Stay-up-to-date to avoid complications and achieve the best treatment for patients.
Takeaways:
- This article discusses the use of oral anticoagulants.
- Indications, side effects and dosing of oral anticoagulants.
- Discussion of oral anticoagulants and indications for drug therapy.
- Patient education for patients taking oral anticoagulants.
MILLIONS of people in the United States take anticoagulants to prevent deep venous thrombosis (DVT), pulmonary embolism (PE), and stroke associated with atrial fibrillation. The mainstays of anticoagulant therapy are heparin and warfarin. Both have disadvantages and limitations. For years warfarin was the only oral anticoagulant, but new medications are just as effective and have fewer disadvantages and limitations.
When you understand how anticoagulants work, you can ensure that patients achieve optimal outcomes and avoid complications.
3 steps of hemostasis
To promote hemostasis, the body engages three processes: vasoconstriction, platelet plug development, and clot formation.
After a vessel injury, vasoconstriction decreases the vessel diameter, reducing blood flow to the injured area. Platelets in the blood aggregate there and stick together to form a platelet plug. Platelets activate fibrin required to help form and maintain the plug. For a clot to form, two intersecting pathways must be initiated. (See Follow the pathway.)
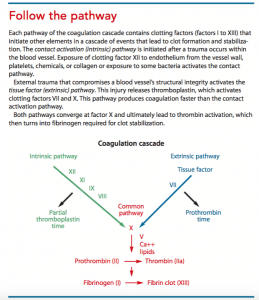

Anticoagulants alter hemostasis by inhibiting a component of the coagulation process. Different anticoagulants focus on different components in the process, but the outcome is either clot prevention or clot extension. Anticoagulants don’t dissolve a clot that’s already formed.
The oral anticoagulants currently available in the United States include warfarin, direct thrombin inhibitors, and direct factor Xa inhibitors. (See Oral anticoagulants: Pros and cons.)


Warfarin
Warfarin affects the four vitamin K–dependent clotting factors: II (prothrombin), VII, IX, and X. In the intestines, warfarin blocks vitamin K epoxide reductase, the enzyme required to convert vitamin K to its active form.
Pharmacokinetics: Warfarin is readily absorbed in the GI tract and metabolized by the liver using the cytochrome P-450 pathway through the CYP1A2, CYP2C9, and CYP3A4 systems. It’s excreted by the kidneys. When warfarin is in the blood circulation, 99% binds to the plasma protein albumin, which may delay its initial response by 8 to 12 hours. When discontinuing the medication, anticoagulant effects are observed for 2 to 5 days as the plasma-bound drug becomes free drug.
Therapeutic uses: Warfarin is primarily used to prevent DVT and PE, especially in patients with prosthetic heart valves and atrial fibrillation. It’s also used to reduce the risk of recurrent transient ischemic attacks and myocardial infarctions.
Dosage: Warfarin is available in various strengths, from 1-mg to 10-mg tablets. The required dosage is determined using prothrombin time/ international normalized ratio (PT/ INR) (a standardized method measuring the blood’s clotting ability). The normal INR is 1. The goal PT/ INR for anticoagulant therapy is 2 to 3.5 times baseline, depending on the condition being treated. Dosages are individualized, so establishing the goal INR can take several weeks because of warfarin’s long half-life. As needed, dosage adjustments are made during therapy based on PT/INR. PT/INR is measured daily during the first 5 days of treatment, two times a week for the next 1 to 2 weeks, and then every 2 to 4 weeks thereafter.
Drug monitoring can be done at an inpatient or outpatient lab or by the patient at home using handheld devices. Findings are monitored by a provider to evaluate whether the dosage should be maintained or changed. Handheld devices provide reliable PT/INR results to improve anticoagulant control, but they’re expensive and aren’t always covered by insurance.
Adverse effects: Warfarin’s main adverse effect is bleeding. When bleeding requires treatment (as opposed to simply discontinuing the medication), vitamin K1 (phytonadione) is used to reverse the drug’s impact on the development of vitamin K1-dependent clotting factors. Vitamin K1 is administered orally (preferred) or intravenously. When given by I.V., it should be diluted and administered slowly; a rapid infusion rate can cause severe anaphylactic reactions including flushing, hypotension, and cardiovascular collapse. The goal of vitamin K1 use is controlling excessive bleeding, not blocking all anticoagulation activity. Therefore, dosages should be small, no matter the administration route. Warfarin reversal with vitamin K1 can take several days.
