Nursing assessments and recognition of risk factors aid prompt recognition and treatment.
Sepsis is a life-threatening condition that occurs when the body’s response to infection causes organ injury. According to the Centers for Disease Control and Prevention, it affects approximately 1.7 million adults each year in the United States and is a contributing factor to as many as one-third of all hospital deaths. Paoli and colleagues report that sepsis is the most expensive medical condition to treat in hospitalized patients, accounting for $24 billion in healthcare costs each year.
If overlooked or mistreated, sepsis can lead to septic shock, organ failure, and possibly death. Early recognition is crucial to halt its progression, improve patient outcomes, decrease mortality rates, and reduce healthcare costs. This article will review the latest definition of sepsis and how to recognize its early manifestations and identify resources available to help initiate timely treatment.
Defining sepsis
Sepsis isn’t a specific disease but rather a collection of symptoms resulting from an infection. It evolves over time with worsening characteristics as the patient continues to decompensate. No specific tests are available to diagnose sepsis, so clinicians must piece together the evidence.
The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3) formally defines the condition as a life-threatening organ dysfunction caused by a dysregulated host response to infection. This definition emphasizes the emergency at hand, rather symptoms. However, the clinical definition used in practice is two or more systemic inflammatory response syndrome (SIRS) symptoms accompanied by suspected or presumed infection.
Recognizing at-risk patients
The first step in recognizing sepsis is to identify those most at risk, including adults 65 years or older, people with weakened immune systems, and those with co-morbidities such as cancer, diabetes, lung disease, and kidney disease. Vaccination status also should be considered when determining risk. Because pneumonia is a common cause of sepsis, those who aren’t vaccinated against Streptococcus pneumoniae and influenza are more vulnerable.
Patient environment is another contributing risk factor. Those residing in long-term care facilities or admitted to the intensive care unit (ICU) are at increased risk for healthcare-acquired infections (HAIs).
When gathering pertinent patient information, be aware of these risk factors before starting your physical assessment.
Head-to-toe approach for sepsis detection
Your thorough patient assessment should include neurologic, respiratory, cardiovascular, GI, and genitourinary function. Also evaluate the patient’s skin and any invasive catheters. (See Head-to-toe approach.)
Knowing risk factors and conducting a thorough assessment can help you identify sepsis quickly.
| Risk Factors | |
| |
| Assessments | |
| Neurologic |
|
| Respiratory |
|
| Cardiovascular |
|
| GI |
|
| Genitourinary |
|
| Skin |
|
| Invasive |
|
| Labs and imaging | |
| |
bpm = beats per minute, BUN = blood urea nitrogen, CT = computed tomography, HR = heart rate, ICU = intensive care unit, MAP = mean arterial pressure, RR = respiration rate, WBC = white blood count
Neurologic assessment
Acute brain dysfunction in sepsis is associated with increased morbidity and mortality. SIRS contributes to a decrease in micro-perfusion to the brain, which leads to tissue hypoxia and may cause irreversible brain damage if not addressed quickly. Although neurologic symptoms may be caused by myriad disease processes (seizures, stroke), a thorough neurologic assessment can help determine a patient’s prognosis.
Start by assessing the patient’s orientation and ability to follow commands. If friends or family members are at the bedside, ask about the patient’s baseline functional and cognitive abilities before admission and note any discrepancies in your assessment. Assess the patient frequently. If the patient becomes more lethargic, confused, or restless, an underlying issue may require further investigation.
Respiratory assessment
The respiratory effects of sepsis may be vague, with initial symptoms such as tachypnea (>20 breaths per min) or dyspnea. During auscultation, you may hear crackles (suggesting fluid in the lungs) or rhonchi (indicating secretions). A patient with a weak productive cough may require assistance with expectoration via suctioning. Also assess the color, consistency, and frequency of sputum. As you monitor the patient for worsening conditions, such as respiratory failure, communicate your findings to the multidisciplinary team.
