Tailor your assessment, prevention, and treatment to individual patient needs and risks.
Takeaways:
- Hypoglycemia places people with diabetes at risk for injury and prevents them from achieving their diabetes management goals.
- Informed RNs can improve patient outcomes by tailoring hypoglycemia assessment, prevention, and treatment to individual needs and potential risks.
- Hypoglycemia prevention builds patients’ confidence in their diabetes management.
Hypoglycemia is generally defined in adults and children with diabetes as a blood glucose (BG) ≤ 70 mg/dL with or without hypoglycemia symptoms. A more comprehensive definition is any drop in BG that exposes a person to harm.
RNs often are the first responders to hypoglycemia, both on the job and in the community. Hypoglycemia is a risk even with effective management and treatment of type 1, type 2, and gestational diabetes. In addition to uncomfortable symptoms, hypoglycemia places people at risk for injury and contributes to an inability to achieve diabetes BG goals. Informed RNs improve patient outcomes by tailoring hypoglycemia assessment, prevention, and treatment to individual needs and potential risks.
Uncontrolled BG
Normal BG regulation occurs when peripheral receptors in the hepatic portal vein and central receptors in the hindbrain and hypothalamus respond to changes in BG and release counter regulatory hormones (glucagon) and neurotransmitters (serotonin, norepinephrine, epinephrine). Together, these hormones and neurotransmitters produce the physical symptoms of hypoglycemia, decrease glucose uptake from peripheral tissues, and activate hepatic glucose production.
Some people with frequent hypoglycemia develop hypoglycemia-associated autonomic failure (HAAF), resulting in hypoglycemia unawareness, defined as not having any physical symptoms preceding the onset of hypoglycemia. When patients don’t have early signs to act upon, hypoglycemia unawareness can lead to severe hypoglycemia (BG < 50 mg/dL) that requires assistance from someone else. Many people can restore hypoglycemia awareness by maintaining a BG above 100 mg/dL for several weeks.
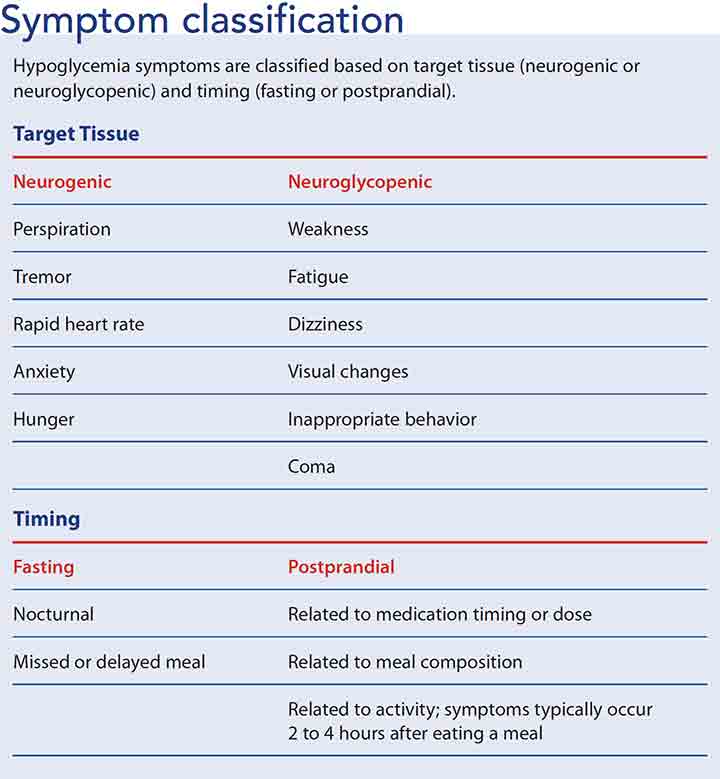
Symptom classification
Accurate knowledge and documentation of hypoglycemia episodes assists with BG pattern identification and acquisition of diabetes supplies. Insurance companies usually require evidence of hypoglycemia severity and frequency to support the need for an insulin pump, continuous glucose monitor (CGM), or increased test strip allowance. Symptoms are classified and documented by target tissue (neurogenic or neuroglycopenic) and timing (fasting or postprandial). BG results combined with these symptoms provide additional evidence of hypoglycemia patterns. (See Symptom classification.)

Target tissue
Neurogenic (sympathetic) symptoms of hypoglycemia include perspiration, tremor, rapid heartbeat, anxiety, and hunger. Neuroglycopenic symptoms include weakness, tiredness, or dizziness; inappropriate behavior; inability to concentrate; confusion; blurred vision; and, in extreme cases, coma and death. Others may mistake the person with neuroglycopenia as being under the influence of alcohol or drugs. Advise patients with diabetes to wear medical alert information to decrease the likelihood of mistreatment.
