Good outcomes require early identification and prompt treatment.
Takeaways:
- Early reperfusion and medical therapy have drastically reduced acute myocardial infarction complications.
- Despite advanced therapies, complications continue to arise.
- Nurses play an integral role in prompt identification of complications related to an acute myocardial infarction and initiating early treatment to prevent long-term adverse outcomes.
CNE
1.41 contact Hours
Learning Objectives
- Describe complications of acute myocardial infarction (AMI).
- Discuss nursing care related to complications of AMI.
The author and planners of this CNE activity have disclosed no relevant financial relationships with any commercial companies pertaining to this activity. See the last page of the article to learn how to earn CNE credit.
Expiration: 4/1/23
About every 40 seconds in the United States, someone experiences an acute myocardial infarction (AMI)—that adds up to about 600,000 AMIs every year. This potentially debilitating condition, which results from myocardial cell death caused by prolonged ischemia, is associated with serious complications and mortality. Early reperfusion and medical therapy have drastically reduced AMI complications, but they still occur. Nurses play an integral role in identifying and managing AMIs and their complications.
AMI care
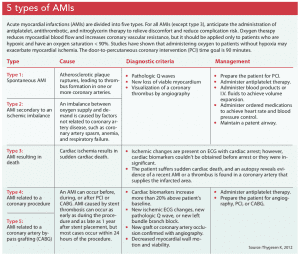
The goals of care for patients with an AMI are prompt symptom recognition and reperfusion therapy to help diminish complications. Identifying and managing an AMI begins when the patient first encounters a medical professional. If a patient reports ischemic symptoms (such as pain in the chest, left arm, or jaw; epigastric discomfort; dyspnea; fatigue; nausea; vomiting; and/or syncope), obtain a 12-lead ECG within 10 minutes of arrival. Other diagnostic evaluations will include laboratory tests for cardiac biomarkers (cardiac troponin 1 and cardiac troponin T) and imaging to assess myocardial wall viability and motion. (See 5 types of AMIs.)
Complications
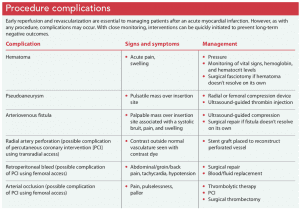
In addition to complications related to thrombolytic reperfusion therapy, patients are at risk for mechanical, electrical, pericardial, and procedural complications, as well as heart failure. (See Procedure complications.)
Mechanical complications
Mechanical complications associated with AMI include ventricular septal rupture (VSR), papillary muscle rupture, and ventricular free wall rupture.
VSR. The incidence of VSR is less than 1%, but because it can quickly progress to left and right ventricular dysfunction and cardiogenic shock, prompt recognition and treatment are necessary. VSR is caused by ischemic necrosis after an AMI; it’s more likely to occur in individuals more than 65 years old, as well as those with hypertension, elevated body mass index, female gender, and no previous history of AMI.
Presentation and diagnosis. Patients will present with either mild symptoms (such as dyspnea, pansystolic murmur best heard at the left sternal border, and a palpable parasternal thrill) or severe symptoms (such as hypotension, oliguria, and pulmonary edema). The gold standard for diagnosis is an echocardiogram, which will identify the size and location of the rupture. If echocardiogram data are unclear, the patient will undergo a right heart catheterization to detect altered left- and right-side pressures caused by shunting of blood from the left to the right ventricle.
Treatment. The goal of medical treatment is airway maintenance and afterload reduction before surgical treatment. The patient may require intubation while being stabilized with angiotensin-converting enzyme (ACE) inhibitors, hydralazine, and phosphodiesterase-3 inhibitors (such as milrinone). Afterload can be reduced by inserting an intra-aortic balloon pump (IABP).
Surgical closure is the definitive treatment for VSR, and mortality and long-term negative outcomes are reduced when coronary artery bypass grafting is accomplished at the same time. Postsurgery care goals include optimizing cardiac output and reversing end-organ dysfunction. IABP, positive inotropes, antiarrhythmics, and vasodilators will be prescribed to reduce preload and afterload.
