Be sure your knowledge is current.
Takeaways:
- Insulin is a critical part of diabetes management for many people.
- Insulin types and delivery methods have changed drastically in the last decade.
- Nurses have an important role in teaching patients about insulin and helping them improve outcomes.
CNE 1.5 contact hours
Learning Objectives
- Differentiate types of insulin.
- Describe insulin-related complications.
- Discuss the nurse’s role in helping patients manage their diabetes.
The author and planners of this CNE activity have disclosed no relevant financial relationships with any commercial companies pertaining to this activity. See the last page of the article to learn how to earn CNE credit.
Expiration: 6/1/23
Approximately 34 million people in the United States have diabetes; 90% to 95% of them have type 2 diabetes. Insulin saves lives and helps people with both type 1 and type 2 diabetes live longer than they would without it, but it isn’t a cure; it’s a treatment. The types of insulin available and how it is delivered have evolved, as have treatment goals.
History of insulin
In 1922, about a year after insulin was first isolated and extracted from pancreatic islet cells, it became commercially available. It was roughly equivalent to today’s Regular insulin, and it was injected subcutaneously several times a day to manage glucose levels. In the 1930s, scientists discovered that adding protein to Regular insulin made it last longer, so many people began taking Regular insulin plus a longer-acting insulin with an added protein, with the goal of managing glucose levels all day and through the night.
Since then, different types of insulin (lente, NPH, semilente, ultralente) and timing protocols have been tried, such as once-a-day injections; twice a day; and Regular and NPH in the morning, Regular at dinner, and NPH at bedtime. In the 1980s, researchers began making insulin in a laboratory using recombinant DNA procedures, resulting in insulin that is amino acid for amino acid the same as human insulin. Before that, all insulin came from cow and pig sources, which is no longer the case in the United States. In the 1990s, insulin analogs were introduced. Analogs are altered in the lab so they work faster or slower, resulting in rapid-acting and long-acting insulins.
When insulin is used
Anyone with type 1 diabetes requires insulin to survive, and many people with type 2 diabetes may progress to needing insulin for effective disease management and to lower their risk for long-term complications. (See Types of diabetes.)
In 1993, results of the landmark Diabetes Control and Complications Trial (DCCT) supported the value of managing diabetes to prevent or delay complications. This study introduced carbohydrate counting and basal/bolus insulin dosing. Healthcare providers teach people with diabetes to keep blood glucose levels in a target range and to avoid variability (bouncing levels) as much as possible.
Each person’s insulin plan should be individualized to his or her unique needs. Some people with type 1 diabetes use multiple daily injections, and others use insulin pumps to deliver insulin around the clock. Either approach can utilize basal/bolus dosing. With multiple daily injections, people take a rapid-acting insulin (lispro, aspart, or glulisine) with breakfast, lunch, dinner, and snacks and then a long-acting insulin (glargine, detemir, or degludec) once or twice a day (typically at least 12 hours apart). The long-acting insulin serves as the basal (background) dose, which works all day and night. The rapid-acting insulin is the bolus dose given with meals and to bring down elevated glucose levels. Many people with type 2 diabetes take only a long-acting insulin at bedtime.
Type 1 diabetes is an autoimmune disease in which the body’s immune system mistakenly attacks the pancreas cells that produce insulin. People with type 1 diabetes don’t make enough insulin to survive and must take exogenous insulin.
Type 2 diabetes occurs when pancreas beta cells still produce insulin, but muscle, liver, and fat cells don’t use it efficiently (insulin resistance).
Latent autoimmune diabetes in adults (LADA) is a slower onset type 1 diabetes. Many older adults with LADA are misdiagnosed with type 2 diabetes because they don’t present with the acute signs typical of type 1 diabetes (extreme thirst, frequent urination, and weight loss). They’re frequently prescribed oral medication instead of the insulin they need. People with LADA who are misdiagnosed with type 2 diabetes tend to get sicker until they develop diabetes-related ketoacidosis.
To help avoid misdiagnosis of LADA, one of the most important questions a nurse can ask a person who presents with elevated blood glucose levels is, “Have you lost weight without trying?” If someone responds yes (or says they were trying to lose weight and were excited by how quickly the pounds were coming off), this can indicate type 1 diabetes (burning fat because not enough insulin is available).
