Understand the benefits, risks, and contraindications.
- When used in conjunction with lifestyle modifications, pharmacologic treatment can be effective in expediting weight loss in the appropriate patient population.
- Successful weight loss and maintenance comes only with continued reinforcement and adherence to healthy lifestyle habits, including diet and physical activity.
- Nurses play a vital role in educating patients about the importance of healthy lifestyle modifications and management strategies to ensure long-term weight-loss maintenance.
Learning Objectives
- Describe pharmacologic weight loss treatments.
- Discuss who can benefit from weight loss medications.
- Describe patient education and nursing implications that can impact successful weight management.
Nancy E. Thompson, a peer reviewer for this article, is on the speakers’ bureau for Merck. The relevant financial relationships have been mitigated. No relevant financial relationships were identified for any other individuals with the ability to control content of the activity.
Caring for adult patients with obesity in primary care
Obesity: Skin issues and skinfold management
Expiration: 9/1/26
Marcy Taylor*, a 48-year-old woman, visits her nurse practitioner, Bryan Thomas, to discuss weight loss management. Marcy is 62 inches tall, weighs 182 lbs, and has a body mass index (BMI) of 33.3 kg/m2. Over the years, she’s tried several treatment options, including intermittent fasting and various diet programs, with no long-term success. Marcy would like to discuss pharmacologic options to “jump start” her weight loss and is interested in semaglutide (Wegovy®) because it’s injected only once a week.
Marcy has a medical history of hypertension, new onset paroxysmal atrial fibrillation, high cholesterol, and prediabetes. Her current medications include telmisartan for hypertension and rosuvastatin for cholesterol. She’s taken phentermine in the past with successful weight loss and likes this medication, but she had to discontinue it because of the paroxysmal atrial fibrillation. She wants to lose 47 lbs to reach her personal goal of 135 lbs. Marcy doesn’t exercise and works a sedentary job as an office receptionist. Her 24-hour diet recall consists of two cups of coffee and a blueberry muffin for breakfast, a turkey sandwich for lunch with two cans of soda, and fried chicken with mashed potatoes for dinner. Her snacks consist of pretzels and a bedtime bowl of vanilla ice cream.
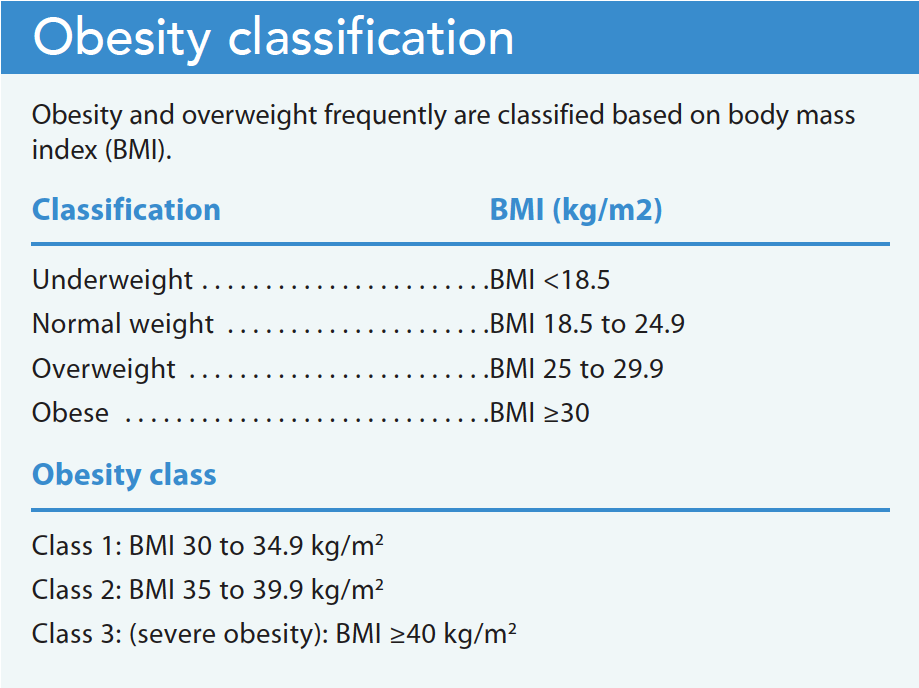

The World Health Organization and the CDC classify individuals with a body mass index (BMI) >25 kg/m2 as overweight and those with a BMI ≥30 kg/m2 as obese. Severe obesity (BMI >40) can reduce life expectancy by 5 to 20 years. Obesity-related comorbid conditions include cardiovascular disease, GI disorders, metabolic syndrome, diabetes, respiratory problems, obstructive sleep apnea, musculoskeletal disorders, depression, and certain cancers. However, only one-third of patients with obesity are formally diagnosed and effectively advised and treated. (See Obesity classification.)
