Follow this protocol to improve patient outcomes.
- Performing cardiopulmonary resuscitation on an adult who’s had cardiac surgery can significantly damage the heart.
- The cardiac advanced life support (CALS) protocol is designed for use with patients after cardiac surgery (except those who’ve had pulmonary surgery) to correct reversible causes of arrest quickly.
- If necessary, immediate resternotomy can be performed by a trained physician.
Nurses working in cardiovascular intensive care units (CVICUs) or mixed ICUs care for patients after heart surgery, including those who’ve undergone cardiac valve repairs, aneurysm repairs, pacemaker insertion, and coronary artery bypass grafting. They receive advanced cardiac life support (ACLS) training to prepare them to care for patients with clinical complexities.
The American Heart Association (AHA) ACLS guidelines build on the foundation of basic life support skills with more in-depth coverage of pharmaceuticals and the use of automated external defibrillators (AEDs). These guidelines require critical review and modification to ensure optimum resuscitation for patients who’ve undergone cardiac surgery or have implanted devices such as intra-aortic balloon pumps (IABPs) or mechanical circulatory support.
Opioid overdose cardiopulmonary emergencies
Strategies for improving survivorship after sudden cardiac arrest
Emergency cardiac drugs: Essential facts for med-surg nurses
AHA guidelines
When performing cardiopulmonary resuscitation (CPR) on an adult, the AHA recommends “pushing hard and fast.” The guidelines recommend initiating CPR by interlocking the hands between the nipples of the patient and then depressing the chest at least 2 inches at a rate of 100 to 120 beats per minute, allowing the chest to recoil fully. The AHA goes on to advise attaching an AED as soon as possible, and administering 1 mg of epinephrine every 3 to 5 minutes if the patient remains pulseless.