In the past, patients were taught to avoid foods with vitamin K (for example, green leafy vegetables, Brussels sprouts, kale, spinach, collard greens, liver). Patients should be taught to maintain consistency in their consumption of foods with vitamin K, not avoid them. Inconsistency is what affects warfarin’s effectiveness.
Contraindications: Contraindications for warfarin include open wounds; active bleeding; severe thrombocytopenia; active ulcer disease; eye, brain, or spinal cord surgery; pregnancy; lactation; uncontrolled hypertension; severe vitamin K deficiency; liver disease; and alcoholism. Warfarin should be used with extreme caution in patients with hemophilia, increased capillary permeability, dissecting aneurysm, and women anticipating an abortion.
Drug-to-drug interactions: Warfarin should not be used with any drugs that will increase bleeding. Because warfarin is metabolized by cytochrome P-450 pathways, it interacts extensively with other medications. The cytochrome P-450 pathway involves a group of enzymes responsible for drug metabolism. When multiple drugs are metabolized through this pathway, metabolism and biotransformation of each of the drugs is altered and can result in changes in toxicity, pharmacologic impact, and drug-to-drug interactions.
Warfarin also has significant interactions with other drugs that bind to plasma proteins. These interactions will either increase bleeding and anticoagulant effects or decrease anticoagulant effects. Before administering warfarin, verify that the patient’s currently prescribed medications are safe to take with warfarin, and instruct the patient to contact his or her healthcare provider before taking any new medication or herbal supplement.
Direct thrombin inhibitors
Direct thrombin inhibitors include dabigatran etexilate, bivalirudin, desirudin, and argatroban. Only dabigatran is available for oral administration. Direct thrombin inhibitors block the action of thrombin circulating in the blood and already bound in a clot, preventing the conversion of fibrinogen to fibrin, the activation of factor XIII, the conversion of soluble fibrin to insoluble fibrin, and thrombin-mediated platelet activation and aggregation.
Pharmacokinetics: Dabigatran etexilate is rapidly absorbed in the GI tract and converted to its active form, dabigatran, by the gut and P-glycoprotein. Food, particularly a high-fat meal, delays dabigatran absorption, but doesn’t reduce the amount of drug absorbed. Peak drug concentrations occur in about 2 hours, and protein binding is minimal. Dabigatran is eliminated unchanged by the kidneys. The drug’s normal half-life is 12 to 17 hours and increases to 18 hours or more with renal impairment.
Therapeutic uses: Direct thrombin inhibitors prevent and treat DVT, stroke, and systemic embolisms in patients with nonvalvular atrial fibrillation (NVAF). They’re also prescribed following knee-or hip-replacement surgery and to treat DVT and PE in patients after treatment with a parenteral anticoagulant for 5 to 10 days.
Dosages: Direct thrombin inhibitor dosages are treatment specific. Instruct patients to swallow dabigatran capsules intact. If the capsule is opened, chewed, or crushed, the rate of absorption can be increased by 75%. Food doesn’t impact medication absorption. (See Dabigatran dosages and indications.)
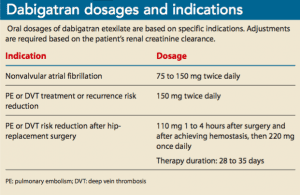

Adverse effects: Bleeding, erosive gastritis, hemorrhagic gastritis, and GI reflux, hemorrhage, and ulcers are the main adverse effects of dabigatran. Some GI symptoms may be reduced by taking a proton-pump inhibitor or histamine receptor blocker. If GI effects aren’t reduced, a different anticoagulant should be tried. Idarucizumab is a dabigatran-specific antidote for life-threatening bleeding or emergency surgery.