Pharmacotherapy for weight loss
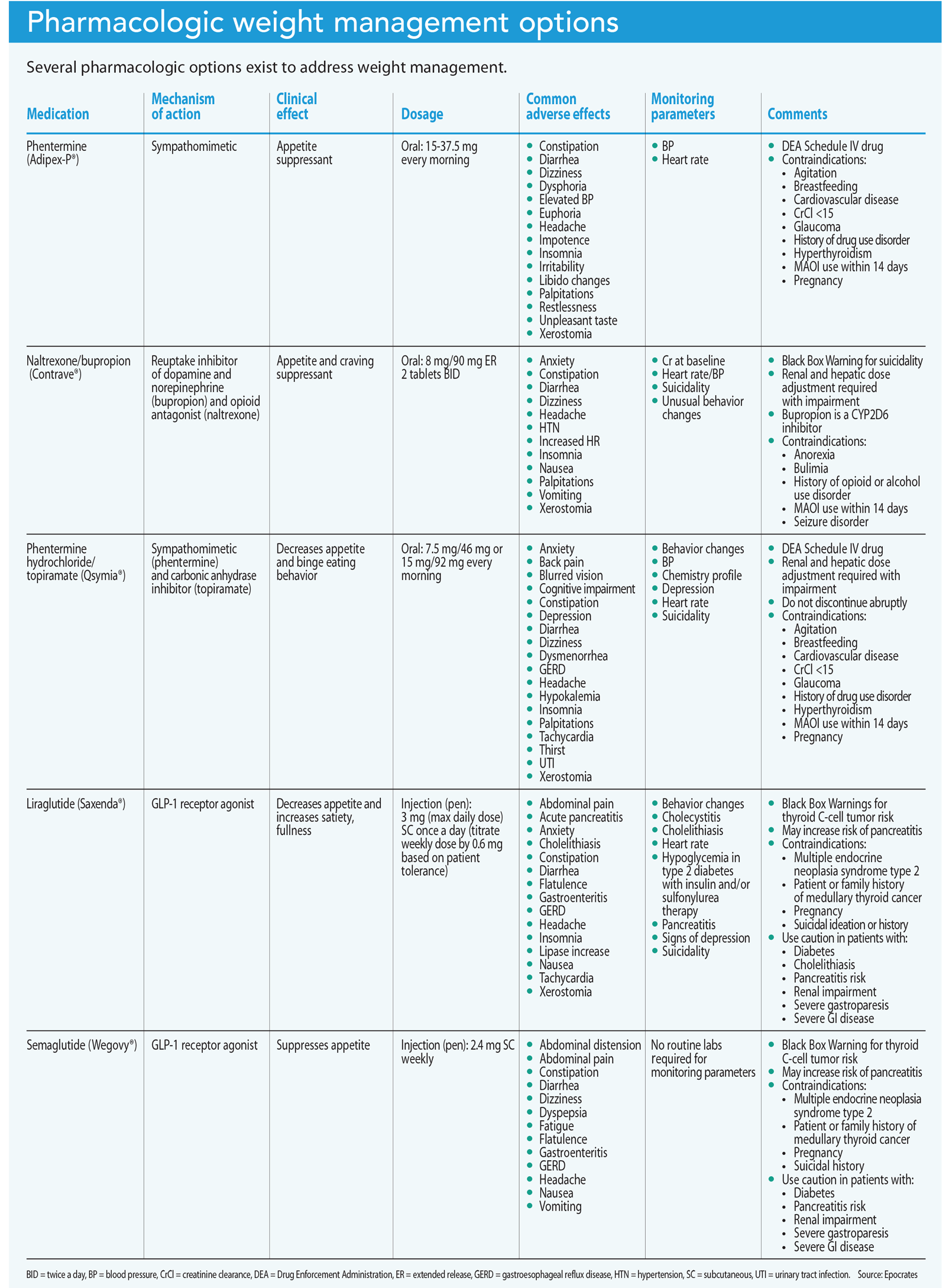
Clinical guidelines support pharmacotherapy as a second-line treatment option in conjunction with diet, exercise, and behavioral modifications for obesity management. The Food and Drug Administration (FDA) has approved specific medications for patients who meet the criteria of overweight or obese and would benefit from weight loss. However, reinforcement of weight loss strategies and healthy lifestyle changes remain imperative for long-term success. A meta-analysis of 29 long-term weight loss studies revealed that more than half of the weight lost was regained within 2 years, and more than 80% was regained by 5 years. Patients must understand that long-term success requires ongoing attention to health behaviors and is unlikely if lifestyle modifications aren’t a priority. (See Pharmacologic weight management options.)
