The Institute for Safe Medication Practices (ISMP) made several recommendations related to the safe administration of I.V. medications based on the results of its survey and has published a compilation of best practices. The Infusion Nurses Society (INS) standards also address I.V. push administration.
Here is checklist of recommendations from ISMP and INS to help ensure safe practice.
For Pharmacists
- Dispense I.V. push medications in ready-to-administer prefilled syringes.
- If a prefilled syringe isn’t commercially available, prepare a syringe in the dose specific to the patient. Label the syringe with the patient’s name, drug name, strength, dose, directions for administration (e.g., slow I.V. push over 3 to 5 minutes), and the beyond-use date/time.
- If stability is a problem, dispense a single-use vial with instructions.
- Be sure manufacturers’ prefilled syringe cartridge holders are readily available to clinicians administering I.V. push medications.
- Avoid dilution of I.V. push medications, but if required, do so in the pharmacy (instead of having clinicians dilute them).
- Provide the rate of administering I.V. push medications on the medication administration record.
For nurses
Before administration
- Do not dilute I.V. push medications unless required to (for example, because stability issues prevent the pharmacist from doing so).
- If you have to dilute, follow the specific directions provided by the pharmacy and prepare the medication in an uncluttered location with the needed supplies.
- Do not use saline flush syringes for diluting medications.
- Do not draw medication from a commercially prepared syringe into another syringe for administration.
- Ask the pharmacy to prepare I.V. push medications whenever possible if a manufacturer-prepared prefilled syringe in not available
- If more than one syringe of medication for a single patient needs to be prepared at the bedside, prepare each one separately and immediately administer it before preparing the next one.
- Label all medication syringes that you prepare when not at the patient’s bedside, even if you think you’ll be administering them right away.
Administration
- Use an appropriately sized syringe (for example, 3 mL) for administering I.V. push medications via a central vascular access device. (A 10 mL syringe is required only for confirming patency of the line, not for administering the medication.)
- Use the needleless connector that’s closest to the patient whenever possible for administering medication.
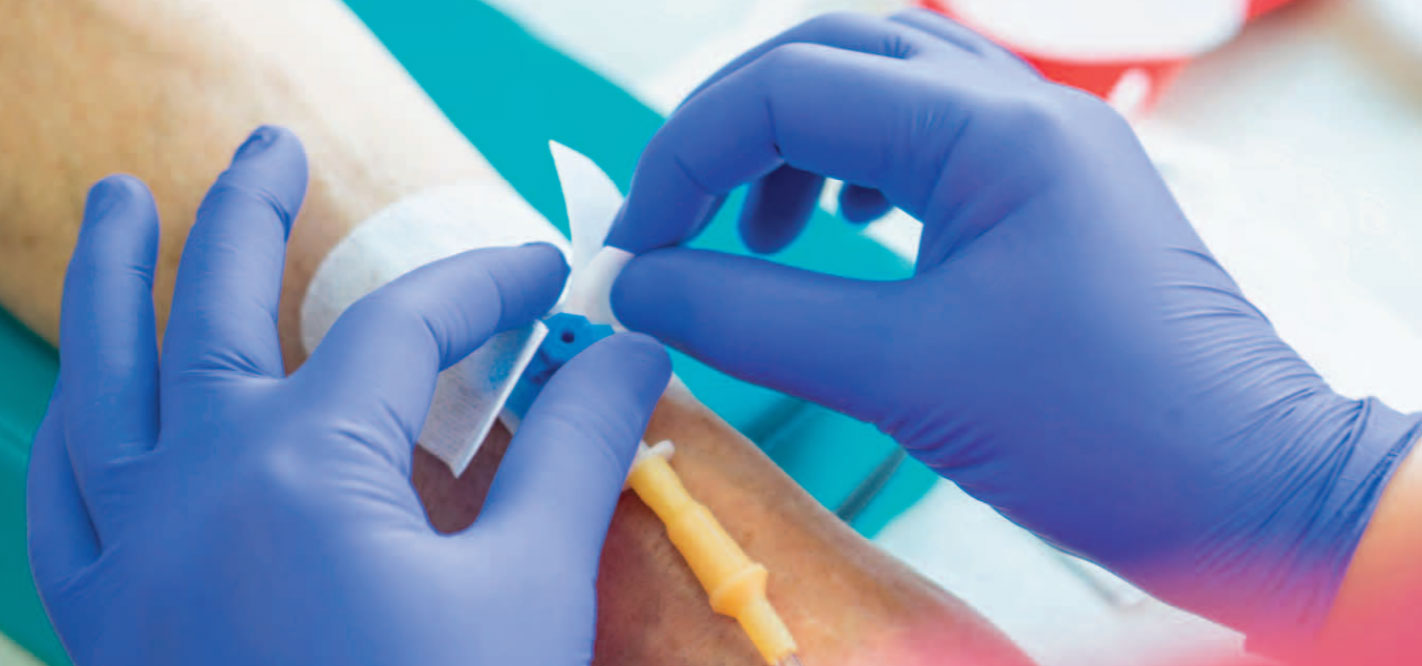
- Disinfect I.V. access ports before administration by scrubbing with the disinfectant used in your organization; allow to dry for 10 seconds.
- Follow the recommended rate for administration and use a watch or clock with a second hand for timing to ensure accuracy.
- Remember to contact a pharmacist if you have a drug information question and a nurse educator or advanced practice nurse if you have a drug administration question.
For organizations
- Conduct a gap analysis of I.V. push medication practices using the ISMP Gap Analysis Tool (GAT) for Safe IV Push Medication Practices, available at ismp.org/resources/gap-analysis-tool-safe-iv-push-medication-practices. Another resource is the Ready-to-administer (RTA) MedSafety Form from The Joint Commission, available at rtamedsafety.com.
- Provide I.V. push education for clinicians during orientation and annually.
- Ensure policies and procedures follow national standards and guidelines.
- Encourage clinicians to report errors and near-misses, which can help identify areas for improvement.
Sources: Infusion Nurses Society. Infusion therapy standards of practice. 2016;39(1S):1-169.
Institute for Safe Medication Practices (ISMP). Part II: Survey results suggest action is needed to improve safety with adult IV push medications. 2018. ismp.org/resources/part-ii-survey-results-suggest-action-needed-improve-safety-adult-iv-push-medications
ISMP safe practice guidelines for adult IV push medications. 2015. ismp.org/sites/default/files/attachments/2017-11/ISMP97-Guidelines-071415- 3.%20FINAL.pdf
ant4-Fresenius-IV-Push-325a