Expand your knowledge for safe, quality care.
Takeaways:
- Acute kidney injury (AKI) in combination with other disease processes, such as COVID-19, significantly impacts patient mortality in the ICU.
- Continuous renal replacement therapy (CRRT) is a treatment commonly used in the ICU to provide kidney support for critically ill patients who are hemodynamically unstable.
- Key aspects of care for patient receiving CRRT can help providing high-quality, safe, effective care.
Critically ill patients can develop acute kidney injury (AKI) as a result of myriad conditions, including COVID-19, sepsis, and heart failure. According to Katulka and colleagues, when these patients require renal replacement therapy (RRT), their mortality rate increases to nearly 60%. Comorbidities such as diabetes, chronic kidney disease, and hypertension are associated with worse outcomes. Despite these dismal statistics, in many cases, RRT can support patients until AKI resolves.
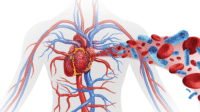
RRT removes fluids and waste (solutes) and exchanges electrolytes, proteins, glucose, and other solutes (collectively referred to as ultrafiltrate) across a permeable membrane by using an extracorporeal blood circuit. Three types of RRT are used in acute care: intermittent hemodialysis (IHD), continuous renal replacement therapy (CRRT), and prolonged intermittent renal replacement therapy (also called slow-low-efficiency daily dialysis [SLEDD]), a combination of IHD and CRRT.
Acute kidney injury: A nursing challenge
Acute kidney injury: Causes, phases, and early detection
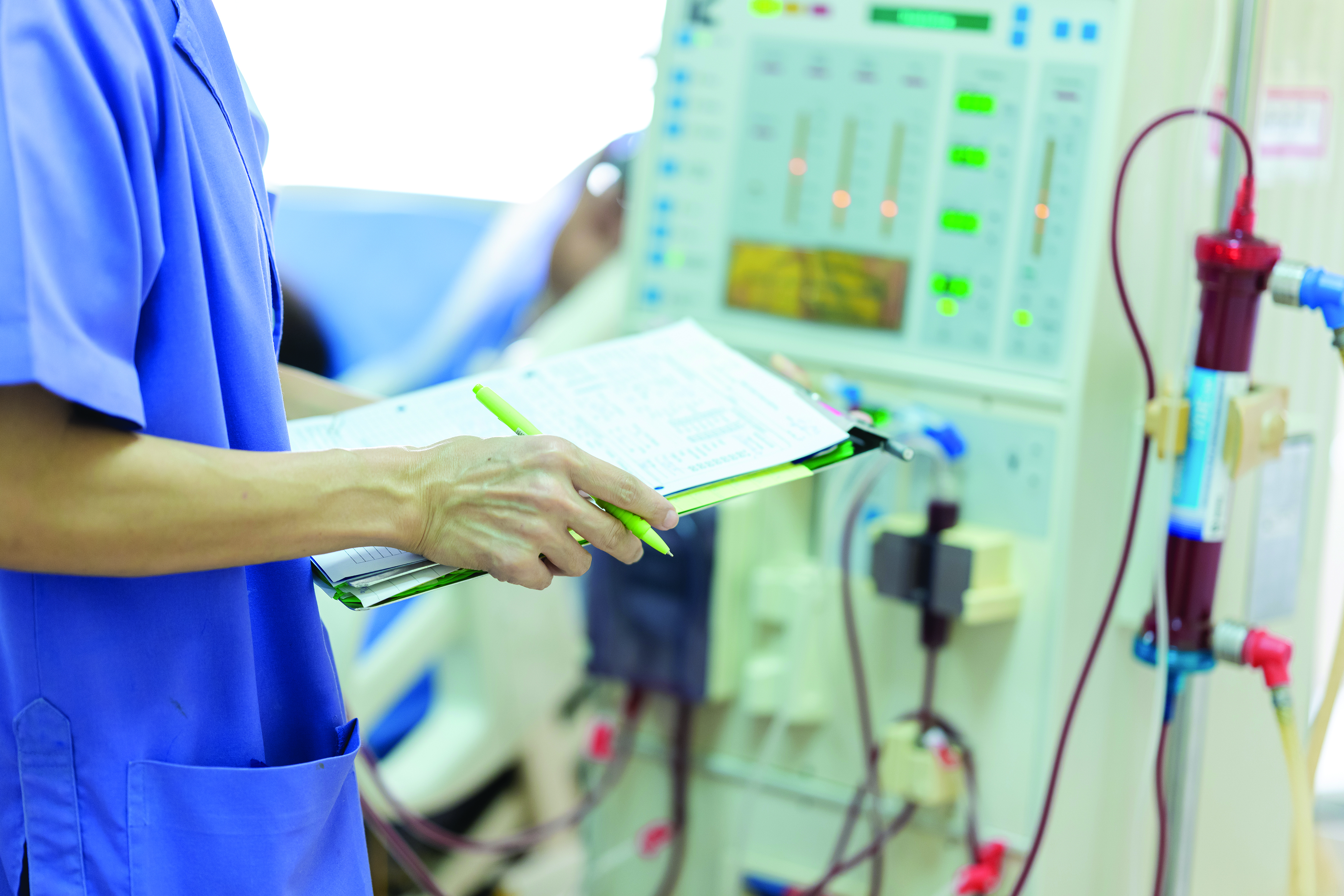
Continuous renal replacement therapy: Dialysis for critically ill patients
IHD quickly removes waste and fluid over a span of 3 to 5 hours. Dialysis nurses can provide this therapy inside and outside the intensive care unit (ICU). SLEDD frequently is provided in the ICU as a bridge therapy when a patient is hemodynamically stable enough for an 8- to 16-hour treatment but too unstable for a 3- to 5-hour treatment.