However, following ACLS guidelines, along with CPR or external cardiac massage (ECM), can significantly damage a heart that’s just undergone surgery. Valve dehiscence can occur along with cardiac chamber or graft rupture, vascular dissections, or myocardial lacerations. Cardiac Advanced Life Support (CALS) offers an alternative.
Cardiac Advanced Life Support (CALS)
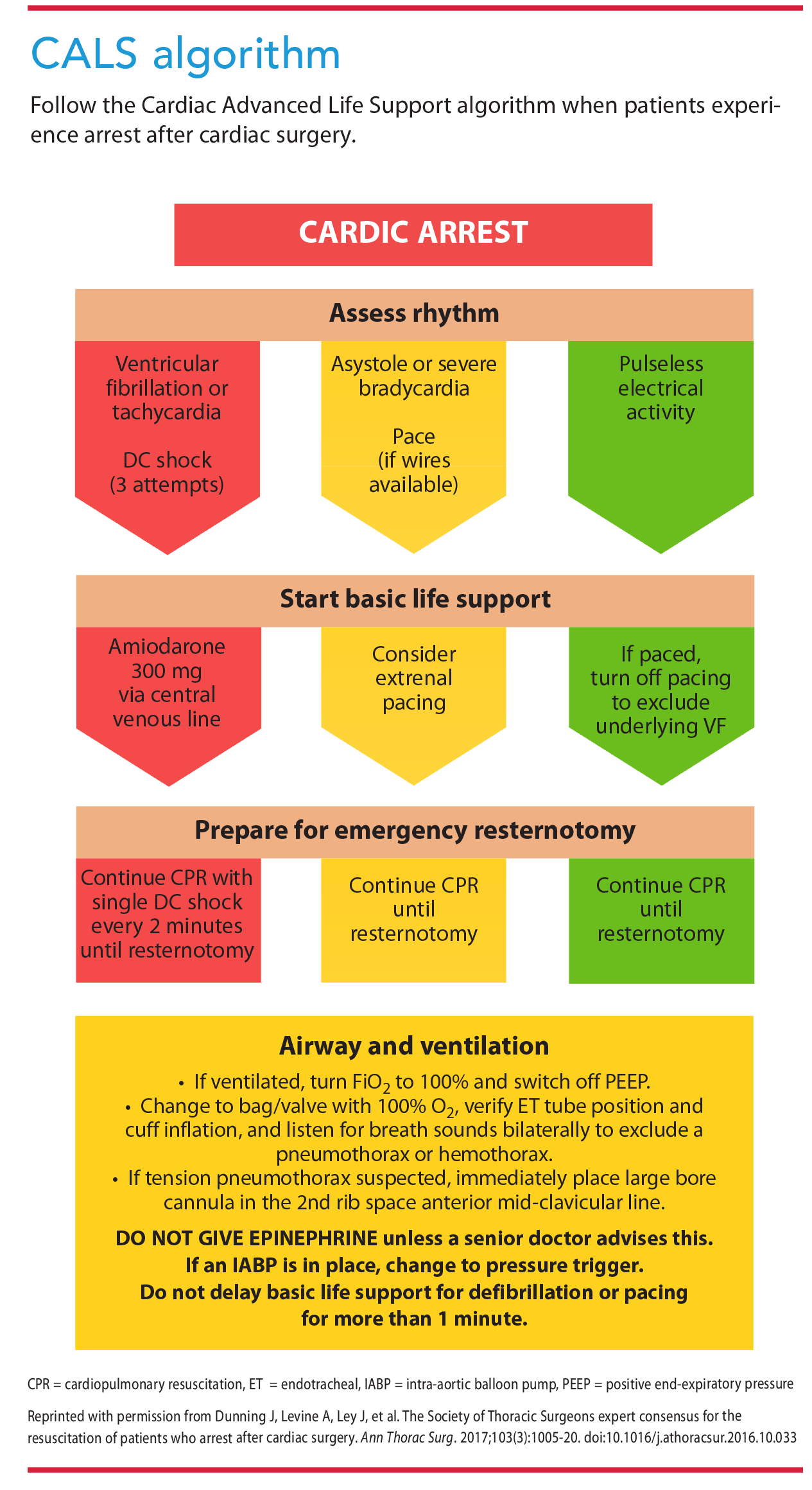
The CALS protocol, a cardiopulmonary resuscitation method developed for CVICUs, is designed for use with any patient who’s had cardiac surgery, excluding those who’ve had pulmonary surgery. The goal of CALS is to correct any reversible causes of arrest quickly, followed, if necessary, by immediate resternotomy performed by a trained physician. When the absence of cardiac pressure and respiratory waveforms is identified, a first responder should initiate the CALS protocol. ECM can be deferred for 1 minute to allow for rapid defibrillation or pacing as indicated. ECM should be performed immediately on patients with pulseless electrical activity (PEA) arrests. (See CALS algorithm.)
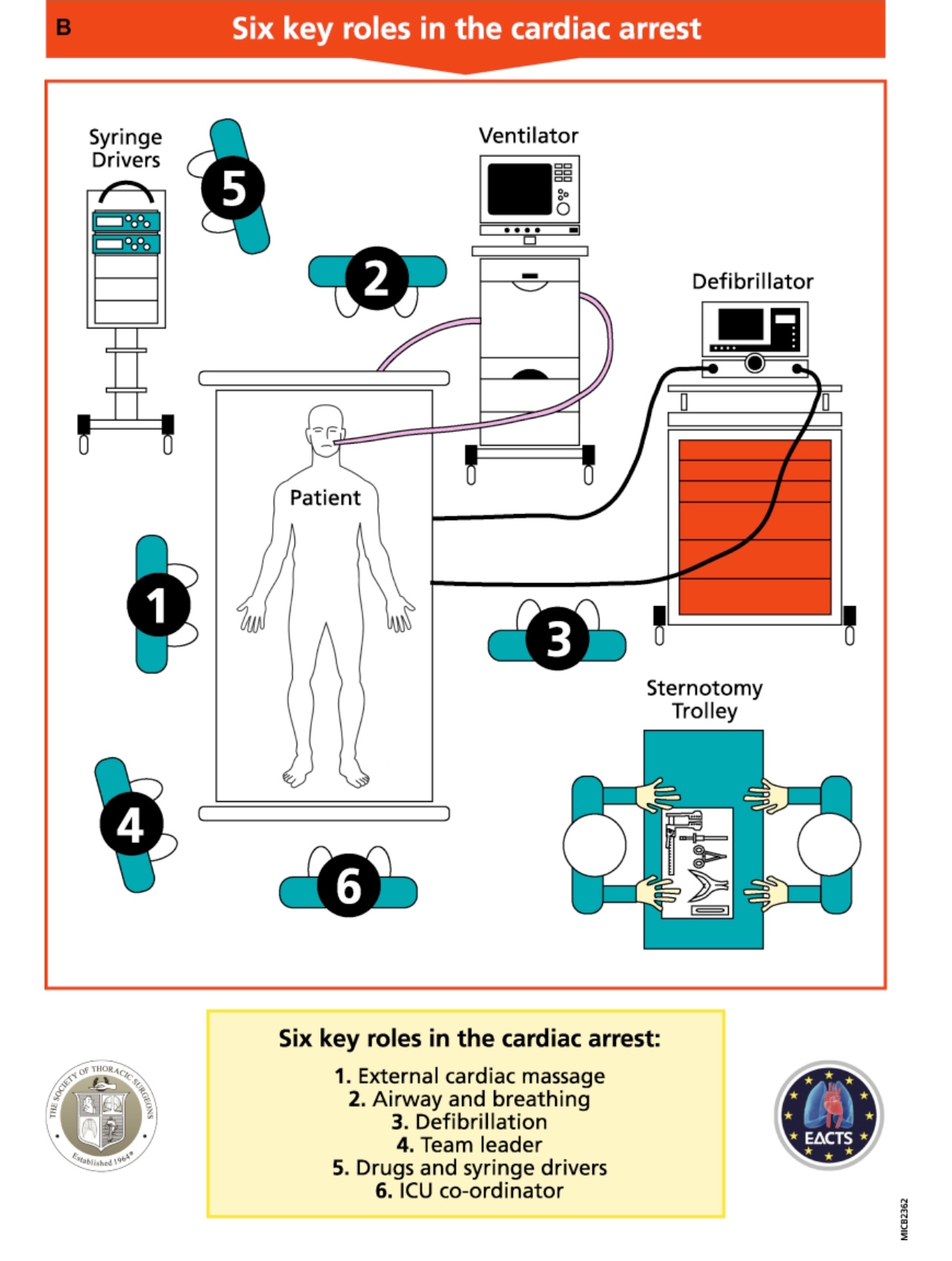
The CALS protocol advises against full-dose epinephrine or 1 mg epinephrine for these patients to avoid significant hypertension, which could potentially harm fresh cardiac grafts. Resternotomy is required for 20% to 50% of patients who’ve had a cardiac arrest after cardiac surgery, but it’s recommended only on postoperative Days 1 to 10 because pericardial adhesions might result in more harm when reopening the patient. (See Key roles.)
Key roles
The Cardiac Advanced Life Support approach identifies the following six key roles for clinical staff:
External cardiac massage
- One team member performs external cardiac massage (ECM) at a rate of 100 to 120 compressions per minute to reach a mean arterial pressure (MAP) of 60 mmHg. Note: Switching team members every 2 minutes helps to ensure high-quality ECM.