As sepsis worsens, respiratory failure may lead to acute respiratory distress syndrome (ARDS) caused by inflamed bronchioles and purulent secretions that occlude the airway and collapse the alveoli. Alveolar collapse reduces gas exchange and causes hypercapnia (pCO2 >50 mmHg) and hypoxemia (pO2 <60 mmHg). Sepsis-induced ARDS is associated with high mortality and accounts for approximately 210,000 U.S. cases annually.
If a patient becomes hypercapnic and exhibits signs of altered mental status, dyspnea, or cyanosis, intensified oxygen delivery devices such as hi-flow nasal cannula, bilevel positive airway pressure, or (in worst-case scenarios) mechanical ventilation may be required.
For patients who are mechanically ventilated with an artificial airway, consider how long they’ve been intubated. Patients who’ve been intubated for 4 days or more are at risk for ventilator-associated events such as pneumonia and sepsis. Although no specific diagnostic criteria exist, an abnormal respiratory assessment may indicate the need for chest radiography.
Cardiovascular assessment
Cardiovascular assessment is the nurse’s most objective tool to determine a patient’s perfusion status. SIRS causes systemic vasodilation, which results in tachycardia (heart rate >100 beats per minute) and hypotension (systolic <90 mmHg, diastolic <60 mmHg). These cardinal signs of dedecompensation require immediate attention. Interventions such as passive leg raise or fluid challenges may indicate patient acuity and level of care needed. Mean arterial pressure (MAP) is another crucial indicator of perfusion status. The Surviving Sepsis Campaign created a treatment guideline for patients with sepsis and identified a MAP ≥65 mmHg to be adequate for supporting organ function.
Another critical factor is body temperature. A temperature >100.4° F (>38°C) or <96.8° F (<36°C) may indicate sepsis. Monitor the patient’s temperature carefully, and report values that are out of range.
GI assessment
GI symptoms may be subtle and include nausea, vomiting, and diarrhea. Depending on the origin of sepsis, the GI system may be more vulnerable to secondary complications such as superinfections or stress ulcers.
Carefully assess the patient’s oral cavity for redness or lesions; then work your way down to the abdomen and inspect for distention, tenderness, or pain. Ask about changes in eating habits, bowel patterns, or stool characteristics. Review the patient’s medication list for antibiotics, which may eliminate natural flora from the GI tract and allow opportunistic infections such as Clostridium difficile or Candida albicans to proliferate.
Genitourinary assessment
Urinary tract infections (UTIs) are the most common type of HAI and may progress to sepsis. In the hospital setting, approximately 75% of UTIs are associated with a urinary catheter. For patients with a urinary catheter, start your genitourinary assessment by determining how long the catheter has been in place, and reviewing its indication. Ensure that the catheter is properly secured to the patient’s thigh and that the drainage bag is below the patient. Monitor urine output for a minimum of 30 mL/hr, which indicates adequate kidney perfusion.
For patients without a urinary catheter, ask if they’re experiencing urinary frequency or burning sensations when voiding. Ask about urine color, clarity, and odor. Characteristics such as hematuria, cloudiness, or malodor may indicate infection. Next, palpate the patient’s suprapubic region and flanks for tenderness, which may indicate bladder or kidney inflammation. Assess the patient’s urine output; a decrease in frequency or amount may be a sign of acute kidney injury. Noting these abnormalities as an acute finding may trigger a diagnostic work up.
Skin assessment
The skin is the largest system in the body and serves as a barrier to pathogens. A break on the skin surface may allow a localized infection to become systemic. Surgical incisions, pressure injuries, and venous or arterial stasis ulcers all have the potential to become infected and septic. Assess the patient’s body thoroughly, look for openings, and monitor for discharge, which may indicate a festering wound. Measure the length, width, and depth of any wound you find, and take pictures per your organization’s policy. If erythema is present from cellulitis, trace it with a marker to monitor its progress.
