesn’t possess metabolites that are risky to renal-compromised patients.
Less commonly used opioids
In the years after the advent of fentanyl, several analogues were synthesized: sufentanil, remifentanil, and alfentanil. Each of these drugs varies according to onset of action, duration of action, and potency. (See Pharmacology of less commonly used opioids.)
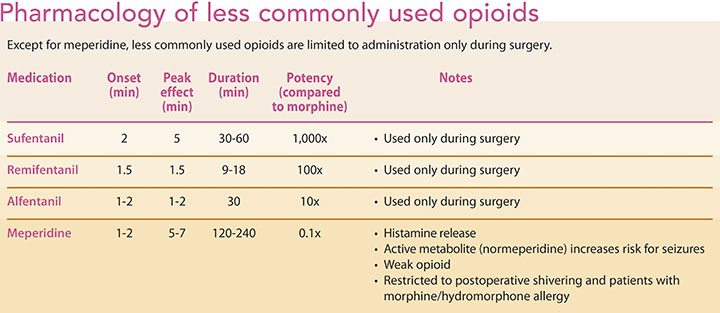

Sufentanil. With a profile similar to fentanyl, sufentanil is 1,000 times more potent than morphine, making it the strongest opioid available for use. It’s primarily indicated in cases that require a high degree of analgesia because of intense surgical stimulation, such as with cardiac and spinal surgery. Sufentanil’s duration of action is shorter than that of morphine and hydromorphone, but it produces a more rapid onset and better analgesia quality. Because the medication’s potency requires patient intubation, its use is confined primarily to the operating room (OR).
Remifentanil. With a potency 100 times greater than morphine, remifentanil is identical to fentanyl in terms of potency; however, it has a faster onset of action and is metabolized more quickly. Its use remains primarily restricted to the OR, and it is reserved for surgeries—such as head and neck, cardiac, spinal, craniotomy, and gastric bypass—that require a high degree of analgesia and a rapid recovery time after administration.
Remifentanil’s duration of action is the shortest of all the short-acting opioids because it’s rapidly metabolized through ester hydrolysis. A patient can be maintained on a high-dose remifentanil I.V. drip over several hours, but when the infusion stops, the drug is metabolized within 9 to 18 minutes with little to no side effects. Because of this, opioids such as hydromorphone or fentanyl are typically administered at the end of surgery to provide the patient with longer-acting analgesia that extends into postoperative care.
Alfentanil. Among all of the fentanyl analogues, alfentanil has the weakest potency (10 times greater than morphine), but the fastest onset. And although alfentanil provides excellent analgesic coverage, its duration of action is short. Its use is restricted primarily to airway management (intubation) as well as cases (such as head and neck surgery) that may be intensely stimulating but require blunting of the painful stimulus for only a short time. Because alfentanil doesn’t provide longer-acting analgesia, fentanyl or hydromorphone may be titrated at the end of surgery.
Meperidine. Originally developed as an anticholinergic drug, meperidine has long been used in surgical settings to treat moderate to severe pain because of its analgesic properties. Similar to other opioids, meperidine’s mechanism of action is through activation of mu receptors; however, it also activates kappa receptors, giving it the ability to treat the poorly understood phenomenon of postoperative shivering.
Recently, meperidine use began to fade because it causes histamine release and possesses an active metabolite (normeperidine) that places patients, especially those with renal failure, at risk for seizures. And because meperidine is an anticholinergic analogue, tachycardia may occur; care must be used when administering it to patients with cardiovascular disease. As a result of its high side-effect profile, meperidine use has been confined to treating postoperative shivering or as a last resort for patients who have an allergy to morphine or hydromorphone.
Non-opioid analgesics
Non-opioid analgesics used to treat moderate to severe pain include dexmedetomidine, I.V. acetaminophen, ketorolac, and ketamine. (See Pharmacology of non-opioid analgesics.)
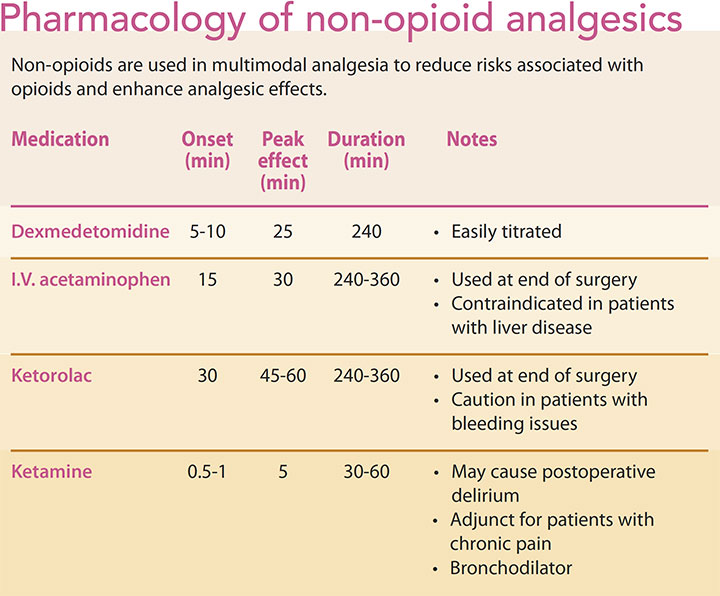

