Reduce complications with thorough nursing assessment and close monitoring.
Takeaways
- Pulmonary embolism risk severity varies.
- Patients at high risk require close monitoring and an individualized prevention plan.
- Patient and family need education includes pharmacologic and nonpharmacologic treatment.
CNE: 1.75 contact Hours
Learning Objectives
- Describe risk factors for and how patients might present with pulmonary embolism (PE).
- Discuss nursing implications to consider when caring for patients with suspected or confirmed PE.
- Identify medication management options and associated safety concerns for patients with PE.
The authors and planners of this CNE activity have disclosed no relevant financial relationships with any commercial companies pertaining to this activity. See the last page of the article to learn how to earn CNE credit.
Expiration: 3/1/25
Pulmonary embolism (PE) occurs when a clot (thrombus) that formed somewhere in the body dislodges, travels through the right side of the heart to the lungs and blocks one of the arteries that supplies blood flow to the lungs. The vascular obstruction can cause sudden death, ventilation-perfusion (V/Q) mismatching, pulmonary hypertension, pulmonary infarction, hemodynamic compromise, right-sided heart failure, and multiple organ failure. Some patients with PE exhibit clinical manifestations such as hypoxemia and chest pain; however, some are asymptomatic, emphasizing the need for a high index of suspicion and close surveillance of patients at high risk. (See PE by the numbers.)
PE by the numbers
According to the European Society of Cardiology, pulmonary embolism (PE) is the third most common cause of acute cardiovascular death (after myocardial infarction and stroke). A high mortality rate persists despite advances made in treatment.
- The exact incidence of PE is unknown, but according to Turetz and colleagues, an estimated 600,000 cases occur in the United States each year, resulting in approximately 100,000 deaths annually.
- PE incidence is rising over time in developed countries, likely due—at least in part—to increasingly sedentary lifestyles.
- PE disproportionally affects the elderly population. Individuals over age 70 years are six times more likely to develop PE than those less than 44 years.
- If left untreated, PE mortality is significant and can be as high as 30%, with 10% of patients with acute PE succumbing to sudden death.
Etiology
Most PE results from deep vein thrombosis (DVT), which occurs when a blood clot that develops in one of the deep veins of the body, usually the leg, breaks free from the vessel wall and travels through the right side of the heart into the lungs. Keep in mind that PE also can consist of fat, air, bacteria, amniotic fluid, or other substances. Clinical presentation, diagnostic approaches, and physiologic alterations are essentially the same, but treatment varies based on embolus composition.
PEs can occur in multiple conditions, including healthy states such as pregnancy and inactivity as well as pathologic states that involve blood vessel injury and surgery. They can be acute or chronic, and treatment and complications may vary in duration and intensity.
Risk factors
Several factors increase an individual’s risk of developing PE. Virchow’s triad (mechanisms underlying the risk factors) include venous stasis, hypercoagulability, and injury to the thin inner lining (tunica intima) of the vessel. Identifying known risk factors and educating patients about lifestyle adjustments can help decrease individual risk. (See PE risk factors.)
PE risk factors
Pulmonary embolism (PE) risk factors are stratified as high, moderate, and low. Patients with high-risk factors require close nursing assessment and monitoring and a standardized screening protocol.
High risk
- Age >60 years
- Atrial fibrillation/flutter
- Cancer
- Erythropoiesis-stimulating agents
- Fracture of the leg or hip
- Genetics
- History of heart attack or stroke
- Hypercoagulable states/thrombophilia
- Major surgery
- Major trauma
- Severe COVID-19 symptoms
Moderate risk
- Blood transfusion
- Family history of embolism
- Infection
- Long trips
- Sedentary lifestyle
- Spinal cord injury
Low risk
- Estrogen or testosterone therapy
- Obesity
- Post-partum period
- Pregnancy
- Smoking
Pathophysiology
Sisson and colleagues describe PE as a partial or complete occlusion of the pulmonary artery or one or more of its branches by an embolus or thrombus. The pulmonary vascular occlusion results in impaired gas exchange and circulation. When the embolus lodges in the pulmonary vasculature, several neurohormonal and inflammatory mediators—including serotonin, catecholamines, histamine, leukotrienes, angiotensin II, thromboxane, and toxic oxygen free radicals—are released. These substances cause widespread vasoconstriction, which impedes blood flow to the lungs and increases pulmonary arterial pressure (pulmonary hypertension). The vasoconstriction results in right ventricular heart failure because the right ventricle has to pump against higher-than-normal pulmonary vascular resistance, which dilates the right ventricle and decreases contractility and cardiac output. Subsequent imbalance between oxygen demand and supply further decreases cardiac contractility, which may lead to hemodynamic instability and cardiogenic shock. (See PE pathophysiology.)