The next step is to send blood for autoantibodies to determine if it is, indeed, autoimmune diabetes. People with LADA need to have a correct diagnosis so they can manage their diabetes effectively. If they are taking oral medications and even basal insulin, they may not be able to achieve their A1c goal, which increases their risk for long-term complications.
Types of insulin
Insulins are classified by how fast they work, when they have their onset of action, their peak effect, and their duration of action. Insulin action profiles are estimates; each may differ slightly and also may differ for individual people. (See Types of insulin.)
Recently, insulin manufacturers have introduced “biosimilars” and insulin “follow-ons” (such as Basaglar). These insulins may fill the need for a generic insulin. They work the same as more expensive insulin analogs for a slightly lower price.
Insulin delivery
Insulin is delivered in the subcutaneous tissue either by injection using an insulin syringe or pen, or with an insulin pump (continuous subcutaneous insulin infusion). Most people in the United States take U-100 insulin, although some take U-200, U-300, or U-500 because these insulins are more concentrated (they may last longer or require smaller doses for people who take large amounts of insulin each day).
Research shows that people of any size can use short (even 6 mm or shorter) insulin needles because skin and subcutaneous tissue thickness don’t differ much from person to person. Syringes come in sizes that can hold 25 to 100 units of insulin. Small children and the elderly may need half-units; the smallest syringes typically can accommodate those doses.
Nurses should instruct patients to rotate insulin injection sites (abdomen, arms, hips, buttocks, and legs) and to use new needles to avoid damaging skin. Some people may develop injection-site lipohypertrophy (lumps under the skin) that affects the insulin absorption rate. Using a larger area within injection sites and rotating sites can help manage or avoid lipohypertrophy.
Insulin types are categorized by their onset of action, peak effect, and duration.
| Type of insulin | Name of insulin | Action |
| Rapid-acting |
|
|
| Short-acting |
|
|
| Intermediate-acting |
|
|
| Long-acting |
|
|
Insulin pumps, which have been available since the late 1970s, have gotten smaller and more effective over time. They deliver insulin around the clock with preprogrammed basal settings and manually entered mealtime and correction bolus doses. People using insulin pumps enter the number of carbohydrate grams they’re about to eat and their glucose level, and the pump calculates the dose, based on settings (insulin-to-carbohydrate ratio and “sensitivity” or “correction” factor) programmed into the device. The pump then uses these settings and built-in algorithms to suggest how much insulin the person should deliver via the pump.
More and more people manage type 1 and type 2 diabetes with insulin pumps, so nurses working in hospital settings should be familiar with them. Ideally, an insulin pump is continued during hospitalization (to maintain glucose levels in the target range and maximize healing), although some circumstances (impaired consciousness or cognition) may require their removal.
Goals and monitoring
The goal in diabetes management is to keep blood glucose levels in a target range (identified by each patient in partnership with his or her diabetes care team) for as long as possible. In general (and for research purposes), this range is considered to be 70 mg/dL to 180 mg/dL; however, people living with diabetes may use a different range, based on age, comorbidities, additional medications, or other individual factors.
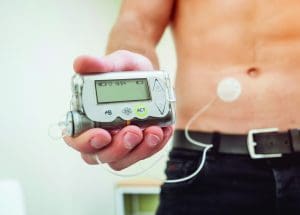

CGM systems, which have become more readily available in recent years, enable people to look at what percent of time they are in their target range, sometimes referred to as “time in range.” They allow someone with diabetes to see glucose patterns and make changes accordingly. This technology can be used with insulin injections as well as pumps and has opened up new horizons for managing diabetes, including the development of closed loop pumps and fine-tuning injection dosing. (See Closed-loop pumps.)
In 2016, the first hybrid closed-loop insulin pump became available. The “loop” refers to the connection between a continuous glucose monitor and a compatible insulin pump; insulin doses are calculated either manually or automatically. Hybrid closed-loop and fully closed-loop systems are becoming more readily available as they receive Food and Drug Administration approval.
Do-it-yourself (DIY) “loop” systems have gained traction recently. Individuals have pieced together older insulin pumps and algorithms to manage their doses. Some commercial insulin pump companies are combining their pumps with “loop” systems. Many people are discussing these DIY systems on social media, and some health professionals are skeptical because no standards have yet been published.
The development of closed-loop pumps is evolving rapidly, so nurses need to ensure their knowledge in this area is current.