Contraindications: Dabigatran shouldn’t be given to patients who have active bleeding or a prosthetic heart valve, and patients who are undergoing surgery of the spinal cord, brain, or eye. Dabigatran shouldn’t be used during pregnancy.Drug-to-drug interactions: Increased or decreased effects of direct thrombin inhibitors are due to CYP pathway inhibition or induction. Concurrent administration of dabigatran with anticoagulants, antiplatelet agents, or nonsteroidal anti-inflammatory agents increases the risk of bleeding. Other drugs that can increase bleeding risk when given with dabigatran include cyclosporine, dronedarone, itraconazole, ketoconazole, tacrolimus, amiodarone, quinidine, erythromycin, verapamil, azole antifungals, and HIV protease inhibitors. Drugs and supplements that can decrease the effectiveness of dabigatran include carbamazepine, dexamethasone, fosphenytoin, phenytoin, rifampin, carbamazepine, dexamethasone, tipranavir, and St. John’s wort.
Direct factor Xa inhibitors
Direct factor Xa inhibitors (rivaroxaban, apixaban, edoxaban, and betrixaban) decrease thrombin production and prevent clot formation. Information about rivaroxaban, the first orally administered direct Xa inhibitor, is presented below. The other oral direct factor Xa inhibitors have similar actions and pharmacokinetics.
Pharmacokinetics: After oral administration, the bioavailability of rivaroxaban is high, even though it binds to protein. A peak plasma level occurs 2 to 4 hours after administration, and the drug has a half-life of 24 hours. Rivaroxaban is partially metabolized by the CYP3A4 isoenzyme in the cytochrome P-450 pathway and is a substrate for P- glycoprotein (a transporter involved in removing rivaroxaban from the body). Rivaroxaban elimination occurs via the urine and feces.Therapeutic uses: Direct factor Xa inhibitors are used to prevent PE and DVT after total hip- or knee-replacement surgery, prevent stroke and intravascular events in patients with NVAF, and treat DVT and PE.
Dosage: Direct factor Xa inhibitor dosage is treatment specific. These medications should be taken at approximately the same time daily, with or without food. (See Direct factor Xa inhibitor dosages and indications.)
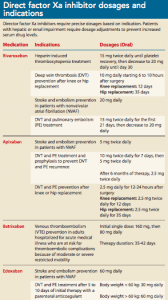

Adverse effects: Adverse effects of direct factor Xa inhibitors include bleeding, hematoma, dizziness, rash, GI distress, anemia, muscle spasms, and arm or leg pain. In addition, when rivaroxaban is discontinued before treatment completion, a thrombotic event or stroke may occur. To prevent this outcome, a different anticoagulant should be used to replace rivaroxaban when it’s discontinued.
Contraindications: All direct factor Xa inhibitors are contraindicated in patients with active bleeding. These medications should be discontinued at least 18 hours or two half-lives before spinal or epidural procedures. Patients with moderate or severe liver impairment (Child-Pugh B or C) or with any liver disease associated with coagulopathy should not take direct factor Xa inhibitors. Safe use of these medications in pregnancy hasn’t been established.
Drug-to-drug interactions: Drugs that inhibit or induce the CYP3A4 enzyme and P-glycoprotein (amiodarone, quinidine, diltiazem, verapamil, ranolazine, and macrolide antibiotics) can increase or decrease direct factor Xa inhibitor levels. Drugs and supplements that decrease the effects of direct inhibitors (carbamazepine, phenytoin, rifampin, and St. John’s wort) can potentially cause a thrombotic event. All medications that increase bleeding—including anticoagulants, antiplatelet agents, and nonsteroidal anti-inflammatory agents—are contraindicated.
Nursing considerations and discharge planning
Oral anticoagulant dosages are prescribed based on the focus of treatment, so medication errors (too much or too little medication) can have deleterious effects. Teach patients the signs and symptoms of bleeding (including increased bruising, petechia, hematomas, increased heart rate, decreased blood pressure, red or black stools, cloudy or discolored urine, headache, and pelvic or lumbar pain).
Discharge planning and patient education should include oral and written instructions about how patients can decrease the risk of bleeding.
- Take medication as prescribed.
- Follow provider instructions when a dosage is missed.
- Monitor for bleeding.
- Attend follow-up monitoring appointments if required.