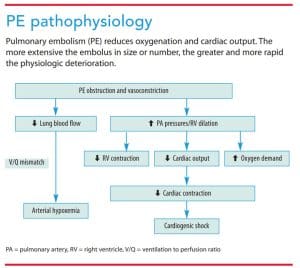

The reduction or cessation of blood flow to lung tissue creates a V/Q mismatch. The blocked vessel can’t carry blood to areas of alveoli that are ventilated, so less oxygenated blood is brought to the left side of the heart, leading to hypoxemia and tissue hypoxia. Although the mechanisms aren’t fully understood, hypoxemia is enhanced by impaired production of surfactant in the alveoli deprived of blood supply, leading to atelectasis.
Clinical manifestations and evaluation
In most cases, PE clinical presentation is nonspecific and depends on the number and size of emboli and the number and size of blood vessels occluded. Patients may be asymptomatic or experience critical hemodynamic instability. The most common symptoms include dyspnea and pleuritic or substernal chest pain, but patients also may experience tachycardia, cough, hemoptysis, cyanosis, and syncope. (See Clinical manifestations.)
Clinical manifestations
Pulmonary embolism clinical manifestations vary based on the size and number of emboli. For example, a massive pulmonary embolus that occludes the pulmonary artery will result in loss of consciousness and cardiac arrest, whereas a small embolus may manifest as tachypnea and tachycardia.
- Altered level of consciousness/mentation
- Anxiety
- Compensatory respiratory alkalosis, followed by respiratory acidosis if embolus persists
- Cough
- Crackles on chest auscultation
- Cyanosis
- Dyspnea
- Fever
- Hemoptysis
- Hypotension
- Hypoxemia
- Metabolic acidosis for hemodynamic compromise
- Pleural friction rub
- Pleuritic chest pain
- Syncope
- Tachycardia
- Tachypnea
Diagnosis requires evaluation of risk factors, patient history, and predisposing factors. Diagnosing non-thrombotic PE can be challenging because micro-emboli may not be detectable with computed tomography (CT). Patients with high clinical suspicion of PE should be evaluated with a CT pulmonary angiogram, which allows visualization of a thrombus or blocked blood flow distal to the thrombus. Patients who lack access to an angiogram or for whom this test is contraindicated should be evaluated with a V/Q scan, which permits assessment of pulmonary arterial blood flow and alveolar ventilation distribution. Although lower extremity doppler ultrasound can’t diagnose PE, Thompson and colleagues recommend it for patients with contraindications for both angiogram and V/Q scan. Ultrasound detects reduced blood flow, but doesn’t provide evidence of the cause.
A chest X-ray can help rule out pleural effusion, pneumonia, and other pulmonary disorders. Keep in mind that chest X-rays usually are unremarkable for the first 24 hours before atelectasis develops. A 12-lead ECG may show right heart strain, tachycardia, and nonspecific ST segment and T wave changes. Arterial blood gas analysis may reveal hypoxemia with respiratory alkalosis secondary to tachypnea to compensate for the hypoxemia; however, hypoxemia and metabolic acidosis are present with a massive PE that significantly reduces perfusion. Results from a basic metabolic panel can be used to evaluate renal function for the contrast dye used for a pulmonary angiogram and for later selection of pharmacologic agents, and a complete blood count may reveal leukocytosis from the inflammatory response to PE. Serum D-dimer may show the products of thrombus degradation (according to Thompson and colleagues, levels over 500 ng/dL increase the likelihood of PE). (See Diagnostic tests.)
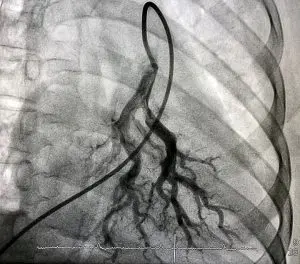

Diagnostic tests
Diagnostic tests for pulmonary embolism are selected based on the severity of the clinical manifestations and the risk factor level.
- Arterial blood gases
- Basic metabolic panel
- Chest X-ray
- Complete blood count
- Computed tomography pulmonary angiogram (shown in photo)
- D-dimer, coagulation studies
- History and physical examination
- 12-lead electrocardiogram
- Ventilation/perfusion scan
Complications
PE complications can be life-threatening and include pulmonary hypertension, pulmonary infarction, right-sided heart failure, multi-organ dysfunction, and, if the clot is large enough to obstruct a pulmonary artery, sudden death. Lack of oxygen to the lung results in lung tissue death (pulmonary infarction). Because the right ventricle normally pumps blood against low pressure in the pulmonary vasculature, the workload of pumping against high pulmonary arterial resistance (pulmonary hypertension) causes the right ventricle to fail, leading to potentially catastrophic cardiac decompensation as reduced cardiac output impairs blood flow to vital organs.