Insulin-related complications
The limiting factor for taking insulin is hypoglycemia (low blood glucose). Hypoglycemia, which can lead to falls, passing out, seizures, or in rare cases, death, typically occurs when someone takes his or her usual insulin and then eats less food or engages in more physical activity than planned. People who take insulin should always carry glucose tablets, juice, nonchocolate candy, fruit, or other fast-acting carbohydrate sources with them. Because insulin is a “storage hormone,” people may gain weight when taking it. However, they can maintain an ideal goal weight with balanced insulin doses, healthy food choices and portions, and regular physical activity.
People also may experience hyperglycemia (high blood glucose), which comes with its own set of problems, especially if left untreated, when it can lead to diabetes-related ketoacidosis (DKA). People with type 2 diabetes typically make enough insulin to avoid DKA, which occurs when insulin isn’t available to pull glucose into cells for energy. Instead, the body burns fat for energy. Ketones, the byproducts of fat metabolism, build up in the bloodstream and cause a pH imbalance that can lead to serious illness or even death.
People with type 1 diabetes can experience DKA when they don’t have enough insulin on board; for example, if they have an underlying illness, infection, or other acute stressor. DKA also can occur if an insulin pump malfunctions and insulin isn’t properly delivered or when people receive inadequate diabetes education and don’t know how to manage their disease. Ask patients about their access to supplies and insulin and if they’ve been taught to properly store, draw up, and administer insulin or use a pump. If necessary, refer them to appropriate support services, including a certified diabetes care and education specialist.
Other issues
Other issues related to taking insulin include possible emotional toll, mistaken beliefs about its effects, and rationing.
In the past, many people with type 2 diabetes who required insulin were made to feel as though they had done something wrong. Some people still may feel ashamed if they need insulin. However, at least eight factors (including genetics) can contribute to a person developing type 2 diabetes, and only a few of them are modifiable. Nurses should approach patients with compassion and remind them that their disease and medication requirements are not their fault. When speaking with them about factors they can modify (such as diet and exercise), nurses should do so without judgment.
When people avoid or delay taking insulin, they may experience serious health problems, including limb loss and blindness. Family members and loved ones who witness this may mistakenly believe insulin caused the devastating outcome. In addition to teaching people how to take insulin safely and effectively, nurses must teach them (and their families) about the value of insulin, including that it can help them maintain their health, feel better, and avoid complications.
Insulin is very expensive. Some people with type 1 diabetes report insulin “rationing” because of the medication’s high cost. Rationing can result in DKA and even death. Discussions among people with diabetes, government leaders, industry leaders, healthcare professionals, and others are ongoing about how insulin gets priced and into the hands of people who need it. Different groups, including the American Diabetes Association, are working to address the situation. Insulin manufacturers have put assistance programs in place. Store-brand insulin (typically Regular and NPH) is available in limited types for a lower price at some pharmacies, and discount coupons are available. Diabetes care and education specialists can help nurses obtain information about these programs for patients.
Nursing role
In addition to providing education, nurses should assist people with decision-making and teach them about the value of managing their diabetes. The goals of taking medications, staying active, making consistent food choices, and checking glucose levels are important for maintaining target blood glucose levels. The result can be a lower risk for or delay of diabetes complications, including eye disease, kidney disease, nerve damage, and heart disease. Nurses should ask people how treatment and lifestyle modifications are working for them. What are their fears and challenges? Can they pay for their medications and insulin? Do they have the supplies they need? Do they have transportation to appointments? What do they need help with? Nurses can then provide referrals and recommend resources as needed. Nurses also should be sensitive to the language that they use. (See Words matter.)
Language can be a powerful tool for enhancing the motivation, health, and well-being of people with diabetes. Using respectful, nonstigmatizing language can decrease psychological distress (for example, guilt and shame) and lead people with diabetes to engage in self-care and seek follow-up care.
A task force consisting of representatives from the American Association of Diabetes Educators and the American Diabetes Association recommended that clinicians use language that:
- is neutral, nonjudgmental, and based on facts, actions, or physiology/biology (for example, “manage” rather than “control”)
- is free from stigma (for example, avoid “compliance” and “adherence”)
- is strengths based, respectful, inclusive, and imparts hope (for example, instead of focusing on potential complications, focus on the fact that people with diabetes are living long and healthy lives and then work collaboratively to develop a plan that works for the person)
- fosters collaboration between patients and providers (for example, avoid language that evokes authority such as “should/shouldn’t” and “must/must not”)
- is person centered (for example, “person with diabetes” not “diabetic”).
