Use, misuse, complications, and withdrawal
- The increased use of BZDs in the United States caused public health and safety concern among state and federal legislators and various specialty organizations.
- To help ensure proper use of BZDs, nurses must recognize the potential for dependence and misuse, the medications’ mechanisms of action, the complications of long-term use, and the effects of discontinuation.
Providers prescribe benzodiazepines (BZDs)—also known as anxiolytics, hypnotics, muscle relaxants, anticonvulsants, and amnestic medications—to manage several symptoms and conditions, including anxiety, insomnia, alcohol withdrawal, sedation, muscle spasms, agitation, and seizures. When prescribed for short-term use, they’re valuable to patient management.
This group of medications originated with chlordiazepoxide and diazepam in the late 1950s. Long-term use of diazepam demonstrated significant dependence, tolerance, social dislocation, and difficult withdrawal. In 1975, clonazepam and lorazepam were released to the U.S. market and branded as anxiolytics. The lower doses of these medications, compared with diazepam, masked their initial potency. For example, 1 mg of clonazepam or lorazepam is equivalent to 20 mg of diazepam. The three most potent BZDs—alprazolam, clonazepam, and lorazepam—are 10 to 20 times more potent than diazepam. (See BZD history.)
Cannabinoid hyperemesis syndrome explained
Doctor shopping and prescription substance use disorders: A nursing response
Awash in “bath salts”: The dangers and skyrocketing use of mephedrone
BZD history
Since the 1950s, benzodiazepines (BZDs) have played a role in managing anxiety, insomnia, alcohol withdrawal, sedation, muscle spasms, agitation, and seizures.
1955: Swiss drug company Hoffman-LaRoche synthesized the first BZD (chlordiazepoxide), which was released in the United States in 1960 under the brand name Librium.
1958: Hoffman-LaRoche patented diazepam (Valium).
1970s: Diazepam was found to produce dependence and withdrawal concerns.
1975: La-Roche branded BZDs as anxiolytics and produced clonazepam (Klonopin); Wyeth released lorazepam (Ativan).
1979: Massachusetts Senator Ted Kennedy led a Senate hearing into the dangers of BZDs. No legislation was passed.
1980s: Most industrialized countries strictly regulated nonemergency BZD use. In the United States, BZD prescriptions increased.
1990: The American Psychiatric Association launched a task force to investigate the increase in BZD prescriptions. It found that 80% of patients experienced withdrawal.
2008: More than 1 in 20 Americans received a BZD prescription.
2010: The Veterans Administration recommended against using BZDs to treat post-traumatic stress disorder.
2016: State and federal attention turned toward addiction and death prevalence associated with BZD use. The Food and Drug Administration (FDA) issued a black box warning for combined BZD and opioid use.
2020: The FDA updated its BZD warning to include the risk of abuse, addiction, physical dependence, and withdrawal reactions.