Airway and breathing
- This team member increases oxygen supplementation to 100% and removes positive end-expiratory pressure. They assess the patient’s airway and breathing to determine if the cause of arrest is a pneumothorax, hemothorax, or an endotracheal tube issue.
Defibrillation
- This team member attaches the defibrillator and administers shocks and pacing as required. They also ensure internal defibrillators are connected during resternotomy.
Team leader
- This senior team member conducts resuscitation management and ensures protocols are followed correctly.
Drugs and syringe drivers
- One team member stops all infusions and administers other drugs as indicated by the team leader.
ICU coordinator
- One member of the ICU staff coordinates other facilitating activities to help supplement resuscitation.
Cardiac arrest causes and actions
Many cardiac arrest conditions are reversible. Determining and treating the cause is essential to optimizing patient outcomes. (See Reversible causes.)
Reversible causes
Reversible causes of cardiac arrest include the following:
- Hydrogen ion excess (acidosis)
- Hypoglycemia
- Hypokalemia/hyperkalemia
- Hypothermia/hyperthermia
- Hypovolemia
- Hypoxia
- Tamponade
- Tension pneumothorax
- Thrombosis (pulmonary embolus/myocardial infarction)
- Toxins
According to the Society of Thoracic Surgeons, 0.7% to 8% of patients who’ve had cardiac surgery suffer from cardiac arrest. Ventricular fibrillation (VF), the most common cause, accounts for 25% to 50% of cardiac arrests. VF treatment begins with defibrillation—up to three shocks at 150 joules. ECM isn’t recommended or beneficial before administering defibrillation. If a return of circulation isn’t achieved after three shocks, begin ECM until a resternotomy can be performed.