Dexmedetomidine. Dexmedetomidine is a sedative agent with analgesic properties. It’s reserved for patients who have a history of chronic pain or who are having surgery that’s expected to cause significant postoperative pain. Administered as an I.V. infusion, dexmedetomidine stimulates alpha-2 receptors. Unlike opioids, it minimally affects the respiratory system and airway reflexes. However, like sufentanil, remifentanil, and alfentanil, its use is primarily restricted to the OR. Because dexmedetomidine is delivered by I.V. infusion, providers rarely order it for postoperative pain management unless the patient is transferred to the intensive care unit for postoperative care, where he or she will be closely monitored.
I.V. acetaminophen. The mechanism of action of I.V. acetaminophen remains unclear, but it may inhibit cyclooxygenase (COX), an enzyme that plays a key role in prostaglandin synthesis, one of a cascade of inflammatory mediators released with surgical stimulation. Because I.V. acetaminophen bypasses the liver’s metabolic function, it provides significantly more analgesia than oral acetaminophen. However, use caution in patients with pre-existing liver disease; hepatotoxicity can occur if more than 4 g are administered to an adult within 24 hours. Pediatric dosing is weight based (10 to 15 mg/kg/dose) and shouldn’t exceed 2.6 g within 24 hours.
Ketorolac. The anesthesia provider may administer ketorolac at the conclusion of surgery. Like other nonsteroid anti-inflammatory drugs (NSAIDs), ketorolac inhibits prostaglandin synthesis by blocking COX. The most commonly administered dose is 30 mg (equivalent to 10 mg of morphine), but it should be decreased to 15 mg for patients 65 years and older who may have decreased kidney function. Ketorolac is excreted primarily through the kidneys, and accumulation of the medication can lead to acute renal failure, nephrotic syndrome, or nephritis. Ketorolac is contraindicated in patients with chronic kidney disease. Because ketorolac inhibits prostaglandin production, its use is contraindicated in patients with bleeding conditions.
Ketamine. Ketamine is a sedative-hypnotic agent that possesses unique analgesic properties that may be useful at low doses to treat patients with chronic pain. Because its mechanism of action occurs through the inhibition of N-methyl-D-aspartate pain receptors, anesthesia providers may administer ketamine to patients with a history of chronic pain. A growing body of evidence supports ketamine in treating postoperative pain. In addition to its potent analgesic effect, the drug maintains a patient’s airway reflexes and also acts as a bronchodilator.
Ketamine can produce a state known as dissociative anesthesia—characterized by catatonia, amnesia, and analgesia—that may result in postoperative delirium.
Nurses’ role
When you receive report from an anesthesia provider about a patient who’s received multiple pain medications during surgery, ask if the patient has a history of chronic pain. He or she may be on opioid therapy at home and have an increased tolerance to opioids. This high tolerance is particularly challenging for the anesthesia provider to treat because the mechanism behind chronic pain is poorly understood, and evidence shows that continued administration of opioids during surgery isn’t effective. Consequently, the provider may use adjuncts, including non-opioid medication, to provide adequate analgesic coverage.
As a nurse, you have the unique opportunity to teach patients about the correct use of opioids for pain control. For example, you can work with patients to establish a plan specific to their surgical and personal pain-management needs for the purpose of addressing their expectations while also appropriately managing their postoperative pain.
When you receive report from the anesthesia provider on Ms. Rodriguez, you anticipate that she may have issues with pain control because of her history of chronic lower back pain, long-term opioid use, and current spinal surgery. You understand that the anesthesia provider used a multimodal plan and expect a hydromorphone patient-controlled analgesic pump will be ordered to help Ms. Rodriguez manage her pain.
While Ms. Rodriguez is in the PACU, you closely monitor her vital signs for any potential side effects she may experience with her pain medications.
*Name is fictitious.
Huy Vo is a senior staff CRNA at the University of California, Los Angeles (UCLA) Medical Center; Erica Clayton is a staff CRNA at Kaiser Harbor City Medical Center in Los Angeles. Jessica Stolyarskaya is an administrative specialist at UCLA Medical Center.
Selected references
American Association of Nurse Anesthetists. A Holistic Approach to Pain Management: Integrated, Multimodal, and Interdisciplinary Treatment: Position Statement. 2016.
American Society of Anesthesiologists Task Force on Acute Pain Management. Practice guidelines for acute pain management in the perioperative setting: An updated report by the American Society of Anesthesiologists Task Force on Acute Pain Management. Anesthesiology. 2012;116(2):248-73.
Butterworth JF, Mackey DC, Wasnick JD. Analgesic agents. In: Morgan & Mikhail’s Clinical Anesthesiology. 5th ed. New York, NY: McGraw Hill; 2013: pp 189-98.
Chen L, Mao J. Nonopioid medications. In: Miller RD, ed. Miller’s Anesthesia. 8th ed. Philadelphia, PA: Saunders; 2015: pp 915-18.
Fukuda K. Opioid analgesics. In: Miller RD, ed. Miller’s Anesthesia. 8th ed. Philadelphia, PA: Saunders; 2014: pp 864-914.
Gulur P, Koury K, Arnstein P, et al. Morphine versus hydromorphone: Does choice of opioid influence outcomes? Pain Res Treat. 2015;482081.
McGaffigan P, Lorincz CY, Gandhi TK. Opioids: What do healthcare professionals want and need to know? Patient Safety & Quality Healthcare. April 13, 2016.
Nagelhout JJ. Intravenous induction agents. In: Nagelhout JJ, Elisha S, eds. Nurse Anesthesia. 6th ed. St. Louis: Elsevier; 2018; 93-109.
Nagelhout JJ. Opioid and non-opioid analgesics. In: Nagelhout JJ, Elisha S, eds.
Nurse Anesthesia. 6th ed. St. Louis: Elsevier; 2018; 128-39.
ant5-CE Analgesics and Anesthesia-416a



















1 Comment.
Would it be possible to list the study/reference that was used regarding the IV acetaminophen data in the article Opioid and non-opioid analgesia during surgery? Thank you.