Source: Benzodiazepine Information Coalition, 2022
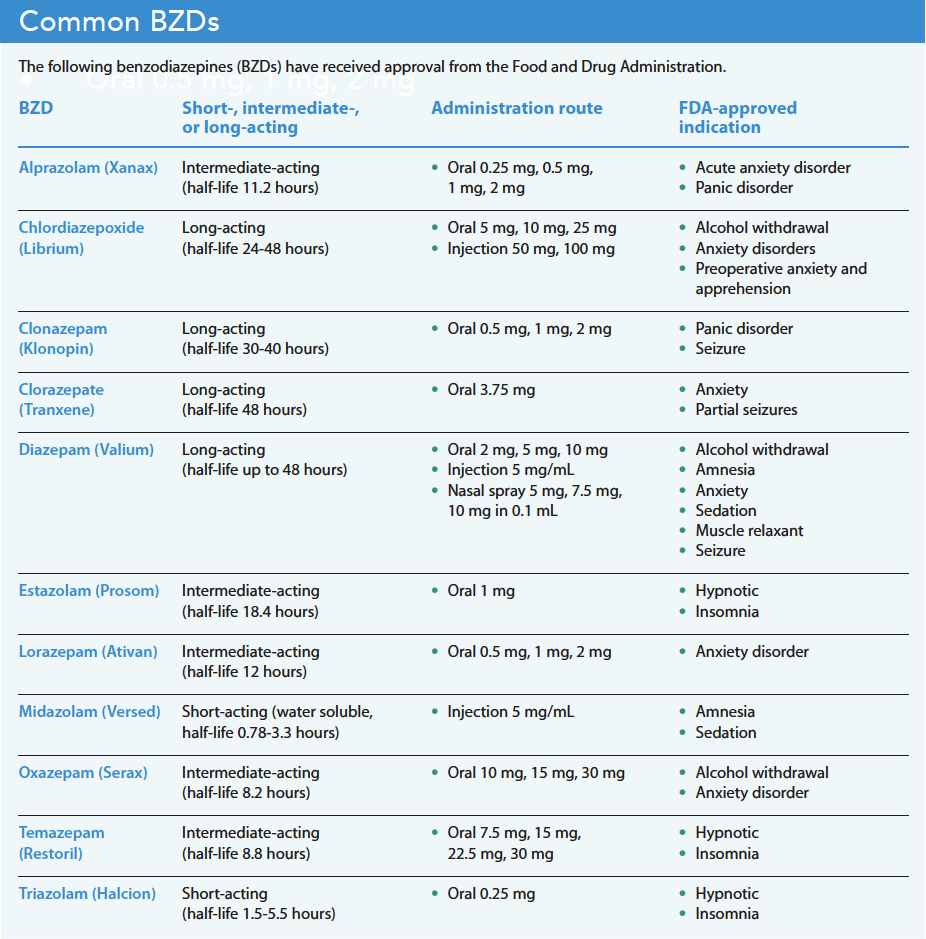
The increased use of BZDs caused public health and safety concerns among state and federal legislators, the American Psychiatric Association, the Veterans Health Administration, and the Food and Drug Administration (FDA). BZD prescriptions in the United States increased by 320% from 1996 to 2013, and overdose deaths associated with BZDs increased over 500%. In 2016, the FDA issued a black box warning for combined BZD and opioid use. The FDA announced that prescribing opioid medications or other central nervous system depressants together with BZDs may result in serious adverse events, including slowed or difficult breathing and death. In 2020, the FDA updated the BZD black box warning to include risks of abuse, addiction, physical dependence, and withdrawal reactions. Despite decades of identifying the dangers of BZD, the number of prescriptions continues to rise, as have the adverse effects associated them. (See Common BZDs.)
BZD use and misuse
A 2019 study by Maust and colleagues evaluated the use and misuse of BZDs based on a National Survey on Drug Use and Health of adults age 18 years and older who reported taking BZDs. Researchers estimated that 30.6 million U.S. adults use BZDs each year, with 25.3 million using them as prescribed and 5.3 million misuing them.
Respondents with a history of misuse and abuse of or dependence on prescription opioids or CNS depressants had the highest risk for BZD misuse. Those without a BZD prescription typically obtained the medication from a close friend or relative. Many adults 50 years and older used BZD to aid sleep. The authors concluded that the number of American adults using BZD was higher than previously reported and of this population, 20% accounted for misuse.
Mechanisms of action
The molecular structure of BZDs, similar to barbiturates, binds within the CNS by enhancing the activity of gamma-aminobutyric acid (GABA), which acts as the main inhibitory neurotransmitter in the CNS, reduces neuronal excitability, and maintains muscle tone. This activation produces sedative, hypnotic, anti-anxiety, anticonvulsant, and muscle relaxant effects.
Complications of long-term use
Evidence suggests that BZDs should be prescribed at the lowest dose and for the shortest amount of time for severe anxiety or insomnia only; they’re not recommended for mild anxiety. Providers should limit their use to 2 to 4 weeks because of the risk of tolerance and dependency. Longer use frequently leads to dose escalation, which can occur after 3 to 4 weeks.
Physical dependence, tolerance, and withdrawal risk can occur with long-term use, which differs from addiction. The American Society of Addiction Medicine defines addiction as a complex chronic disease that involves brain physiology, genetics, the environment, and individual life experience. Those with addiction use substances despite the harm they cause.