Make lifestyle changes to reduce bleeding risk:
- shave with an electric razor
- use a soft-bristle toothbrush and waxed floss
- wear cut-resistant gloves when using knives, and keep knives sharpened to reduce slippage
- avoid activities and behaviors (playing contact sports, increased alcohol consumption) with the potential for falling or self-injury
- remove or rearrange items (furniture, area rugs) in the home that may pose an injury or fall risk
- wear a helmet when bike riding
- wear a medical alert bracelet
- contact a healthcare provider before taking any newly prescribed or over-the-counter medication, vitamin, or herbal supplement.
Stay current
Warfarin has been the mainstay of oral anticoagulant therapy for more than 50 years, but newer medications are replacing it as standard therapy. These newer oral anticoagulants work by directly inhibiting thrombin or factor Xa.
As the population ages, the use of oral anticoagulants to prevent DVT, PE, and stroke will increase.
Stay up-to-date about these medications’ indications and adverse effects to ensure the best treatment for your patients.
Nancy Haugen is associate dean, prelicensure and undergraduate programs, at the Samuel Merritt University School of Nursing in Oakland, California.
Selected references
Albertsen IE, Larsen TB, Rasmussen LH, Overvad TF, Lip GY. Prevention of venous thromboembolism with new oral anticoagu- lants versus standard pharmacological treatment in acute medically ill patients. A systematic review and meta-analysis. Drugs. 2012;72(13):1755-64.
Bajorek B, Saxton B, Anderson E, Chow CK. Patients’ preferences for new versus old anticoagulants: A mixed-method vignette- based study. Eur J Cardiovasc Nurs. 2018; 17(5):429-38.
Burcham JR, Rosenthal LD. Anticoagulant, antiplatelet, and thrombolytic drugs. In:Lehne’s Pharmacology for Nursing Care. 10th ed. Philadelphia, PA: Elsevier; 2018: 604-32.
Dans AL, Connolly SJ, Wallentin L, et al. Concomitant use of antiplatelet therapy with dabigatran or warfarin in the randomized evaluation of long-term anticoagulation therapy (RE-LY) trial. Circulation. 2013;127(5): 634-40.
Kearon C, Akl EA, Ornelas J, et al. Antithrombotic therapy for VTE disease: CHEST guideline and expert panel review. CHEST. 2016;149(2):315-52.
Kumar S, Danik SB, Altman RK, et al. Non- vitamin K antagonist oral anticoagulants and antiplatelet therapy for stroke prevention in patients with atrial fibrillation: A meta-analysis of randomized controlled trials. Cardiol Rev. 2016;24(5):218-23.
Maruyama K, Yamamoto T, Aoyagi H, et al. Difference between the upper and the lower gastrointestinal bleeding in patients taking nonvitamin K oral anticoagulants. Biomed Res Int. 2018:7123607.
Miller CS, Dorreen A, Martel M, Huynh T, Barkun AN. Risk of gastrointestinal bleeding in patients taking non–vitamin K antagonist oral anticoagulants: A systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2017;15(11):1674-83.
Providência R, Grove EL, Husted S, Barra S, Boveda S, Morais J. A meta-analysis of phase III randomized controlled trials with novel oral anticoagulants in atrial fibrillation: Comparisons between direct thrombin inhibitors vs. factor Xa inhibitors and different dosing regimens. Thromb Res. 2014;134(6):1253-64.
Skelley JW, Thomason AR, Nolen JC, Candidate P. Betrixaban (Bevyxxa): A direct-acting oral anticoagulant factor Xa inhibitor. P T. 2018;43(2):85-120.
Smith SA, Travers RJ, Morrissey JH. How it all starts: Initiation of the clotting cascade. Crit Rev Biochem Mol Biol. 2015;50(4):326-36.
Vallerand AH, Sanoski CA. Davis’s Drug Guide for Nurses. 16th ed. Philadelphia, PA: F.A. Davis Company; 2018.



















3 Comments.
This is amazing! We arent eating it for a couple more hours and I have yet to put in the coconut milk. But what I have tasted is awesome! Thanks for the recipe!!!
You can take the test by clicking on the box below the article that says Register & take test.
How do I do the post-test online?