Papillary muscle rupture. Like VSR, papillary muscle rupture occurs after altered ventricular function as a result of a lack of blood flow. The papillary muscles work to prevent regurgitation, but abnormal ventricular wall function may lead to mitral regurgitation. Mitral regurgitation secondary to papillary muscle rupture is more likely to occur after an inferior AMI because of the single blood supply; the anterolateral papillary muscle is more protected because of its dual blood supply from the left anterior descending and circumflex coronary arteries.
Presentation and diagnosis. Patients will have a new pansystolic murmur and symptoms of pulmonary edema, hypotension, and potential cardiogenic shock. An echocardiogram will be performed to diagnosis the rupture.
Treatment. After the diagnosis is confirmed, the patient will be started on afterload reduction medications such as vasodilators and inodilators. An IABP also may be inserted to achieve afterload reduction. Although these interventions will help stabilize the patient, surgical intervention is necessary to reverse the mitral regurgitation. Postoperative care is the same as the care provided to a patient after VSR surgical repair.
Ventricular free wall rupture. Ventricular free wall ruptures are categorized into three types. Type I occurs when a clot lodges in a ventricular wall tear or after thrombolysis treatment. Necrotic tissue develops, leading to a “blowout rupture.” Type II occurs with erosion between infarcted and normal tissue. Type III occurs when the myocardium becomes thinner than normal and the center of a ventricular aneurysm perforates.
Presentation and diagnosis. Patients will have varying symptoms depending on the type of rupture. Hypotension, nausea, chest pain, and low cardiac output commonly occur in patients experiencing a type II or III rupture; patients with a type I rupture may experience sudden death. After diagnosis is confirmed with an echocardiogram, the patient will be taken for emergent surgery.
Treatment. Ventricular free wall ruptures can be repaired using different techniques. One method is a pericardiocentesis, which may provide short-term hemodynamic stability but can cause adverse effects that may potentially harm the patient. The more traditional approach is direct suture of the myocardium over the infarcted tissue. After the rupture is repaired, angiography is completed.
Electrical complications
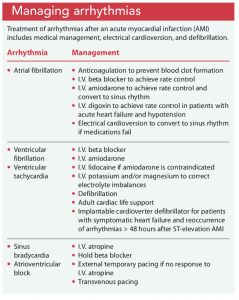
Although early reperfusion reduces the incidence of arrhythmias and sudden cardiac death, patients may experience life-threatening arrhythmias after an AMI.
Presentation and diagnosis. Patients experiencing arrhythmias may present with fatigue, weakness, dizziness, syncope, dyspnea, palpitations, and/or chest pain. Initiate continuous ECG monitoring on admission to watch for atrial fibrillation, ventricular tachycardia, ventricular fibrillation, sinus bradycardia, or a heart block. Some patients may have pre-existing arrhythmias, while others will experience new-onset arrhythmias caused by ischemia, electrolyte imbalances, or restoration of coronary blood flow.
Treatment. In the event of new onset arrhythmias, the goal is achieving and maintaining hemodynamic stability with beta blockers and antiarrhythmics and correcting underlying causes such as electrolyte imbalance and ischemic cardiac disease. Anticoagulants may be given to prevent thrombosis. In some cases, electrical cardioversion or defibrillation is required. (See Managing arrhythmias.)
Pericardial complications
The pericardium is an elastic sac that surrounds the heart, protecting it from infection and trauma. It has two layers that are separated by a small amount of fluid. An AMI can cause inflammation of the pericardium (pericarditis). Peri-infarction pericarditis and Dressler syndrome are forms of pericarditis that can develop after an AMI. Peri-infarction pericarditis develops soon after an AMI; Dressler syndrome occurs anywhere between 2 and 6 weeks after.
Presentation and diagnosis. Symptoms of peri-infarction pericarditis and Dressler syndrome include pleuritic chest pain, pericardial friction rub, ECG changes, pericardial effusion, fever, malaise, decreased appetite, irritability, palpitations, and dyspnea. Pleuritic chest pain differs from chest pain experienced with an AMI; it’s sharp, centralized, and relieved by sitting up and leaning forward. A pericardial friction rub (which sounds like superficial scratching) is best heard by placing a stethoscope over the left sternal border. An echocardiogram is performed to evaluate pericardial fluid and make a diagnosis. Other diagnostic imaging procedures include cardiac magnetic resonance imaging, chest X-ray, and ECG.