Timing
Fasting hypoglycemia appears during sleep or after a prolonged period of not eating. Postprandial hypoglycemia typically occurs 2 to 4 hours after eating a meal. Assess and document these symptoms and time periods to identify BG patterns and guide hypoglycemia prevention and treatment strategies.
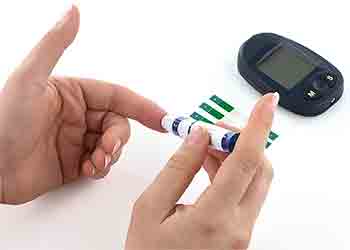
Patient-centered assessment
A patient-centered assessment will aid in individualizing prevention and treatment. Ask patients about their hypoglycemia history and risk factors and how they assess BG.
Hypoglycemia history
Discuss hypoglycemia history with all patients who have type 1, type 2, or gestational diabetes regardless of their HbA1c levels. Review the patient’s medication history; commonly prescribed cardiac medications such as beta blockers and warfarin increase the risk for hypoglycemia unawareness. Your goal is to identify common situations that increase hypoglycemia risk, such as exercise, alcohol use, and meals that are poorly timed with medication type and/or dosage. (See Hypoglycemia history.)

BG assessment
Patients can assess their BG with a glucometer or a CGM that also requires periodic glucometer checks. These tools have improved patients’ ability to self-manage their diabetes.
Glucometers measure capillary BG via blood obtained with a fingerstick. They provide a single data point, so patients can only respond to the information obtained at that time.

Understanding the limitations of glucometers and CGMs will help you facilitate accurate hypoglycemia assessment.
Glucometer malfunction is relatively rare; erroneous results usually are related to blood sampling technique or test strip exposure to heat or moisture. In addition, glucometers perform with up to a 15% margin of error. At the low end of the BG scale, this makes a clinical difference in treatment decisions.
CGM reliability depends on twice-daily calibrations with capillary BG checks. Due to variable measurement between capillary and venous blood samples, many hospital protocols require BG confirmation with a venous sample at the time of hypoglycemia treatment.
Despite these caveats, BG monitoring remains a cornerstone of hypoglycemia prevention and diabetes self-management.
Risk identification
Risk for hypoglycemia depends on patient behaviors—particularly as they relate to timing of meals and medications—age, and type of diabetes medication.
Patient-behavior challenges to hypoglycemia prevention include erratic schedules, disordered eating, and exercise without medication and meal adjustments. A common cause of hypoglycemia is an imbalance between a medication’s onset of action and its administration related to a meal’s timing or composition. In addition, the timing and intensity of physical activity contributes to hypoglycemia.
Age-related physical changes place some people at greater risk for hypoglycemia. For example, growth hormone fluctuations, neurotransmitter aging, gastroparesis, and decreased kidney and liver function all impact glucose regulation.
Most diabetes medications carry some risk for hypoglycemia; however, some have a higher risk than others due to their mechanism of action or fixed dose ratio. For example, people are more likely to experience hypoglycemia if they use sulfonylureas (glyburide, glipizide), meglitinides (repaglinide), or premixed insulins (70/30 NPH/R or 70/30 NPH/lispro). Rapid-acting and short-acting insulin contribute to hypoglycemia risk more than long-acting insulin because of their different pharmacodynamics. Hypoglycemia risk frequently coincides with the peak effect of insulin. The onset, peak effect, and duration of rapid-acting insulin is < 15 minutes, 30 to 90 minutes, and 3 to 5 hours, respectively. Short-acting insulin has an onset of 0.5 to 1 hour, peak effect of 2 to 4 hours, and a duration of 4 to 8 hours. In contrast, long-acting insulin begins working in 1 to 2 hours, has no peak effect, but has a duration of up 20 to 24 hours.
Prevention
Hypoglycemia prevention is preferred to hypoglycemia treatment for achieving diabetes management goals. Hypoglycemia indirectly contributes to HbA1c elevation because of the hyperglycemia that often follows overtreatment with carbohydrate. In addition, fear of hypoglycemia can prevent patient adherence to medication dosages or even prevent prescribing glucose-lowering medications. Both over treatment of hypoglycemia and aversion to diabetes medications may worsen diabetes control.
Design prevention strategies based on each person’s unique experience with hypoglycemia. For example, people with diabetes can prevent hypoglycemia by decreasing their diabetes medication dose before physical activity or by eating more in anticipation of activity. Insulin pump users can decrease basal insulin before, during, or after exercise. Likewise, they can decrease the rapid-acting insulin dose at the meal that precedes activity.