Obesity, type 2 diabetes, and cardiovascular conditions such as hypertension frequently are interrelated, which increases morbidity and mortality. Research has suggested that weight loss can help improve hypertension and type 2 diabetes in some patients. A study by Blumenthal and colleagues demonstrates the benefits and efficacy of lifestyle modifications, including behavioral changes, exercise, and dietary counseling on resistant hypertension. Clinical trials suggest remission in type 2 diabetes, and reduced cardiovascular complications may occur in patients who can maintain long-term weight loss. The Diabetes Remission Clinical Trial (DIRECT) data showed that 86% of patients who lost ≥15 kg could maintain diabetes remission. After 2 years, more than one-third of patients remained in remission.
Oral medications
Short- and long-term oral pharmacologic options exist, and many patients prefer their convenience over injectable medications.
Phentermine
The FDA-approved phentermine (Adipex-P®) is one of the most prescribed medications for short-term obesity treatment. It’s taken for 12 weeks, but has been used longer with some supporting evidence. Successful weight loss can continue with prolonged use after 12 weeks, but increased risks and adverse outcomes must be considered, monitored, and evaluated. In a study by Lewis and colleagues, patients using phentermine continuously for more than 12 months lost 7.4% more weight than the comparison group, with no significant difference in adverse outcomes.
Many patients report liking the flexibility of taking phentermine, which is chemically related to amphetamine and sometimes referred to as an “atypical amphetamine.” It stimulates the central nervous system and can increase heart rate and blood pressure, making patient selection and evaluation essential. Primary side effects—including blood pressure and pulse elevation, palpitations, dizziness, insomnia, and constipation—are related to its sympathomimetic properties.
Phentermine is a Drug Enforcement Administration (DEA) Schedule IV controlled substance with a low potential for abuse and dependence. Misuse can cause addiction, overdose, or death. Prescribing restrictions may differ from state to state. Contraindications include pregnancy, breastfeeding, creatinine clearance (CrCl) <15, heart disease (coronary artery disease, arrhythmias, congestive heart failure, stroke), severe or uncontrolled hypertension, hyperthyroidism, glaucoma, agitation or nervousness, history of drug use disorder, and use of monoamine oxidase (MAO) inhibitors within 14 days. Phentermine may be prescribed alone or in combination with topiramate. The success rates of phentermine can be favorable with adjunctive lifestyle changes and modifications.
Naltrexone/bupropion and phentermine/topiramate
Two common oral medications for the long-term treatment of obesity include naltrexone/bupropion (Contrave®) and phentermine/topiramate (Qsymia®). Both can be continued for up to 12 weeks, but should be discontinued if weight loss is <5%.
Naltrexone/bupropion.
Bupropion inhibits norepinephrine and dopamine reuptake, reduces appetite, and increases energy expenditure via increased dopamine activity and proopiomelanocortin neuronal activation. Naltrexone is an opioid receptor antagonist, which aids weight loss by working to suppress the brain’s eating reward pathways. It can potentially increase growth hormone release, which can lead to increased fat metabolism.