CRRT is provided 24 hours a day. It effectively removes fluids and wastes in many critical situations where large volumes of blood flow and fluid shifts using IHD aren’t possible because the patient is hemodynamically unstable. Some of the most common diagnoses that benefit from CRRT are progressive acute kidney injury (caused by conditions such as rhabdomyolysis and acute brain injury), electrolyte dysfunction (hyperkalemia, hyponatremia), metabolic acidosis (as a result of conditions such as sepsis and COVID-19), and volume overload (which occurs in situations such as heart failure exacerbation). CRRT has become the treatment choice for patients who have COVID-19 and AKI as well as other patients in the ICU who might benefit from RRT. (See How CRRT works.)
When renal injury or failure occurs, frequently the provider consults with the nephrologist to manage renal perfusion medications and decide the best form of RRT. Using quality indicators and recommendations from experts in nephrology, hospitals can create policies and procedures based on best practices to help with initiating and managing RRT, including CRRT, the focus of this article.
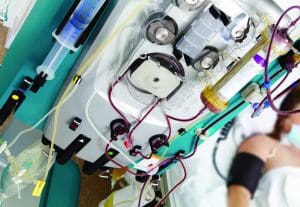

How CRRT works
Continuous renal replacement therapy (CRRT) is a 24-hour blood filtration process. Via hemodialysis access, it slowly purifies the patient’s blood using a cartridge and special filter that removes fluids, toxins, and other substances. To achieve filtration, CRRT uses the four principles of waste and fluid removal:
- convection
- diffusion
- hemofiltration
- ultrafiltration
Intensive care unit and dialysis nurses are responsible for setting up the CRRT machine per manufacturer guidelines.
Initiating and managing CRRT
A collaborative care approach (including the critical care provider, physician’s assistant, advanced practice RN, ICU nurse, nephrology nurse, nephrologist, pharmacist, dietitian, case manager, chaplain services, and palliative care team) can improve CRRT outcomes. Outside of the ICU, nurses can alert providers of compromised kidney function and help identify AKI.
Developing a care plan, including setting goals, begins with early identification of the need for CRRT. Evaluation of the plan, with revisions as needed, continues through treatment discontinuation or transition to SLEDD or IHD. Daily interdisciplinary ICU patient rounds promote early recognition of renal dysfunction and potential risks and benefits of CRRT therapy for each patient. (See Common treatment delays.)
Managing CRRT includes staff education, patient monitoring, medications, nutrition, and prevention of treatment interruptions.