For cardiac arrest caused by asystole or severe bradycardia, pace the patient. If the patient is attached to an epicardial pacemaker, set it to dual-chamber pacing at 80 to 100 beats per minute, or press the emergency pacing button. Attach a transcutaneous pacemaker to the patient if epicardial wires aren’t in place.
If an intrinsic or pacing rhythm is noted on the ECG, but the patient is pulseless, begin ECM and prepare for resternotomy. If neither a defibrillator nor pacing is feasible within 1 minute, start ECM at a rate of 100 to 120 beats per minute. Modify cardiac depressions to produce a mean arterial pressure of 60 mmHg or greater.
Causes of arrest that can lead to VF, bradycardia, or asystole after cardiac surgery include hypovolemia or bleeding, graft or valve failure, low cardiac output, and tamponade. Due to the nature of cardiac surgery and the typical patient population, coagulopathy can occur as a result of residual heparin effects, platelet dysfunction, clotting factor deficiencies, or extensive fibrinolysis. Treatment with appropriate clotting factors or protamine can help reverse these causes.
Surgical bleeding can require surgical re-exploration. Bleeding most commonly occurs at sternal wire sites or anastomotic graft sites. Assess chest tube output for excess bleeding, and follow facility guidelines for provider notification and surgical re-exploration. Typically, chest tube output of >400 mL in the first hour, >200 mL in 2 consecutive hours, or >100 mL per hour in 4 consecutive hours should prompt re-exploration. Keep in mind that chest tube misplacement can mask bleeding and cause fluid collection. If a patient who’s hemodynamically unstable shows low to no chest tube output, notify a provider to consider ordering a chest x-ray or transesophageal echocardiogram.
Cardiac tamponade, a surgical emergency, occurs when an accumulation of fluid or blood in the pericardial sac compresses the heart and reduces ventricular filling and cardiac output. Assess the patient for the Beck’s triad (the three classic signs of cardiac tamponade): hypotension, venous distention, and muffled heart sounds. Arrhythmias or cardiac arrest can be the first signs of tamponade in these patients.
Airway and breathing
If the patient is still on a ventilator when a cardiac arrest occurs, change the ventilator settings to an oxygen concentration of 100% and remove positive end-expiratory pressure to help decrease intrathoracic pressure and increase venous return. If the patient isn’t intubated, use a bag-valve mask at a ratio of two breaths to 30 compressions. Consider causes of respiratory arrest such as hemothorax, pneumothorax, or improper endotracheal tube (ETT) placement by taking the following steps:
- Check ETT placement.
- Listen for air leaks and check cuff inflation.
- Look for fog formation in the ETT.
- Palpate the trachea for proper position.
- Assess for symmetrical chest rise and fall.
- Auscultate lung sounds for bilateral air entry.
- Check capnography readings for appropriate end-tidal carbon dioxide.
If a tension pneumothorax is identified, alert the physician. An appropriate provider will place a large-bore cannula into the second intercostal space at the midclavicular line.
Medications
If a cardiac arrest occurs, pause all vasoactive, ionotropic, chronotropic, and sedation infusions to remove any variables that could interfere with successful resuscitation. Epinephrine and vasopressin aren’t recommended during resuscitation of patients after cardiac surgery. If necessary, only trained providers familiar with these medications should administer them. No evidence exists to support the use of atropine in patients experiencing bradycardia or asystole post-cardiac surgery. Amiodarone should be given to patients with VF or pulseless ventricular tachycardia after three unsuccessful defibrillation attempts. Start with a 300 mg bolus, followed by a 150 mg dose, then an infusion of 900 mg over 24 hours.
Sternal wound infection or sepsis can occur after a resternotomy, so administer an antiseptic washout and additional antibiotics as ordered.
Assistive devices
IABPs help decrease the workload of the heart, allowing it to pump blood more efficiently. However, these devices can present an obstacle in the event of cardiac arrest. Placed in the proximal descending aorta, just below the origin of the left subclavian artery, the balloon inflates during diastole and deflates during systole. It can be triggered by the ECG or arterial pressure waveforms. PEA can be difficult to identify in patients with an active pacemaker because the ECG will show a paced rhythm whether or not a patient has a pulse. Using the ECG trigger mode on an IABP will cause the balloon to pump in conjunction with the ECG reading, but using the pressure trigger mode will cause it to pump in conjunction with the patient’s actual pulse/pressure. For this reason, it’s more accurate when performing CPR to use the pressure trigger mode with counterpulsation set at maximal augmentation during PEA when the patient has no pulse or pressure.