Treatment. Management focuses on keeping the patient comfortable and monitoring for cardiac tamponade, which can occur as pericardial fluid accumulates and compresses the heart’s chambers. Nonsteroidal anti-inflammatory drugs (NSAIDs)—such as aspirin, ibuprofen, and naproxen—are administered to reduce inflammation. If NSAIDs are unsuccessful, corticosteroids can be tapered over 4 weeks.
Heart failure
Patients who’ve had an AMI may experience new-onset heart failure, which can progress to cardiogenic shock.
New-onset heart failure. Approximately 60% of patients 65 years and older will develop heart failure within 2 years of an AMI. Heart failure increases the patient’s risk for mortality, making it an important condition to identify and treat immediately; however, the treatment plan can be challenging because it differs from typical AMI treatment.
Presentation and diagnosis. Patients with new-onset heart failure will report exertional dyspnea, orthopnea, edema, syncope, and fatigue. Left ventricular diastolic pressure and pulmonary artery wedge pressure are assessed via heart catheterization. An echocardiogram will help determine the patient’s ejection fraction. Diagnostic blood tests include a comprehensive metabolic panel, complete blood count, and type B natriuretic peptide.
Treatment. Typical pharmacologic treatment for a patient admitted to the hospital with an AMI includes initiation of a beta blocker within 24 hours; however, for patients with an AMI complicated by heart failure, beta blockers aren’t recommended. Instead, an ACE inhibitor is administered within the first 24 hours, unless otherwise contraindicated (for example, if the patient has a history of anaphylaxis related to an ACE inhibitor, bilateral renal artery stenosis, or acute kidney failure). For these patients, an angiotensin receptor blocker will be ordered. Early initiation of an aldosterone antagonist reduces the patient’s risk for mortality and sudden cardiac death. To ensure treatment adherence, provide the patient with a medication list containing the indications and potential side effects.
Educate the patient and caregiver about common signs and symptoms of heart failure, such as shortness of breath, fatigue, edema, dizziness, palpitations, persistent cough, and a decrease in appetite. Encourage the patient to obtain a daily weight every morning after emptying his or her bladder and record it to identify weight fluctuations. Provide instruction on healthy lifestyle choices, including daily physical activity, limiting salt intake, and smoking cessation.

Presentation and diagnosis. Patients have signs of decompensation and low perfusion, such as hypotension, weak pulse, decreased urine output, cool extremities, and mental status changes. An echocardiogram is performed to assess ventricular function and determine the cause.
Treatment. Early revascularization, with either percutaneous coronary intervention or coronary artery bypass grafting, serves as the most beneficial treatment option for patients with cardiogenic shock. Guidelines recommend invasive cardiac monitoring with a pulmonary artery catheter for continuous cardiac pressures readings to help guide treatment.
The treatment plan focuses on improving organ function and patient stability. Intravenous inotropes and vasopressors, such as dobutamine and norepinephrine, are administered to improve hypotension, cardiac output, and organ function. When adequate perfusion is achieved, diuretic therapy will be initiated. If the patient doesn’t respond well to diuretic therapy, ultrafiltration will be considered. Temporary mechanical circulatory support, such as an IABP or extracorporeal membrane oxygenation, is used to achieve immediate stabilization.
Preventing future AMIs
Advances in medical therapy have drastically reduced the complications associated with AMI, but they remain a leading cause of death. Even as you are managing the complication, start preparing the patient and caregiver for discharge. Depending on the type of complication, education may be complex. Developing a therapeutic and trusting relationship with the patient and caregiver can help promote adherence to a comprehensive treatment plan.