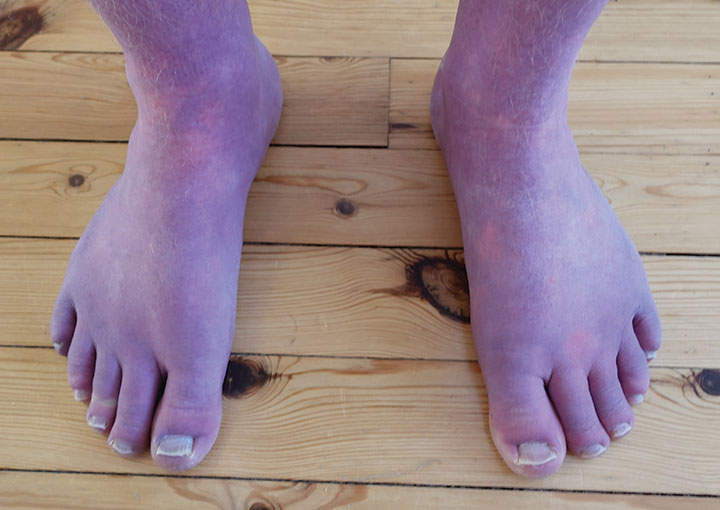
If the patient’s skin is intact, early manifestations of sepsis will be subtle. As the body begins to compensate by shunting blood away from skin and into vital organs, the lack of perfusion may result in cool and clammy skin. As it evolves, patches of skin discoloration will develop as it transitions to a mottled appearance if perfusion isn’t restored.
Invasive catheter assessment
Central venous devices (including central venous catheters, hemodialysis catheters, and implanted ports) can increase a patient’s risk for a central line-associated bloodstream infection (CLABSI), a common HAI that accounts for approximately 250,000 cases annually, according to Haddadin and colleagues. Redness, swelling, pain, and purulent drainage may be localized at the insertion site.
In addition to your physical assessment, determine how long the catheter has been in place, the date of the last dressing change, and the indication for its use. If you suspect CLABSI, notify the provider and suggest catheter removal. A culture of the tip may be necessary to determine treatment options.
Imaging studies and laboratory tests
An x-ray, ultrasound, or computed tomography scan can detect infiltrates or inflammation, helping to screen patients for sepsis while ruling out other disease processes.
Laboratory tests are critical for putting together all of the information. Blood tests such as a complete blood count, basic metabolic panel (BMP), and arterial blood gas (ABG) will help clarify patient acuity. Although the Sepsis-3 committee has disputed using an abnormal white blood count (>12,000/mm3 or <4,000/mm3) to identify sepsis, it’s still clinically significant to support an indirect sepsis diagnosis.
A BMP can identify fluid status, electrolyte imbalances, and kidney function. Sepsis affects fluid shifts and tissue perfusion, so values such as sodium, potassium, blood urea nitrogen, and creatinine may be out of range. This signifies decompensation of the body as sepsis emerges.
An ABG is essential to determining the patient’s respiratory status. It measures critical values such as carbon dioxide and lactate. Carbon dioxide >45 mmHg or lactate >2 mmol/L signifies an acid–base imbalance that implies a metabolic disease such as sepsis or organ dysfunction. Monitor these laboratory trends, and connect your physical findings to help you recognize sepsis.
One of the most important tests to follow-up are specimen cultures. Most healthcare organizations collect cultures as soon as sepsis is suspected. Although it may take 48 to 72 hours for results, a positive finding will determine the underlying pathogen and its susceptibility to anti-infectives. The results guide appropriate infection management and treatment. For example, a patient may require special isolation precautions, such as contact or droplet. In addition, specific pathogens may require mandatory reporting to the department of health or the Centers for Disease Control and Prevention for statistical tracking. Monitoring specimen cultures will protect healthcare staff, visitors, and the community. (See Quick assessment.)


Win the race
Sepsis is a medical emergency that requires immediate treatment to reduce morbidity and mortality. Nurses and providers must race against time as the patient’s body begins to decompensate. Your thorough assessment skills and clinical judgement are critical to early detection and prompt treatment. Incorporate all available data (blood tests, imagining studies, and cultures) to further support your suspicions. Consider using the qSOFA tool if you’re concerned about poor patient outcomes and encourage your facility to integrate a sepsis algorithm into the electronic health record to help manage patient information more efficiently and effectively. (See Take advantage of the EHR.)