Some patients who’ve undergone cardiac surgery have implanted mechanical circulatory support, such as a left ventricular assist device to help pump blood from the left ventricle to the rest of the body. These devices display pump flow, which can help identify loss of blood flow. Follow the CALS algorithm with these patients: defibrillate patients in VF, pace those in asystole, and start ECM on patients in PEA.
Resternotomy
If all causes of cardiac arrest have been considered and all attempts at non-ECM have been exhausted, resternotomy is recommended within 5 minutes to perform internal cardiac massage. Compared to ECM, internal cardiac massage performs better at achieving cardiac resuscitation by increasing coronary/end-organ blood flow and spontaneous circulation. ICUs should have a resternotomy kit or cart on standby to expedite this procedure. At a minimum, the kits should include a scalpel, a wire cutter, a heavy needle holder, a sternal retractor, and a sucker.
Special considerations
Use the CALS resuscitation guidelines for patients undergoing heart, heart–lung, or double-lung transplants through a sternotomy. If patients come out of surgery with an open chest with packing and then experience cardiac arrest, perform ECM at the midpoint of the chest over the packs of gauze. Assess arterial pressure waveforms for effectiveness as less force may be required.
Therapeutic hypothermia
Therapeutic hypothermia is an effective treatment for patients who are comatose after cardiac arrest and undergo a return of spontaneous circulation (ROSC). The goal is to maintain a core temperature between 32° C and 36° C (89.6° F and 96.8° F) for 24 hours. Lowering core temperatures helps to reduce cerebral metabolic demand by reducing the rate of oxygen consumption. Therapeutic hypothermia also reduces cerebral edema by preventing inflammatory cytokines from penetrating the blood brain barrier. If poor cerebral blood flow is assumed after resuscitation and achieving ROSC, consider therapeutic hypothermia.
Apply an evidence-based approach
Although cardiac arrest after cardiac surgery is uncommon, all clinical staff caring for these patients should be aware of and familiar with the CALS approach for resuscitation. Consider all causes of cardiac arrest and treat them accordingly. Nursing units caring for these patients should have protocols in place and the necessary supplies to act with haste in the event of cardiac arrest. For efficiency, assign a team of six with key roles to help ensure successful resuscitation. All hospitals have protocols for cardiac resuscitation, but they also should consider the evidence-based CALS approach.
Madison Carpenter is a post-anesthesia care unit nurse at UCI Health in Orange, California.
American Nurse Journal. 2023; 18(7). Doi: 10.51256/ANJ072314
References
Applefield D, Krishnan S. Protamine. StatPearls. January 8, 2023. ncbi.nlm.nih.gov/books/NBK547753
Brand J, McDonald A, Dunning J. Management of cardiac arrest following cardiac surgery. BJA Educ. 2018;18(1):16-22. doi:10.1016/j.bjae.2017.11.002
Dunning J, Levine A, Ley J, et al. The Society of Thoracic Surgeons expert consensus for the resuscitation of patients who arrest after cardiac surgery. Ann Thorac Surg. 2017;103(3):1005-20. doi:10.1016/j.athoracsur.2016.10.033
Khan TM, Siddiqui AH. Intra-aortic balloon pump. StatPearls. June 3, 2022. ncbi.nlm.nih.gov/books/NBK542233
Merchant RM, Topjian AA, Panchal AR, et al. Part 1: Executive summary: 2020 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation. 2020;142:S337-57. doi:10.1161/CIR.0000000000000918
Oliver TI, Sadiq U, Grossman SA. Pulseless electrical activity. StatPearls. January 23, 2023. ncbi.nlm.nih.gov/books/NBK513349/
Pacha HM, Soud M, Alraies MC. Beyond Beck’s Triad: A rare cause of cardiac tamponade and hemoptysis. Ochsner J. 2018;18(3):271-3. doi:10.31486/toj.17.0081
Key words: resuscitation, CPR, cardiac surgery, cardiac advanced life support, resternotomy