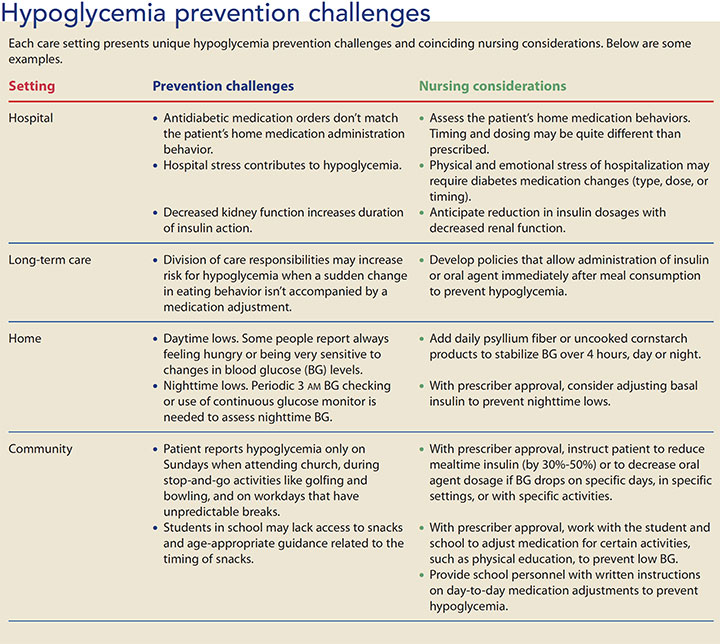
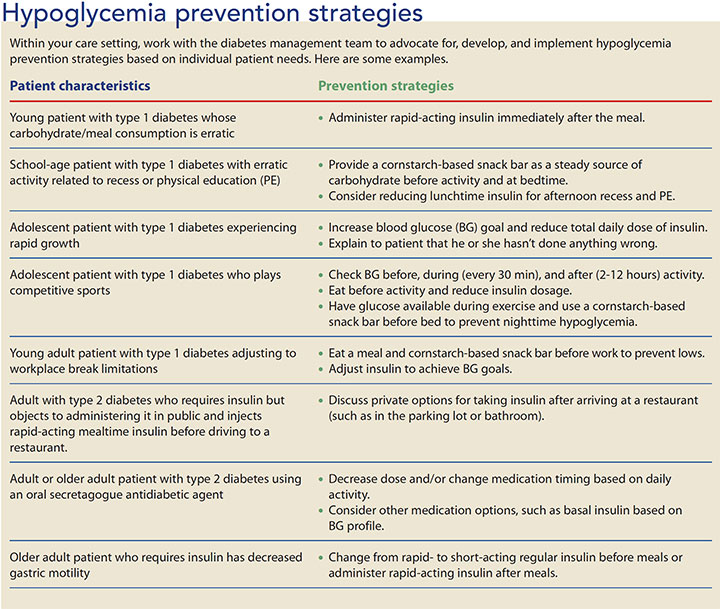
Each patient-care setting presents unique challenges for hypoglycemia prevention and treatment. (See Hypoglycemia prevention challenges.) Advocate for implementation of prevention strategies in your care setting. You and the diabetes management team can create practical solutions. (See Hypoglycemia prevention strategies.)


Treatment
Adopt evidence-based hypoglycemia treatment protocols for use in acute-care, long-term care, home, and school settings. Consider hypoglycemia a critical incident and evaluate its frequency in your care setting and the treatment protocol’s effectiveness. Base treatment protocols on the patient’s condition (conscious, I.V. access, NPO status) and BG level. The patient’s condition will guide whether a meal, oral glucose, I.V. dextrose, or glucagon is appropriate treatment. Follow protocol instructions for ongoing BG monitoring and evaluation of treatment effectiveness (for example, see this protocol from the American Association of Clinical Endocrinologists: inpatient.aace.com/protocols-and-order-sets).
A meal
For a balance of short- and long-acting fuel that maintains body functions, patients with mild hypoglycemia that occurs at mealtimes should eat a meal that contains complex carbohydrates, protein, fats, fiber, and simple sugars. Instruct patients taking mealtime diabetes medications to follow prescribed dosage adjustments before a meal when BG is < 100 mg/dL. For example, they may decrease their insulin dose 25% to 50%. Omitting insulin completely at mealtime contributes to subsequent hyperglycemia. Provide patients with specific written instructions for common BG scenarios to build their self-management confidence. It’s important not to over-treat hypoglycemia. For instance, consuming more than 15 to 20 g of carbohydrate in response to hypoglycemia causes hyperglycemia.
Oral glucose
Oral glucose elevates BG in about 15 minutes. Instruct patients to use measured hypoglycemia treatments such as instant glucose, chewable glucose tablets, or 4 oz to 6 oz of regular soda. The carbohydrate dose is 15 g to 20 g for adults and weight based for children (generally 200 mg/kg). Using glucose is especially important for patients taking acarbose or miglitol. These starch-blocking medications slow absorption of complex carbohydrates and delay BG elevation. Glucose deprivation in the brain inhibits clear thinking, so instruct patients to set a timer after BG treatment to overcome the natural instinct to overeat. Family and friends often force overeating because of hypoglycemia fears. To help family members understand and trust the treatment protocols, include them in the hypoglycemia treatment plan.