Taking steps when you suspect a patient is at high risk for sepsis can halt its progress. Ultimately, you’ll improve patient outcomes, decrease length of stay, and reduce overall healthcare costs.
Chun Chung Tse is an assistant professor of nursing at Queensborough Community College, City University of New York in New York City.
References
Amland RC, Hahn-Cover KE. Clinical decision support for early recognition of sepsis. Am J Med Qual. 2019;34(5):494-501. doi:10.1177/1062860619873225
Bourcier S, Joffre J, Dubée V, et al. Marked regional endothelial dysfunction in mottled skin area in patients with severe infections. Crit Care. 2017;21(1):155. doi:10.1186/s13054-017-1742-x
Centers for Disease Control and Prevention. Healthcare-associated infections: Catheter-associated urinary tract infections (CAUTI). Updated October 16, 2015.
Centers for Disease Control and Prevention. Nursing homes and assisted living (long-term care facilities [LTCFs]). Updated June 22, 2020.
Centers for Disease Control and Prevention. Sepsis: Clinical information. December 7, 2020.
Centers for Disease Control and Prevention. Ventilator-Associated Event (VAE). January 2021.
Center for Disease Control and Prevention. What is Sepsis? Updated January 27, 2021.
Dasgupta S, Das S, Chawan NS, Hazra A. Nosocomial infections in the intensive care unit: Incidence, risk factors, outcome and associated pathogens in a public tertiary teaching hospital of Eastern India. Indian J Crit Care Med. 2015;19(1):14-20. doi:10.4103/0972-5229.148633
Englert JA, Bobba C, Baron RM. Integrating molecular pathogenesis and clinical translation in sepsis-induced acute respiratory distress syndrome. JCI Insight. 2019;4(2):e124061. doi:10.1172/jci.insight.124061
Finkelsztein EJ, Jones DS, Ma KC, et al. Comparison of qSOFA and SIRS for predicting adverse outcomes of patients with suspicion of sepsis outside the intensive care unit. Crit Care. 2017;21(1):73. doi:10.1186/s13054-017-1658-5
Haddadin Y, Annamaraju P, Regunath H. Central line associated blood stream infections. StatPearls. Updated December 14, 2020.
Jiang J, Yang J, Mei J, Jin Y, Lu Y. Head-to-head comparison of qSOFA and SIRS criteria in predicting the mortality of infected patients in the emergency department: A meta-analysis. Scand J Trauma Resusc Emerg Med. 2018;26(1):56. doi:10.1186/s13049-018-0527-9
Joynes E. More challenges around sepsis: Definitions and diagnosis. J Thorac Dis. 2016;8(11):E1467-9. doi:10.21037/jtd.2016.11.10
Levy MM, Evans LE, Rhodes A. The Surviving Sepsis Campaign Bundle: 2018 update. Intensive Care Med. 2018;44(6):925-8. doi:10.1007/s00134-018-5085-0
Novosad SA, Sapiano MRP, Grigg C, et al. Vital Signs: Epidemiology of sepsis: Prevalence of health care factors and opportunities for prevention. MMWR Morb Mortal Wkly Rep. 2016;65(33):864-9. doi:10.15585/mmwr.mm6533e1
Paoli CJ, Reynolds MA, Sinha M, Gitlin M, Crouser E. Epidemiology and costs of sepsis in the United States—An analysis based on timing of diagnosis and severity level. Crit Care Med. 2018;46(12):1889-97. doi:10.1097/CCM.0000000000003342
Singer M, Deutschman CS, Seymour CW, et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA. 2016;315(8):801-10. doi:10.1001/jama.2016.0287
Sonneville R, de Montmollin E, Poujade J, et al. Potentially modifiable factors contributing to sepsis-associated encephalopathy. Intensive Care Med. 2017;43(8):1075-84. doi:10.1007/s00134-017-4807-z
Yazan H, Annamaraju P, Regunath H. Central line associated blood stream infections. StatPearls. August 11, 2021.














1 Comment.
Serum Lactate or Lactic Acid levels are increasingly being used to identify / monitor impending sepsis. This is another key component to monitor for impending sepsis along with other labwork identified in the article.