Common side effects of naltrexone/bupropion include nausea, vomiting, constipation, headache, dizziness, insomnia, and dry mouth. In addition, the FDA has imposed a Black Box Warning for this medication and recommends monitoring patients for worsening suicidal ideation or behaviors.
This medication has a long list of contraindications and cautionary prescribing, including MAO inhibitor use within 14 days, seizure disorder, hypertension, and patients under age 25. Careful screening and review of patient history are essential. This medication shouldn’t be prescribed to patients with a history of opioid or alcohol use disorder, psychiatric disorders, or suicide.
Phentermine/topiramate.
Some studies have supported the success of phentermine/topiramate and have shown its superiority to other oral agents. According to a review by Idrees and colleagues, significant weight loss of at least 5% was associated in 75% of those taking this medication over the course of a year. They also showed a 4.1% decrease in weight from baseline compared to 3.6% for those taking phentermine alone after 20 weeks.
Combining topiramate with phentermine inhibits carbonic anhydrase, which promotes taste aversion and reduces caloric intake. Topiramate also is FDA-approved for treating seizures and as migraine prophylaxis. This medication may be a compelling choice for patients with obesity who have a history of migraines.
The FDA requires a risk evaluation and mitigation strategy for phentermine/topiramate to ensure the benefits outweigh the risks and that patients of reproductive age are informed about the dangers of taking this medication during the first trimester. Common adverse effects may include paresthesia, dizziness, dry mouth, dysgeusia, constipation, insomnia, somnolence, cognitive dysfunction, mood disturbances, depression, and anxiety. Contraindications include pregnancy, breastfeeding, hyperthyroidism, glaucoma, and MAO inhibitor use within 14 days.
Gradual discontinuation of this medication is essential; abrupt withdrawal of topiramate may increase the risk of seizures. Phentermine/topiramate is a DEA Schedule IV controlled substance with low-to-moderate abuse potential. State prescribing requirements and restrictions may vary.
Injectable medications
Convenient dosing options have many patients interested in injectable options for weight loss. FDA-approved injectable weight loss medications include the GLP-1 agonists liraglutide (Saxenda®) and semaglutide (Wegovy®). GLP-1 agonists delay gastric emptying, which promotes appetite suppression to reduce hunger and calorie intake. Some prescribers voice concern about the risk of pancreatitis and cholecystitis with these medications, but more recent data suggest that it’s not as prevalent as previously thought; however, patients should be monitored. Contraindications for these GLP-1 antagonists include a personal or family history of medullary thyroid cancer or multiple endocrine neoplasia type, as listed on the Black Box Warning.
Liraglutide
Liraglutide, a daily subcutaneous injection, starts with 0.6 mg daily and increases by 0.6 mg weekly until reaching the target or maximum dose. This titration method helps to mitigate possible GI side effects, such as nausea, vomiting, and abdominal pain.
Semaglutide
In June 2021, the FDA approved semaglutide at the 2.4 mg dose for adults meeting the criteria for overweight or obesity. Under the brand names Ozempic® and Rybelsus®, the FDA approved semaglutide to treat type 2 diabetes, but at a different dosing regimen than that used for weight loss. For weight loss, semaglutide is dosed as a once-weekly subcutaneous injection.
Recent clinical trial data show encouraging results. Over 68 weeks, trial participants lost an average of 15% to 20% of their body weight. Also encouraging, many participants saw risk reduction for diabetes and cardiovascular disease. The most frequently reported side effects include nausea, diarrhea, vomiting, constipation, and abdominal pain. Dehydration may occur, which can increase the risk of acute kidney injury. To prevent these effects, patient education should include the need for increased water intake.
Tirzepatide
Tirzepatide (Mounjaro™), an FDA-approved injectable medication for type 2 diabetes, is under accelerated review for treating obesity. This GLP-1 and glucose-dependent insulinotropic polypeptide agonist has shown impressive results in clinical trials, with participants reaping a weight loss of over 22.5% (surpassing semaglutide) in addition to treating their type 2 diabetes.