Discuss behavioral and pharmacologic recommendations to help prevent another AMI. Recommendations may include dietary modifications, blood pressure management, and cardiac rehabilitation. Encourage the patient to participate in exercise programs to promote weight loss and heart health. Review signs and symptoms of an impending AMI, and remind the patient and caregiver to seek immediate emergency treatment if symptoms occur.
Kayla M. Little is a clinical nurse specialist in cardiovascular medicine at the Cleveland Clinic in Cleveland, Ohio.
References
Al-Khatib SM, Stevenson WG, Ackerman MJ, et al. 2017 AHA/ACC/HRS guideline for management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: Executive summary: A report of the American College of Cardiology/American Heart Association task force on clinical practice guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2018;72(14):e91-220.
Anderson JL, Morrow DA. Acute myocardial infarction. N Engl J Med. 2017;376(21):2053-64.
Bahit MC, Kochar A, Granger CB. Post-myocardial infarction heart failure. JACC Heart Fail. 2018;6(3):179-86.
Barker T, Ramnarine I, Woo E, et al. Repair of post-infarct ventricular septal defect with or without coronary artery bypass grafting in the northwest of England: A 5-year multi-institutional experience. Eur J Cardiothoracic Surg. 2003;24:940-6.
Burton LV, Beier K. Papillary muscle rupture. StatPearls. June 4, 2019. ncbi.nlm.nih.gov/books/NBK499976
Benjamin EJ, Muntner P, Alonso A, et al. Heart disease and stroke statistics—2019 update: A report from the American Heart Association. Circulation. 2019;139(10):e56-528.
Ibanez B, James S, Agewall S, et al. 2017 ESC guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation: The task force for the management of acute myocardial infarction in patients presenting with ST-segment elevation of the European Society of Cardiology (ESC). Eur Heart J. 2018;39(2):119-77.
Guensch DP, Fischer K, Shie N, Lebel J, Friedrich MG. Hyperoxia exacerbates myocardial ischemia in the presence of acute coronary artery stenosis in swine. Circ Cardiovasc Interv. 2015;8(10):e002928.
Hofmann R, Witt N, Lagerqvist B, et al. Oxygen therapy in ST-elevation myocardial infarction. Eur Heart J. 2018;39(29):2730-9.
Korczyk D, Kaye G. Fast Facts: Heart Failure. 2nd ed. Oxford, United Kingdom: Health Press Limited; 2017.
Kutty RS, Jones N, Moorjani N. Mechanical complications of acute myocardial infarction. Cardiol Clin. 2013;31(4):519-31.
Leib AD, Foris LA, Nguyen T, Khaddour K. Dressler syndrome. StatPearls. July 4, 2019. ncbi.nlm.nih.gov/books/NBK441988
Montrief T, Davis WT, Koyfman A, Long B. Mechanical, inflammatory, and embolic complications of myocardial infarction: An emergency medicine review. Am J Emerg Med. 2019;37(6):1175-83.
O’Gara PT, Kushner FG, Ascheim DD, et al. 2013 ACCF/AHA guideline for the management of ST-elevation myocardial infarction: A report of the American College of Cardiology Foundation/American Heart Association task force on practice guidelines. Circulation. 2013;127(4):e362-425.
Stub D, Smith K, Bernard S, et al. Air versus oxygen in ST-segment-elevation myocardial infarction. Circulation. 2015;131(24):2143-50.
Thygesen K, Alpert JS, Jaffe AS, et al. Third universal definition of myocardial infarction. Circulation. 2012;126(16):2020-35.
Thygesen K, Alpert JS, Jaffe AS, et al. Fourth universal definition of myocardial infarction. Circulation. 2018;138(20):e618-51.




















4 Comments.
I would like to obtain the post test and receive contact hours. Where do I find it?
Thank you, Theresa
Please visit the ANA’s website to take the CE: https://www.nursingworld.org/continuing-education/recognizing-and-managing-complications-of-acute-my/
good morning, I am wondering how to obtain the post test, and contact hours certificate after I complete the course. Thank-you, Karen
Please visit the ANA’s website to take the CE: https://www.nursingworld.org/continuing-education/recognizing-and-managing-complications-of-acute-my/