Studies of functional brain changes resulting from long-term use and withdrawal of BZDs note a down-regulation of GABA neuroreceptors in the CNS and a reduction and desensitization of the receptor sites from continual BZD binding. Frequently, prolonged BZD exacerbates anxiety, insomnia, and agitation.
Almost all BZD risk factors are associated with long-term use. However, patients who are physically dependent on BZD (beyond 2 to 4 weeks) can’t tolerate abrupt cessation, which may trigger a potentially dangerous withdrawal syndrome. According to the American Psychiatric Association’s Benzodiazepine Task Force on Benzodiazepine Dependence, Toxicity and Abuse, 40% to 80% of patients who discontinue a BZD experience withdrawal symptoms, which can begin within 24 hours to several days. Patients with a psychological dependence may require further management, such as counseling, therapy, or nonaddictive anti-anxiety medications (for example, buspirone or an antidepressant) (See Effects of long-term use.)
Long-term use discontinuation
Discontinuing long-term use of BZD can improve cognitive and psychomotor functioning. Patients ready to discontinue BZDs require education and support, and a slow reduction in dosing allows patients to readjust to life events. Providers should individualize the tapering rate, which can occur over weeks, months, or years. Many patients benefit from switching from a short-acting to a long-acting BZD, such as diazepam. Some patients may require coping strategies to help them live without the medication.
Support patient safety
Nurses should understand the risk of BZD dependence and tolerances; keep in mind the differences among short-acting and long-acting BZDs when considering tolerance, dependence, and withdrawal; and recognize the FDA’s black box warning for combined CNS depressant and BZD use and at-risk patient populations. Nurses can help ensure best practices that support patient safety and health.
Kim Kuebler is the director and founder of Multiple Chronic Conditions Resource Center in Pleasant Lake, Michigan.
American Nurse Journal. 2023; 18(5). Doi: 10.51256/ANJ052323
References:
Benzodiazepine Information Coalition. A brief history of benzodiazepines. benzoinfo.com/a-brief-history-of-benzodiazepines/
Community Behavioral Health. Clinical guidelines for the prescribing and monitoring of benzodiazepines and related medications. August 4, 2020. Division of Behavioral Health and Intellectual disability Services. cbhphilly.org/wp-content/uploads/2020/08/2020-08-06__clinical_guidelines_prescribing_monitoring_benzodiazepines.pdf
Food and Drug Administration. Benzodiazepine drug information. September 23. 2020. fda.gov/drugs/information-drug-class/benzodiazepine-drug-information
Food and Drug Administration. FDA drug safety communication: FDA warns about serious risks and death when combining opioid pain or cough medicines with benzodiazepines; requires its strongest warning. September 20, 2017. fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-warns-about-serious-risks-and-death-when-combining-opioid-pain-or
Food and Drug Administration. FDA requiring boxed warning updated to improve safe use of benzodiazepine drug class. January 14, 2022.
fda.gov/drugs/fda-drug-safety-podcasts/fda-requiring-boxed-warning-updated-improve-safe-use-benzodiazepine-drug-class
Hirschtritt ME, Olfson M, Kroenke K. Balancing the risks and benefits of benzodiazepines. JAMA. 2021;325(4):347-8. doi:10.1001/jama.2020.22106
Kennedy KM, O’Riordan J. Prescribing benzodiazepines in general practice. Br J of Gen Pract. 2019;69(680):152-3. doi:10.3399/bjgp19X701753
Maust DT, Lin LA, Blow FC. Benzodiazepine use and misuse among adults in the United States. Psychiatr Serv. 2019;70(2):97-106. doi:10.1176/appi.ps.201800321
Ogbru A. Benzodiazepine drug class. RxList. December 29, 2022. rxlist.com/benzodiazepines/drug-class.htm
Royal Australian College of General Practitioners. Evidence-based guidance for benzodiazepines. March 3, 2020. https://www.racgp.org.au/clinical-resources/clinical-guidelines/key-racgp-guidelines/view-all-racgp-guidelines/drugs-of-dependence/part-b/evidence-based-guidance-for-benzodiazepines
Key words: benzodiazepines, medication misuse, withdrawal, medication discontinuation