Who can benefit from weight loss medications?
After a physical examination and discussion, Marcy and Bryan decide to begin semaglutide treatment with a 0.25 mg subcutaneous injection weekly for 4 weeks, followed by an increase to 0.5 mg weekly for 4 weeks with a follow-up visit at 8 weeks. If Marcy tolerates the medication well, Bryan will increase the dose to 1.7 mg weekly for 4 weeks, then increase it to 2.4 mg weekly. In addition, Marcy will follow a customized management plan that includes lifestyle modifications, diet, and exercise. Bryan provides a referral to a dietician to help with meal planning.
At her 8-week follow-up visit, Marcy describes meeting with the dietician and adopting a healthy diet and meal plan. She’s been diligent in adhering to lifestyle modifications, including exercising 4 days a week. She’s tolerating the current dose of semaglutide well with mild nausea, which has subsided. She’s lost a total of 12 lbs. As a result, Bryan increases the dose of semaglutide to 1.7 mg for 4 weeks followed by an increase to 2.4 mg weekly. A follow-up appointment is scheduled for 8 weeks.
The risks, complications, and medical costs associated with obesity are well studied and documented. For the appropriate patient, pharmacologic options can help achieve successful weight loss and improve overall outcomes. Patients like Marcy, who meet the criteria for obesity and especially with comorbidities, can benefit from weight loss medications.
The best candidates for pharmacologic management are motivated patients who’ve tried to lose weight and are committed to implementing lifestyle changes needed for long-term success. Considerations when prescribing weight-loss medications include patient health history, comorbidities, preferences, cost, and insurance coverage. Many insurers require prior authorization, and some may require documentation of failed medications and therapies. Some insurances don’t cover pharmacologic treatment for weight loss at all, forcing patients to pay out of pocket for the medication, which can prove very expensive. These barriers can limit patient access.
Healthcare professionals can be instrumental advocates, playing a pivotal role in education, health promotion, identifying weight-loss strategies, setting realistic goals, and providing patients with support, resources, and motivation during their weight loss journey. (See Resources.)
Implications for nursing
Nurses serve as advocates, educators, and even health coaches for their patients with obesity. They’re instrumental in providing support and information about the importance of lifestyle modifications, including healthy eating, self-care, coping mechanisms, behavior modification, and physical activity for successful weight loss and maintenance. The weight loss journey can be long and challenging for many patients. Nurses can help them set realistic goals and provide resources. In addition, nurse practitioners, who also serve in the prescriber role, participate in shared-decision making on weight loss medications and other treatment options. Understanding that every patient is unique and requires an individualized management plan, including monitoring and re-evaluation, helps to ensure overall patient safety, outcomes, and success.
References
Several evidence-based practice guidelines and online resources are available to share with patients during their weight loss journey and to help nurses and other healthcare providers learn more.
- American Society for Metabolic and Bariatric Surgery (asmbs.org/patients): The organization’s patient learning center includes videos, answers to frequently asked questions, and recommendations for finding a provider.
- Dietary Guidelines for Americans (dietaryguidelines.gov): This downloadable PDF provides guidance regarding scientific findings related to weight and weight loss, nutrition facts, and recommendations based on age and other demographics.
- National Heart, Lung, and Blood Institute (nhlbi.nih.gov/health/educational/lose_wt/BMI/bmicalc.htm): Patients can use the institute’s body mass index calculator to determine their weight category.
- National Institute of Diabetes and Digestive and Kidney Diseases (niddk.nih.gov/health-information/weight-management): Patients can find information on many topics, including choosing a successful weight loss program, binge eating disorders, and bariatric surgery.
- The Obesity Society (obesity.org): This scientific membership organization focuses on advancing a science-based understanding of the causes, consequences, prevention, and treatment of obesity.
- Prevention (prevention.com/weight-loss/a20468312/best-weight-loss-apps): Baum I, Smith J. 17 best weight loss apps to help you achieve your goals, according to experts.
- USDA Dietary Guidance Interactive Tools (www.nal.usda.gov/human-nutrition-and-food-safety/nutrition-tools-and-curricula): This site provides tools and calculators for managing weight.