Staff education
Initial and ongoing healthcare staff education is central to successful uninterrupted CRRT. Typically, education is both didactic (for example, reasons for use, treatment modalities, patient scenarios, documentation) and simulated (for example, competency checks, machine setup, troubleshooting). Also beneficial are subject-matter experts (clinical nurse specialists, educators, advanced bedside nurses) who serve as resources for ICU nurses. Intentional rounds, just-in-time discussions (at the bedside) about patients on CRRT, and periodic competency checks also can help identify potential gaps and areas for improvement.
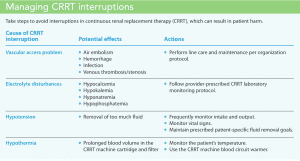
Common treatment delays
Factors that may delay continuous renal replacement therapy include
- acute priority conditions (for example, active bleeding, arrhythmias, active seizures)
- anticoagulation requirements
- family decision-making about treatment risks and benefits
- lack of available equipment
- lack of qualified staff to care for the patient
- severe acidosis
- severe hypotension
- vascular access challenges
Patient monitoring
Most patients who require CRRT are considered high acuity for at least the first 24 to 48 hours. In some organizations, CRRT management consists of a collaboration between the critical care nurse and the dialysis nurse, whereas in others, the ICU nurse has complete responsibility.
Electrolyte balance and fluid and waste removal must be tailored to each patient’s needs. Throughout treatment, nurses should monitor patients closely, watching for signs of electrolyte disturbances, vital sign changes, and urine output improvement.
Medications and nutrition
Vasopressors, sedatives, antibiotics, and nutrition can pose challenges when a patient is receiving CRRT. Medication dosages and nutrition should be guided by factors such as the patient’s renal clearance, residual kidney function, and protein binding. Prompt communication about interruptions and treatment discontinuation will allow the pharmacy and dietary teams to adjust medications and nutrition accordingly.
Preventing treatment interruptions
Reducing treatment interruptions requires proper care of the vascular access catheter and circuit clotting prevention (See Managing CRRT interruptions.)

Vascular access. Well-functioning vascular access is critical to prevent CRRT interruptions. The two most common types of CRRT central venous catheter access used in the acute care setting are temporary hemodialysis catheters and tunneled, cuffed hemodialysis catheters. Temporary hemodialysis catheters can be placed at the bedside by a trained provider or in the operating room. Tunneled, cuffed hemodialysis catheters are placed in the operating room.
The KDIGO (Kidney Disease: Improving Global Outcomes) clinical practice guideline for acute kidney injury recommends placing hemodialysis catheters using ultrasound guidance to decrease the risks (multiple attempts, hematoma formation, pneumothorax, and hemothorax) that accompany catheter placement. Large-bore hemodialysis catheters (13–14 Fr) placed in the internal jugular, femoral, or subclavian vein, are the most effective. The right internal jugular vein is the preferred site because of its proximity to the right atrium. Catheter length and appropriate tip placement, determined by the inserting provider, also are important for adequate flow. Suggested lengths are 12 to 15 cm in the right internal jugular vein, 15 to 20 cm in left internal jugular vein, and 19 to 24 cm in the femoral vein.
The ICU nurse can help identify potential problems related to vascular access . Effective prevention and early detection techniques include pulsatile flushing of the hemodialysis catheter to prevent biofilm buildup, assessing flow before CRRT initiation, and monitoring the CRRT machine pressure trends during treatment.
If CRRT will continue for more than 3 weeks, the patient should be evaluated for permanent hemodialysis catheter placement. Typically, arteriovenous fistulas and grafts aren’t used to deliver CRRT because of the risks of needle dislodgment, hematoma formation, and access damage. These risks increase with continuous flow through the access over an extended period.
Circuit clotting. Blood flow through the CRRT circuit can affect the potential for filter clotting. Depending on the CRRT machine, manufacturer flow recommendations range from 150 to 300 mL/min. Current CRRT machines offer modalities that allow for blood dilution as it passes through the filter. This process uses dialysate/replacement fluid to assist with lowering the risk of filter clotting. It also can be beneficial to patients for whom anticoagulation is contraindicated but who have been identified as at high risk for circuit clotting.