Source: Dickinson et al. 2017
The future of diabetes and insulin
Since the early 2000s, work has been underway on an artificial or “bionic” pancreas, which is a closed loop pump that doesn’t require input from the patient. And some researchers are investigating a “smart insulin” that when injected can determine how much someone needs and when. However, living with diabetes still requires people to be aware of what they’re doing, eating, and drinking. Until diabetes can be prevented or cured, nurses must work effectively with and advocate for patients affected by the disease so they can benefit from insulin’s life-saving potential.
Jane K. Dickinson directs and teaches in the solely online Master of Science in Diabetes Education and Management program at Teachers College, Columbia University in New York, New York.
References
American Diabetes Association. Standards of medical care in diabetes—2020. Diabetes Care. 2020;43(suppl 1):S1-212.
Bliss M. The Discovery of Insulin. Toronto: McClelland & Stewart; 1982.
Cefalu WT, Dawes DE, Gavlak G, et al. Insulin access and affordability working group: Conclusions and recommendations. Diabetes Care. 2018;41(6):1299-1311.
Centers for Disease Control and Prevention. National Diabetes Statistics Report, 2020. cdc.gov/diabetes/library/features/diabetes-stat-report.htm
DeFronzo RA. From the triumvirate to the ominous octet: A new paradigm for the treatment of type 2 diabetes mellitus. Diabetes. 2009;58(4):773-95.
Diabetes Control and Complications Trial Research Group; Nathan DM, Genuth S, Lachin J, et al. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med. 1993;329(14):977-86.
Dickinson JK, Guzman SJ, Maryniuk MD, et al. The use of language in diabetes care and education. Diabetes Care. 2017;40(12):1790-9.
Frid A, Hirsch L, Gaspar R, et al. The third injection technique workshop in Athens (TITAN). Diabetes Metab. 2010;36(suppl 2):S19-29.
JDRF. Glucose-responsive insulin. jdrf.org.uk/our-research/about-our-research/treat/smart-insulin
Kwon J, Brown A. Omnipod first insulin pump partner with Tidepool Loop. diaTribe Learn. November 20, 2018. diatribe.org/omnipod-first-insulin-pump-partner-tidepool-loop
Kwon J, Kennedy L, Brown A, Close K. Glycemic outcomes beyond A1c: Consensus on measuring what matters. diaTribe Learn. July 22, 2017. diatribe.org/measuringwhatmatters
Quianzon CC, Cheikh I. History of insulin. J Community Hosp Intern Med Perspect. 2012;2(2). tandfonline.com/doi/full/10.3402/jchimp.v2i2.18701
Runge A, Brown A. The FDA approves Medtronic’s MiniMed 670G hybrid closed loop system. diaTribe Learn. October 12, 2016. diatribe.org/fda-approval-medtronic-minimed-670g-hybrid-closed-loop-system
Schaeffer R. Beta Bionics receives FDA approval to begin bionic pancreas study recruitment. Healio. May 21, 2018. healio.com/endocrinology/diabetes/news/online/%7B227e7a76-f5fa-4ed7-a6ad-fa7f22dbdbad%7D/beta-bionics-receives-fda-approval-to-begin-bionic-pancreas-study-recruitment
Sherr J, Tamborlane WV. Past, present, and future of insulin pump therapy: Better shot at diabetes control. Mt Sinai J Med. 2008;75(4):352-61.
Sisson EM, Zimmerman, KM. Pharmacology for glucose management. In: Cornell S, Halstenson C, Miller DK, eds. The Art and Science of Diabetes Self-Management Education Desk Reference. 4th ed. Chicago: American Association of Diabetes Educators; 2017: 469-517.
Umpierrez GE, Klonoff DC. Diabetes technology update: Use of insulin pumps and continuous glucose monitoring in the hospital. Diabetes Care. 2018;41(8):1579-89.
White J, Goldman J. Biosimilar and follow-on insulin: The ins, outs, and interchangeability. J Pharm Technol. 2019;35(1):25-35.



















8 Comments.
Very informative article…Thank You
I am already registered but got locked out and now I am unable to get back in to take the test. WHAT do I do????
Excellent review of newest insulins and management of diabetes.
Very good article/good information
a great refresher article for any level provider
Timely information.
Good read
thank you.