I.V. dextrose
I.V. dextrose, which is available in a prefilled 50-mL syringe, elevates BG immediately. It’s administered as 50% (D50W) by I.V. push to hospitalized patients with I.V. access who are unable to take an oral hypoglycemia treatment. Give half of a syringe or a whole syringe based the patient’s BG level and as directed by the standing orders.
Glucagon
Glucagon elevates BG by stimulating the liver to convert stored glycogen to glucose. In severe hypoglycemia, administer glucagon as a 1-mg intramuscular injection to an unconscious person without I.V. access. Vomiting is a common side effect, so instruct caregivers to place the person on his or her left side after injecting the gluca gon. BG elevation continues for about 1 hour after administration. To maintain normal BG, the patient must eat a meal after regaining consciousness.
Confirm an emergency glucagon kit prescription for all patients with type 1 diabetes, those with a history of severe hypoglycemia, and those at risk for severe hypoglycemia. Teach caregivers how to administer the glucagon injection, and instruct patients and their family to keep the kit easily accessible. Living situations change, so each year identify all friends and family members who need the glucagon information.
Prevention builds confidence
Every RN can contribute to the assessment, prevention, and treatment of hypoglycemia. You can improve outcomes with patient education tailored to each individual’s potential risks. Inform patients that hypoglycemia may contribute to higher HbA1c. Counsel them to wear medical alert identification, identify hypoglycemia symptoms, check their BG, and anticipate problem circumstances. Ask patients to keep a hypoglycemia journal and bring it to medical appointments. Hypoglycemia prevention builds patients’ confidence in their diabetes management.
Dana E. Brackney is an assistant professor at Appalachian State University in Boone, North Carolina.
Selected references
American Diabetes Association. Standards of Medical Care in Diabetes—2018 abridged for primary care providers. Clin Diabetes. 2018;36(1):14-37.
Butler S, Abel KL, Clark L. The unique needs of the preschool and early elementary school-age child with type 1 diabetes. NASN Sch Nurse. 2013;28(5):230-2.
Carey M, Boucai L, Zonszein J. Impact of hypoglycemia in hospitalized patients. Curr Diab Rep. 2013;13(1):107-13.
Desjardins K, Brazeau AS, Strychar I, Rabasa-Lhoret R. Are bedtime nutritional strategies effective in preventing nocturnal hypoglycemia in patients with type 1 diabetes? Diabetes Obes Metab. 2014;16(7):577-87.
Francescato MP, Geat M, Stel G, Cauci S. Accuracy of a portable glucose meter and of a Continuous Glucose Monitoring device used at home by patients with type 1 diabetes. Clin Chim Acta. 2012;413(1-2):312-8.
Jesitus J. Hypoglycemia guidelines: AAP vs PES. Contemp Pediatr. 2016;33(6):22-6.
Ly TT, Maahs DM, Rewers A, Dunger D, Oduwole A, Jones TW; International Society for Pediatric and Adolescent Diabetes. ISPAD Clinical Practice Consensus Guidelines 2014. Assessment and management of hypoglycemia in children and adolescents with dia- betes. Pediatr Diabetes. 2014;15(suppl 20):180-92.
McCrimmon RJ. Update in the CNS response to hypoglycemia. J Clin Endocrinol Metab. 2012;97(1):1-8.
Pivovarov JA, Taplin CE, Riddell MC. Current perspectives on physical activity and exercise for youth with diabetes. Pediatr Diabetes.2015;16(4):242-55.
Polonsky WH, Fisher L, Hessler D, Edelman SV. Identifying the worries and concerns about hypoglycemia in adults with type 2 diabetes. J Diabetes Complications. 2015;29(8): -1171-6.
Salacinski AJ, Alford M, Drevets K, Hart S, Hunt BE. Validity and reliability of a glucometer against industry reference standards. J Diabetes Sci Technol. 2014;8(1):95-9.
Seaquist ER, Anderson J, Childs B, et al. Hypoglycemia and diabetes: A report of a work-group of the American Diabetes Association and the Endocrine Society. Diabetes Care. 2013;36(5):1384-95.
Sircar M, Bhatia A, Munshi M. Review of hypoglycemia in the older adult: Clinical implications and management. Can J Diabetes. 2016;40(1):66-72.
Taborsky GJ Jr, Mundinger TO. Minireview: The role of the autonomic nervous system in mediating the glucagon response to hypoglycemia. Endocrinology. 2012;153(3):1055-62.