*Name is fictitious.
Kimberly Lynch is an adult nurse practitioner at Associated Physicians in Libertyville, Illinois, and assistant FNP program director and associate professor of MSN/FNP programs at Olivet Nazarene University in Bourbonnais, Illinois.
American Nurse Journal. 2023; 18(9). Doi: 10.51256/ANJ092306
References
Adipex-P. Epocrates. epocrates.com/online/drugs/3527/adipex-p
Apovian CM, Aronne LJ, Bessesen DH, et al. Pharmacological management of obesity: An Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2015;100(2):342-62. doi:10.1210/jc.2014-3415
Blumenthal JA, Hinderliter AL, Smith PJ, et al. Effects of lifestyle modification on patients with resistant hypertension: Results of the TRIUMH randomized clinical trial. Circulation. 2021;144(15):1212-26. doi:10.1161/CIRCULATIONAHA.121.055329
Centers for Disease Control and Prevention. Adult obesity facts. cdc.gov/obesity/data/adult.html. May 17, 2022.
Centers for Disease Control and Prevention. Defining adult overweight & obesity. June 3, 2022. cdc.gov/obesity/basics/adult-defining.html
Contrave. Epocrates. https://www.epocrates.com/online/drugs/6957/contrave
Hall KD, Kahan S. Maintenance of lost weight and long-term management of obesity. Med Clin North Am. 2018;102(1):183-97. doi:10.1016/j.mcna.2017.08.012
Idrees Z, Cancarevic I, Huang L. FDA-approved pharmacotherapy for weight loss over the last decade. Cureus. 2022;14(9):e29262. doi:10.7759/cureus.29262
Ko JH, Kim TN. Type 2 diabetes remission with significant weight loss: Definition and evidence-based interventions. J Obes Metab Syndr. 2022;31(2):123-33. doi:10.7570/jomes22001
Kulak-Bejda A, Bejda G, Waszkiewicz N. Safety and efficacy of naltrexone for weight loss in adult patients—A systematic review. Arch Med Sci. 2020;17(4):940-53. doi:10.5114/aoms.2020.96908
LeBlanc ES, Patnode CD, Webber EM, Redmond N, Rushkin M, O’Connor EA. Behavioral and pharmacotherapy weight loss interventions to prevent obesity-related morbidity and mortality in adults: Updated evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2018;320(11):1172-91. doi:10.1001/jama.2018.7777
Lewis KH, Fischer H, Ard J, et al. Safety and effectiveness of longer-term phentermine use: Clinical outcomes from an electronic health record cohort. Obesity 2019;27(4):591-602. doi:10.1002/oby.22430
May M, Schindler C, Engeli S. Modern pharmacological treatment of obese patients. Ther Adv Endocrinol Metab. 2020;11:2042018819897527. doi:10.1177/2042018819897527
Montan PD, Sourlas A, Olivero J, Silverio D, Guzman E, Kosmas CE. Pharmacologic therapy of obesity: Mechanisms of action and cardiometabolic effects. Ann Transl Med. 2019;7(16):393. doi:10.21037/atm.2019.07.27
Mounjaro. mounjaro.com/hcp
Qsymia. Epocrates. epocrates.com/online/drugs/6442/qsymia
Saxenda. Epocrates. epocrates.com/online/drugs/7106/saxenda
U.S. Preventative Services Task Force. Final recommendation statement: Weight loss to prevent obesity-related morbidity and mortality in adults: Behavioral interventions. September 18, 2018. uspreventiveservicestaskforce.org/uspstf/document/RecommendationStatementFinal/obesity-in-adults-interventions#bootstrap-panel–3
Wegovy. Epocrates. epocrates.com/online/drugs/10221/wegovy#black-box-warnings
Wilding JPH, Batterham RL, Calanna S, et al. Once-weekly semaglutide in adults with overweight or obesity. N Engl J M. 2021;384(11):989-1002. doi:10.1056/nejmoa2032183
World Health Organization. Obesity. who.int/health-topics/obesity#tab=tab_1
Key words: Obesity, obesity treatment, weight management, weight-loss