If a clot persists and the patient continues to have significant respiratory distress that results in hypoxemia and hypercapnia, acute respiratory failure may occur. Pulmonary hypertension caused by a persistent clot may result in scarring and chronic thromboembolic pulmonary hypertension. Increased right ventricular workload, from acute or chronic PE, may cause arrhythmias.
Pharmacologic treatment
Pharmacologic PE treatment includes anticoagulants, thrombolytics, and antiplatelet therapy.
Anticoagulants, administered either intravenously or subcutaneously, are first-line PE treatment. Rather than breaking up a clot, anticoagulants prevent them from enlarging. Bleeding is the main complication.
A thrombolytic may be administered if the PE is large and life-threatening and the existing clot must be dissolved to avoid hemodynamic and respiratory compromise. Although thrombolytics increase bleeding risk, they can be lifesaving if a patient is hemodynamically compromised. Thrombolytics are administered locally near the clot via a long catheter or systemically via I.V.
Antiplatelet therapy may be initiated for patients who are at high risk for developing a PE during a planned surgical procedure, particularly if the procedure is of long duration. Antiplatelet medications prevent platelet aggregation in the blood and subsequent clot formation. Evidence shows that these medications significantly reduce the risk of fatal and non-fatal PEs. (See Pharmacologic treatment options.)
Pharmacologic treatment options
Understanding the mechanisms of action and the parameters used to monitor pulmonary embolism (PE) treatment aids safe patient care and prevents treatment complication. Prophylactic and therapeutic ranges vary, with the latter being higher. Nursing considerations common to all PE pharmacologic treatment include monitoring for bleeding from over-anticoagulation and clotting from under-anticoagulation.
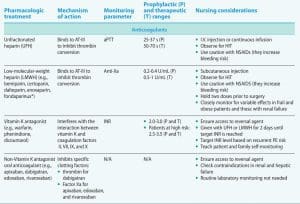



*Fondaparinux is not an LMWH; however, its half-life and selective Factor Xa inhibition are similar to LMWH.
aPTT = activated partial thromboplastin time, AT-III = antithrombin-III, HIT = heparin-induced thrombocytopenia, INR = international normalized ratio, NSAIDs = nonsteroidal anti-inflammatory drugs, rt-PA = recombinant tissue plasminogen activator, s=seconds
Nonpharmacologic treatment
Patients who have contraindications or are unresponsive to pharmacologic therapy may require clot removal. The least invasive option is suction thrombectomy, in which a catheter is threaded through a major vessel to the clot and the clot is removed. If this treatment isn’t an option, surgical embolectomy may be required.
Placement of an inferior vena cava filter, preferably a retrievable one, is reserved for patients with recurrent PE despite appropriate management with medication, those who are unable to take anticoagulants and don’t require surgical intervention, and patients with active bleeding. Jaber and colleagues reported improved outcomes with these filters, including reductions in 3-month mortality rate and recurrent PE. When used for patients who subsequently develop active bleeding from concurrent anticoagulation, the European Society of Cardiology guidelines recommend removing the filter and re-initiating anticoagulation when the bleeding stops and the patient is stable.
Providers can use oxygen therapy to treat hypoxemia from the V/Q mismatch of perfusion to under-ventilated lung regions that results from redistribution of pulmonary blood flow. Some organizations have success using extracorporeal membrane oxygenation (ECMO) as a bridge for diagnosis and treatment of life-threatening, massive PE. ECMO pumps oxygenated blood to the body, bypassing the failing heart and lungs. Keeling and colleagues recommend that PE centers have ECMO readily available for treatment. Click here to learn more about ECMO.
Post-treatment evaluation and prevention
Patients receiving acute thrombolytic therapy require close observation to monitor for clinical manifestations of allergic and anaphylactic reactions. Chronic thromboprophylaxis may be used for months or years to prevent subsequent PE. Bleeding risk varies by drug, dosage, and individual patient factors. The provider may use a bleeding risk score (such as the Pulmonary Embolism Severity Index) to determine the duration, type, and dose of chronic anticoagulation. Successful treatment requires periodic bleeding risk reassessment, hepatic and renal function tests, and patient adherence with and tolerance of the prescribed regimen.
For immobilized patients, mechanical prophylaxis with intermittent pneumatic compression can help prevent recurrent PE, but low patient adherence has been reported by many authors, including Dunn and Ramos. The devices may disrupt sleep and discourage mobility in bed. According to Dunn and Ramos, the devices are misapplied easily and present risks for skin breakdown.
Anti-embolism stockings (thromboembolic deterrent hose) increase venous blood flow velocity, decreasing venous stasis and the risk of developing DVTs. Appropriate pressure distribution to prevent blood pooling in the lower extremities requires proper sizing of the stockings. The elastane in the fabric loses its compressibility and effectiveness over time, so stockings should be replaced monthly or more frequently as needed to maintain their preventive effect.