Using anticoagulation agents such as heparin or regional citrate to maintain filter patency varies widely among hospitals. Tandukar and Palevsky cite studies that estimate 30% to 60% of hospitals don’t use anticoagulation with CRRT, often because of the risk of bleeding in patients who have coagulopathy or thrombocytopenia. When anticoagulants are used, the goal is to infuse the lowest dose possible to minimize the systemic effect and achieve sustained filter patency. Keep in mind that each anticoagulation agent has its own risks (for example, bleeding and hypocalcemia with calcium citrate) that require monitoring.
For patients who can’t tolerate anticoagulation, intermittent saline flushes through the CRRT circuit can be used to wash away fibrin strands that might stick to the filter membrane and cause clogging.
Discontinuing treatment
Considerations for CRRT discontinuation include hemodynamic and metabolic status improvements. However, even with these assessment measures in place, no clear consensus exists on the best timing for discontinuation. Current practice uses clinical improvement as a guide. The KDIGO guideline recommends discontinuing therapy when the patient’s kidney function meets their needs or if the care goals no longer can be met. Some patients may be left with inadequate kidney function and require IHD, which can be performed by a dialysis nurse outside the ICU.
Knowledge improves care
Although CRRT is performed only in an ICU, acute care nurses outside the ICU also play an integral part in the care of the acute care patient before and after therapy. When all nurses expand their knowledge of the effects that many disease processes have on kidney function and available treatments, they can help provide safe, high-quality care.
References
Bonnassieux M, Duclos A, Schneider AG, et al. Renal replacement therapy modality in the ICU and renal recovery at hospital discharge. Crit Care Med. 2018;46(2):e102-10. doi:10.1097/ccm.0000000000002796
Cepeda CD, Mathur P, Mehta RL. Continuous renal replacement therapies for acute kidney injury. In: Nissenson AR, Fine RE, eds. Handbook of Dialysis Therapy. 5th edition. New York City, NY: Elsevier; 2016; 356-79.
Counts CS. Core Curriculum for Nephrology Nursing. 6th ed. Pitman, NJ: American Nephrology Nurses’ Association; 2015.
Dirkes S, Wonnacott R. Continuous renal replacement therapy and anticoagulation: What are the options? Crit Care Nurs. 2016;36(2):34-41. doi:10.4037/ccn2016623
Katulka RJ, Al Saadon A, Sebastianski M, et al. Determining the optimal time for liberation from renal replacement therapy in critically ill patients: A systematic review and meta-analysis (DOnE RRT). Crit Care. 2020;24(1):50. doi: 10.1186/s13054-020-2751-8
Kidney Disease: Improving Outcomes. KDIGO Clinical Practice Guideline for Acute Kidney Injury. 2012. kdigo.org/wp-content/uploads/2016/10/KDIGO-2012-AKI-Guideline-English.pdf.
Macedo E, Bihorac A, Siew ED, et al. Quality of care after AKI development in the hospital: Consensus from the 22nd Acute Disease Quality Initiative (ADQI) conference. Eur J Intern Med. 2020;80:45-33. doi:10.1016/j.ejim.2020.04.056
Macedo E, Mehta RL. Continuous dialysis therapies: Core curriculum 2016. Am J Kidney Dis. 2016;68(4):645-57. doi:10.1053/j.ajkd.2016.03.427
Nadim MK, Forni LG, Ravindra LM, et al. COVID-19-associated acute kidney injury: Consensus report of the 25th Acute Disease Quality Initiative (ADQI) Workgroup. Nat Rev Nephrol. 2020;16(12):747-64.
National Kidney Foundation. Ultrafiltration. June 21, 2019. kidney.org/atoz/content/ultrafiltration.
Rewa OG, Bagshaw SM. Beginning and ending continuous renal replacement therapy in the intensive care unit. In: Ronco C, Bellomo R, Kellum JA, Ricci Z, eds. Critical Care Nephrology. 3rd ed. New York City, NY: Elsevier; 2019; 993-9.
Tandukar S, Palevsky PM. Continuous renal replacement therapy: Who, when, why, and how. Chest. 2019;155(3):626-38. doi:10.1016/j.chest.2018.09.004
Clarissa Welbaum is a clinical nurse specialist at AdventHealth Orlando in Orlando, Florida.