Nursing implications
Sudden onset and high mortality of PE make continuous surveillance of at-risk patients imperative. Nurses should know the risk profile for each patient and plan surveillance accordingly. For many patients who die from PE, the diagnosis is made post-mortem, so the condition should be at the top of the differential diagnosis list, along with myocardial infarction, when a patient has acute chest pain, dyspnea, or a cardiopulmonary arrest.
According to Rivera-Lebron and colleagues, because PE treatment options will vary based on provider experience and skill, medical specialty, organization, region, and available resources, protocols may be different within and across clinical settings. To account for these variations, individualize nursing surveillance, assessment, and patient education.
Patients with PE who have hemodynamic compromise may require fluid resuscitation. Monitor them closely for fluid overload effects on a poorly functioning right ventricle. Maintain good communication with the prescribing provider about the patient’s blood pressure, heart rate, respiratory rate, jugular vein distention, and clinical manifestations of perfusion so that fluid resuscitation can be balanced to enhance cardiac output vs. cardiogenic shock.
Manage pain in patients who are symptomatic and take steps to assuage fear and anxiety. Provide close surveillance (face-to-face or electronic), ensure the call bell is within reach, and offer patient and family education.
Education about acute or chronic anticoagulation should be appropriate to the setting and include discussion of the risk of PE recurrence, the importance of following the therapeutic regimen, and the consequences of missing medication doses. Provide information about monitoring for bleeding and clinical manifestations of thrombus formation; foods, drugs, and herbs to avoid as appropriate for the prescribed anticoagulant; and lifestyle modifications to prevent or minimize medication adverse effects and PE recurrence. Although most pulmonary pathology will resolve after about 3 months, advise patients and their families to report new onset of dyspnea or stamina deterioration to their healthcare provider.
The authors are affiliated with the research department at the University of Texas Health Science Center at Houston, Cizik School of Nursing. Jamie L. Gilley, Amanda Holmes, and Evaline Sims are PhD students. Sandra K. Hanneman is the Jerold B. Katz Distinguished Professor for Nursing Research.
References
Dunn N, Ramos R. Preventing venous thromboembolism: The role of nursing with intermittent pneumatic compression. Am J Crit Care. 2017;26(2):164-7. doi:10.4037/ajcc2017504
Haberecker M, Schwarz EI, Steiger P, et al. Autopsy-based pulmonary and vascular pathology: Pulmonary endotheliitis and multi-organ involvement in COVID-19 associated deaths. Respiration. 2021;:1-11. doi:10.1159/000518914
Haugen N. Oral anticoagulants: Pharmacologic management update. Am Nurse Today. 2019;14(6):12-17.
Jaber WA, Fong PP, Weisz G, et al. Acute pulmonary embolism: With an emphasis on an interventional approach. J Am Coll Cardiol. 2016;67(8):991-1002. doi:10.1016/j.jacc.2015.12.024
Keeling WB, Sundt T, Leacche M, et al. Outcomes after surgical pulmonary embolectomy for acute pulmonary embolus: A multi-institutional study. Ann Thorac Surg. 2016;102(5):1498-1502. doi:10.1016/j.athoracsur.2016.05.004
Konstantinides SV, Meyer G, Becattini C, et al. 2019 ESC Guidelines for the diagnosis and management of acute pulmonary embolism developed in collaboration with the European Respiratory Society (ERS). Eur Heart J. 2020;41(4):543-603. doi:10.1093/eurheartj/ehz405
Rivera-Lebron B, McDaniel M, Ahrar K, et al. Diagnosis, treatment and follow up of acute pulmonary embolism: Consensus practice from the PERT Consortium. Clin Appl Thromb Hemost. 2019;25:1076029619853037. doi:10.1177/1076029619853037
Sisson TH, Claar D, Chesnutt MS, Prendergast TJ. Pulmonary disease. In: Hammer GD, McPhee SJ, eds. Pathophysiology of Disease: An Introduction to Clinical Medicine. 8th ed. New York, NY: McGraw Hill; 2019; 245-307.
Thompson BT, Kabrhel C, Pena C. Clinical presentation, evaluation, and diagnosis of the nonpregnant adult with suspected acute pulmonary embolism. UpToDate. January 14, 2021.
Turetz M, Sideris AT, Friedman OA, Triphathi N, Horowitz JM. Epidemiology, pathophysiology and natural history of pulmonary embolism. Semin Intervent Radiol. 2018;35(2):92-8. doi:10.1055/s-0038-1642036














1 Comment.
excellent overview outlined in the article